Research Article
Physiological and Biochemical Adaptation of Triticum vulgaris L. to pH Stress by Hormonal Application
Department of Botany, Faculty of Science, Ain Shams University, Abbassia, Cairo, Egypt
Plants differ in their responses to pH conditions. Most crop plants grow well in soil that is neutral, mildly acidic or mildly basic. The dependence of the structure and ionization states of many of the molecular constituents of the cell insures that cellular processes are sensitive to pH[1]. Acidic stress induces changes in the cellular biochemistry and physiology of the whole plant. Biological effects are numerous and complex and include visible symptoms of injury (chlorosis and or, necrosis, reduction in root and shoot growth)[2,3] and invisible effects such as presence of high concentrations of H+ and Al+3 in exchangeable form[4], effects on membrane and ion transport properties[5], reduce photosynthesis, altered water balance and variation of several enzyme activities[6].
In addition, acid stress is accompanied by changes in endogenous hormones which in turn perform changes in the related physiological process. Exogenous application of hormones can stimulate resistance against unfavourable environmental factors and improve plant growth under stress[7]. However, plants can become acclimated to various stress factors by developing tolerance against the stress factor that induced the change[6,8].
The goal of the present work is the using of exogenous application of the hormone (IAA) as a tool to ameliorate the harmful effect of pH stress on the growth and to improve the yield productivity and quality of Triticum vulgaris L. plants; as it is one of the most important economic crops for human nutrition in Egypt and all over the world. The work is done also to show the possible adaptive mechanism under pH stress.
Plant material: Caryopsis of wheat (Triticum vulgaris L. cv. Sakha 8 and Sids 1) were brought from the Agricultural Research Center (ARC), Giza, Egypt. This work was carried out at Botany Department, Faculty of Science, Ain Shams University, Cairo, Egypt during the growing season 2001/2002.
The grains were surface sterilized with 1% sodium hypochlorite for 20 min, then rinsed with distilled water several times. A range of pH treatment was used (2-11) as a preliminary experiment, the highly acidic and slightly alkaline showed lethal effects were neglected. Then three pHs were chosen (4.5, 7 and 8.5). Equal groups of grains (Sakha 8 and Sids 1) were soaked in phosphate buffer alone at different pH (4.5, 7 and 8.5) or in combination with IAA at (1 and 10 μM) for different times (5, 10 and 15 h). After 10 day of growth in plastic boxes, the suitable conditions were chosen for further study. New sets of grains of cv. Sods 1 were soaked in phosphate buffer at pH (4.5, 7 and 8.5 for 15 min and similar groups were prepared in combination with IAA (1 μM). The control was distilled water at pH 6.5. The grains were sown in pots containing garden soil having pH at 8., then irrigated at 80% water saturation capacity, then left to grow at the natural day conditions. Samples of 35 days were collected for morphological and physiological analysis. Another plants were left until the yield maturation for analysis. Parameters were statistically analyzed according to SAS Program[9].
Estimation of chlorophyll: Chlorophyll a and b contents were determined according to Todd and basler[10]. Chlorophyll stability to heat (57°C, 30 min) was assessed according to Murty and Majumder[11]. Chlorophyll stability index was modified as follows:
![]()
Estimation of sugars: Soluble sugars were extracted following the method adopted by Homme et al.[12]. Sugar’s free residue (insoluble sugars) were extracted with perchloric acid following the method described by Hassid and Newfeld[13]. Both the soluble and insoluble sugars were determined with the anthrone reagent[14].
Estimation of proline: Free proline was determined according to Bates et al.[15] method.
Estimation of element content: Total nitrogen and phosphorus were determined in the acid digested sample (1 ml 50% H2SO4 and 1ml 30% perchloric acid), using Berthelot reaction[16] and modified Fiske-Subborow method[17]. Potassium, calcium and magnesium were estimated in the same extract by the atomic absorption.
Protein analysis: Soluble proteins were determined according to the method described by Bradford[18].
Electrophoretic protein profile was analyzed by SDS-Page technique[19]. Data were analyzed and identified by gel documentation system (GDS) which comparing polypeptide maps which include the use of band intensity, molecular weight and the rate of mobility of each polypeptide with standard markers using Gel Proanlyzer Version 3 Media Cyberene Tice Imaging Experts Software.
IAA: Indole acetic acid (IAA) was extracted and separated following the method adopted by Shindy and Smith[20]. Identification and quantification was carried by HPLC using the method described by Shindy and Smith[20].
Estimation of polyamine content: A certain weight of shoot plant samples was homogenized in a Waring Blender in 5% perchloric acid (PCA) according to the method described by Shalaby[21]. The blended mixture was centrifuged and adjusted to a final volume of 250 ml with 5% PCA. The mixture was made alkaline with NaOH and extracted with n-butanol / chloroform (1:1 v/v). The combined organic phase was extracted with 0.02 N HCl and the aqueous extract was dried. Saturated sodium bicarbonate solution (0.5%) was added to the residue of the danylated derivatives of the amines. Then, 1 ml of dansyl chloride reagent (500 mg in 100 ml acetone) was added while using a vortex mixer. They were left to stand for 1 h, thereafter, the dansylamines were extracted by adding water and several portions of diethylether. The combined ether extracts were evaporated to dryness and the residue was re-dissolved in acetonitrite. Polyamine [Putresine (put) {H2N(CH2.NH2)}, Spermidine(spd) {H2N.(CH2)3.NH.(CH2)4.NH2} and Spermine (spm) {H2N.(CH2)3 (CH2) 4NH.CH2}3 ] levels were determined using high performance liquid chromatography as described by Smith And Davies[22].
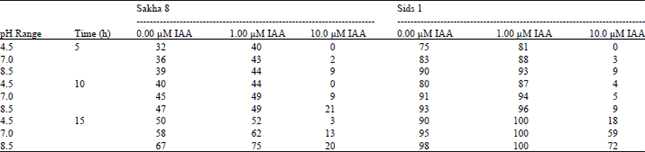
The success of germination is the first step towards the plant survival. Some investigations were carried out to show the effect of pH on the germination process[23]. The percentage of germination of control wheat grains as well as those of subjected to various treatments are illustrated in Table 1. Data show direct relationship between the time of soaking wheat grains in different pH values and the percentages of germination. It is also observed that the percentages of germination of Triticum vulgaris cv. Sids 1 are greater than those of Triticum vulgaris cv. Sakha 8, thus the Sids 1 cultivar is more tolerant than Sakha 8. In addition, soaking in acidic pH (4.5) decreased % of germination than those of either neutral (pH 7) or alkaline (pH 8.5).
| Table 1: | Effect of pre-soaking of Triticum vulgaris L. (cultivars Sakha 8 and Sids1) grains in phosphate buffer at different pH values and IAA concentrations for different times on the percentage of germination |
 | |
| Table 2: | Effect of pre-soaking of Triticum vulgaris L. (cultivars Sakha 8 and Sids 1) grains in phosphate buffer at different pH values and IAA concentrations for different times on the growth parameters of 10 days old seedlings. Each value is a mean of 10 samples |
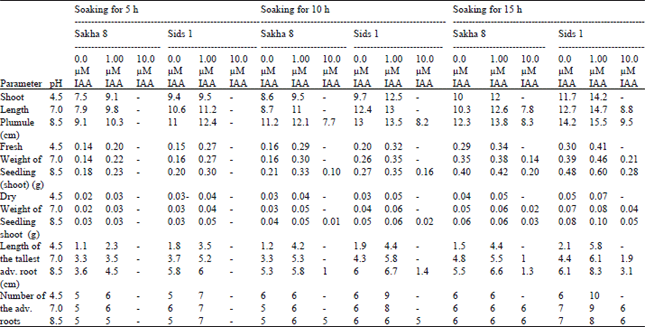 | |
Moreover, addition of 10 μM of IAA (higher concentration) showed inhibition of the germination; this may due to ethylene formation. In contrast, the lower concentration 1 μM of IAA has stimulator effect on such percentages. It is suggested that IAA applied at the optimal level for growth can reduce the inhibitory action of acidic pH on elongation, plastic and elastic extensibility and water absorption leading to increasing the percentage of germination, while supra-optimum concentration didn’t oblish the inhibitory action. The growth characters of the seedlings (Table 2) showed similar pattern as that of the germination. This result is in agreement with that obtained by Kershengal and Rogozhin[23] who found that germination of wheat occurred between pH 5.0-11.0, with peaks at about 6.0 and 9.0. Moreover, seed germination of rap seed germinated at pH 3.0-11.0 was highest at pH 6.0 and lowest at pH 3.0[24]. From the above results, the further study was carried out using Triticum vulgaris cv. Sids 1, 15 h time of soaking (control groups) and 1 uM IAA (treated group).
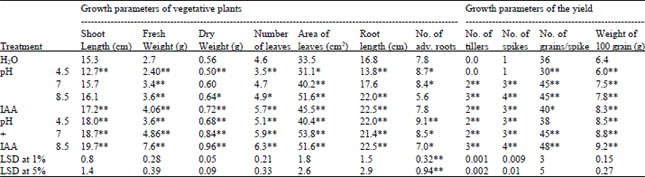
The growth characteristics of wheat plant expressed as shoot length, number of internodes and leaves, area of leaves, fresh and dry weights of shoot are illustrated in Table 3. It is apparent that, the low pH (4.5) generally significantly decreased all the above characters as being compared to those of the corresponding controls. These results are confirmed by those obtained by Bu and Xing[25], who concluded that the leaf area, dry weight of shoot and fruit yield of cucumber plant increased at pH 8 rather than pH 5 or pH 10. Also, Saito[26] deduced that seedling growth, dry matter and length of young or intermediate rice seedling (were raised in boxes placed on soil with pH 5, 6.8 or 8) decreased as soil pH increased. In addition, Fageria and zimermann[27] found that increasing soil pH enhanced dry matter accumulation in roots and tops of wheat.
| Table 3: | Effect of pre-soaking of Triticum vulgaris L. cv. Sids 1 grains in phosphate buffer at different pH values and IAA on the growth parameters of 35 days old plants (vegetative stage) and the resulted yield. Each value is a mean of 10 samples |
 | |
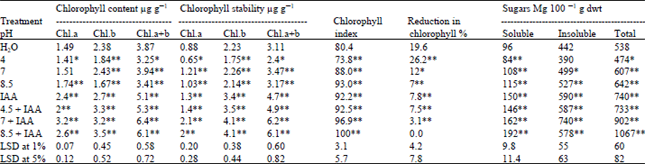
| Table 4: | Effect of soaking of Triticum vulgaris L. cv. Sids 1 grains in phosphate buffer at different pH values and IAA on the chlorophyll contents, chlorophyll stability, chlorophyll index and sugar contents of the 35 days shoots. Each value is a mean of 3 samples |
 | |
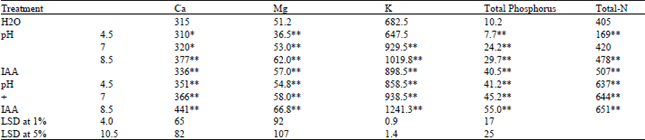
| Table 5: | Changes in the element contents in shoots of Triticum vulgaris L. cv. Sids 1 exposed to various treatments of phosphate buffer at different pHs and IAA for 35 days. Each value is a mean of 3 samples. Values are expressed as mg 100-1 g dry weight |
 | |
Moreover, Kallenback et al.[28] added that the dry matter of three birds foot trefoil (Lotus corniculatus) genotypes grown at soil pH levels of 5, 6,7 and 8 in green houses was lower at low soil pHs than at a higher pH, this was due to the high uptake of calcium and phosphorous. Moreover, number of leaves and dry matter of leaves and stem of hairy beggar-tick (Bidens pilsoa) were reduced at 3.5[29]. Cheng Can et al.[24] obtained similar result in rap seedling grown at pH 3-11. Seedling growth was highest at pH 6 and lowest at pH 3. Also, Marler[30] found that plant dry weight of waimanalo, in the pH 3 treatment was reduced compared with plants at other pHs (3-9). Ila-ava et al.[31,32] obtained similar result using 4 sweet potato cultivars.
Lastly, Gudrupa et al.[33] found that Sedun rubroticum plants grown at pH values up to 6.0 had extremely limited increase in stem length within a growth period of 8 weeks. At higher pH, increase in stem length was associated with increase in both the number of internodes as well as internode length. Most probably, the effect of pH on shoot elongation is related to pH-induced changes of availability of nutrients[34]. Indirectly this is supported by the fact that root growth was more significantly affected by substrate pH as compared with shoot elongation. As an alternative, pH itself may act as an endogenous signal[35].
Data in Table 3 show that, the application of exogenous IAA can reduce the reduction in plant growth characters at acidic condition (pH 4.5), while increased such characters at pHs (7 and 8.5). These results were confirmed by Botia et al.[35] who found that elongation of etiolated hypocotyl segments (5 mm long) of lupinus (Lupinus albus cv. Multolupa) was stimulated by acid pH (4.5 vs 6.5) and by IAA for periods of up 4 h.
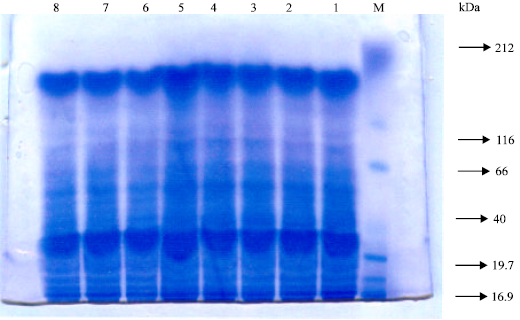 | |
| Fig. 1: | Changes in protein banding patterns of Triticum vulgaris L. cv. Sids 1 of grains treated with phosphate buffer at different pH values and IAA concentrations. Lane M: protein molecular weight marker, lane 1: control (H2O), lane 2: pH 4.5, Lane 3: pH 7, lane 4: pH 8.5, lane 5: IAA, lane 6: pH 4.5 + IAA, lane 7: pH 7 + IAA and lane 8: pH 8.5 + IAA |
After this time, the segments were unable to grow further. In the presence of an optimal IAA concentration (10 μM), acid pH increased the growth rate but had no effect on final growth. With sub-optimal IAA (0.1 μM), however, acid pH increased growth in a more than additive way, suggesting a synergistic action between the two factors. It is suggested that this synergism could be explained by the increased IAA uptake and decarboxylation seen at an acid pH. Vanadate inhibited growth and IAA uptake and decarboxylation suggesting that this inhibitor probably inhibits growth not only by decreasing ATPase-mediated acidification but also by decreasing H+-dependent IAA uptake from the apoplast. It is suggested that this dependence of IAA uptake on ATPase may be mediated by apoplastic acidification. The amount of IAA decarboxylated increased when the assay conditions favoured the growth of segments indicating that IAA could be destroyed by decarboxylation during the auxin-induced growth.
Concerning the chlorophyll contents and chlorophyll stability to heat(CSI), chl.a and b contents were decreased progressively with increasing the acidity compared with the control (pH 6.5). Moreover, the reduction in chlorophyll and chlorophyll index was accompanied with significant and non-significant reductions in soluble and insoluble sugar contents, respectively (Table 4). Drazkiewicz[36] and Borrell et al.[37] obtained similar results. In addition, Cheng Can et al.[24] found that chlorophyll and soluble sugar contents were highest at pH 6 comparing to pH 3 in growing rap seedling. Moreover, dipping plant leaves of 7 tree species of pH range (2-6) for up to 48 h, the chlorophyll content of 4 species decreased after immersion at the lower pH (2) but remained almost constant at the other pHs[38].
The reduction in chl.a and b contents in response to acidic pH may be due to impaired uptake of essential elements such as Mg and Fe, damage of photosynthetic apparatus or enhancement of chlorophyll degradation by chlorophyllase[36]. Furthermore, these alterations in the pigment contents reflect the changes in the composition and structure of the light harvesting complex[39] leading to a disturbance in the chloroplast architecture[40]. Borrell et al.[37] added that the loss of chlorophyll of the plant leaves under stress is accompanied by reduction in the levels of thylakoid protein D1 and D2 and cytochrome F as well as stromal rubisco large subunit. In contrast, in the present work, IAA application increased both the chlorophyll contents and their stability, in plants grown at acidic pH and consequently their sugar contents showed significant increases.
Osmotic adjustment and turgor potential maintenance are known to be accomplished by the uptake of ions[41]. The changes in the absorption of mineral in response to different pH values of wheat plants are shown in Table 5. It is clear that the acidic pH (4.5) decrease the uptake of element, while the neutral or alkaline pH, these contents significantly increased.
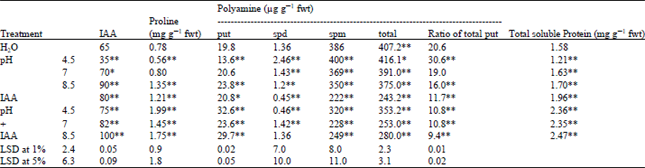
| Table 6: | Changes in IAA, proline, polyamines and total soluble protein contents in shoots of Triticum vulgaris L. cv. Sids 1 exposed to various treatments of phosphate buffer at different pHs and IAA for 35 days. Each value is a mean of 3 samples |
 | |
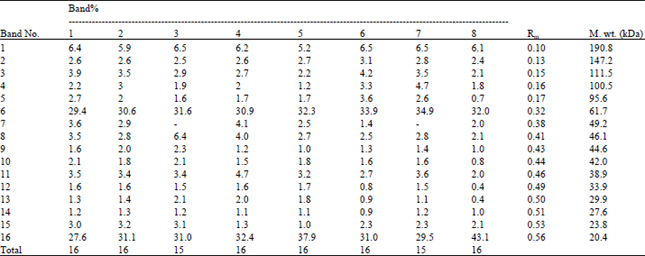
| Table 7: | Comparative analysis of relative concentrations (band %), molecular weight (M.wt.) and mobility rate (Rm) of the different types of protein bands in shoots of Triticum vulgaris L. cv. Sids 1 exposed to various treatments of phosphate buffer at different pHs and IAA for 35 days |
 | |
| Lane 1: control (H2O), Lane 2: pH 4.5, Lane 3: pH 7, Lane 4: pH 8.5 Lane 5: IAA, Lane 6: pH 4.5 + IAA, Lane 7: pH 7 + IAA, Lane 8: pH 8.5 + IAA | |
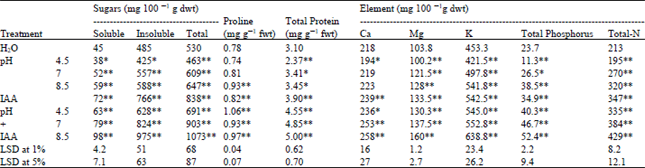
| Table 8: | Effect of pre-soaking of Triticum vulgaris L. cv. Sids 1 grains in phosphate buffer at different pH values and IAA on the sugar, proline, total protein and element contents of the produced yield. Each value is a mean of 3 samples |
 | |
However, treatment with IAA in the present work could minimize the reduction of the element uptake. Phytohormones may participate in stress perception signaling and possibly initiate a cascade of stress-induced responses[42]. In addition, osmotic adjustment and turgor potential maintenance are known to be accomplished by the uptake of ions[41]. These results herein are in agreement with those obtained by Obara et al.[29], Fageria and Zimmermann[27], Kershengol and Rogozhin[23] and Fageria and Baligar[43]. Soil pH affects the membrane and ion transport properties in cereals[5].
Obara et al.[29] found that the contents of P, S, Ca and Mg in leaves were reduced at pH 3.5. In addition, Zoldos et al.[44] concluded that there was marked decrease in the K+ efflux at decreased pH of the medium, however, an increase loss of K+ from the roots was observed.
Moreover, soil acidity cause insolubility of Phosphorus, potassium and other minerals and deficiency of calcium and magnesium[45]. So, the effect of pH on the absorption of major elements was small and insignificant but the effect on minor nutrient absorption was substantial in grown gerbera[46]. Calcium concentration in leaves and stems of Lotus corniculatus was highest at soil pH 8. Also, phosphorus concentration in leaves was lowest at acidic low soil pHs and tend to increase with increasing pH[28]. In general, nutrient uptake , especially of K, increased with increasing pH from 4.5 to 8[31]. In acidic soils (in most humid areas with rainful), calcium and other exchangeable bases are leached from the surface and replaced by hydrogen ions[45].
In general, the reduction in mineral uptake under acidic stress may be due to altering the absorption of ions at the root epidermal plasma membrane[4]. Moreover, at pH less than 5, H+ ions could be passively transported into the germinated wheat grains previously soaked for 15 h in 0.1 M buffer solutions at pH 3.0-13.4 or distilled water and germinated on filter paper moistened with these solutions, causing proportional acidification of the medium, but hydroxyl ions scarcely penetrated the grains, even at a negative pH gradient of 4.3 units[23].
When plants subjected to various form of stresses, their endogenous hormonal level are undergone various and rapid changes[47]. In this experiment, the auxin content was markedly decreased (Table 6). The reduction in IAA content in the acidic treatment may be due to the inhibition of biosynthesis of certain enzymes responsible for the indole auxin compounds or directing the activity of these enzymes towards formation of other compounds rather than tryptophan, the precursor of indole compounds or increase IAA oxidase activity[48]. In addition, soluble protein and the free amino acid proline contents were reduced under acidic condition (Table 6). Gadallah and Sayed[7] obtained similar results. The reduction may be due to inhibition of their de novo synthesis and/or increased degradation in acid stressed plants. In contrast, soluble proteins and proline in the present work were high in IAA treated plants. This suggest that proline would be a good candidate as a second messenger derived from the primary signals. It acts as a free radical scavenger[49] and limit the cytoplasm acidification[50]. It also protect plant tissues against stress by acting as a storage compound for both carbon and nitrogen sources, as osmo-compatible solute, protecting cytoplasmic enzymes and cellular structure[51].
Changes in the polyamine level estimated in wheat shoots subjected to various treatments are recorded in Table 6. Putresine, Spermidine and Spermine were detected in control plants, pH stress (pH 4.5) induced 80% and 3.6% in both spd and spm levels coupled with 31% fold decrease in put content. Meanwhile 11.8 and 9% decrease in spd and spm contents associated with 20% increase in put were recorded in the plants grown in alkaline condition.
Moreover, the ratio of total polyamine/putresine which is an indication of the deleterious injury induced in the plant cells under acidity stress, was significantly increased compared to the control. Thus acidification stress may induce its inhibition on wheat growth by decreasing the putresine content.
IAA could alleviate the harmful effect of acidification on wheat plants by increasing the putresine content (1.6 fold) as being compared with the control plants. It also enhance putresine production in the slightly alkaline grown plant. External oxidant stress can increase plant`s level of the polyamine biosynthetic enzymes, thus increasing the polyamine especially putrescine. Putrescine was constitutively elevated (2.5-5.7) fold in 2–week–old resistant plants.
The ameliorative effect of IAA on the plant growth inhibition may be due to the accumulation of the diamine putresine which has a protective role in the plant cell by its binding to the antioxidant enzymes such as superoxidase dismutase or be conjugated to the small antioxidant molecules allowing them to permit to the site of oxidative stress within the cell[52]. These suggestions were supported with my results that the increase in endogenous polyamine contents was accompanied by increases in chlorophyll contents and their stability and total carbohydrates in wheat plants exposed to acidic pH and treated with IAA. Furthermore, in stressed plants, methionine may be directed towards the biosynthesis of ethylene, however, the addition of IAA in the present investigation may direct the same amino acid towards the biosynthesis of spermine instead of ethylene.
The protective action of polyamines could be explained by their polycationic nature, stabilize membrane by associating with negatively charged phospholipid and negatively charged proteins[37,53,54], their ability to bind with photosynthetic membranes, resulting in conformational changes. Polyamines also acts as free radical scavengers. Moreover, they have a role in maintenance of cellular pH and hormonal action in the plant cells[55].
As regard, the protein profile, mostly 16 bands are separated of molecular weights ranged between 190.8-20.4 kDa except at treatment with pH 7 alone or in combination with IAA where one band was disappeared (49.2 kDa). However, the changes seem to be quantitative especially at 61.7 and 20.4 kDa. The most pronounced increment was at the lower molecular weight (20.4 kDa) in all treatments as being compared with the control (Table 7).
Lastly, it is apparent from Table 8 that the present work managed to verify its aim since plants under acidic stress managed to grow well and produce satisfied yield. Moreover, IAA application improve the yield productivity and quality of plants grown in slightly alkaline condition.
The author would like to express her great thanks to Prof. Dr. Raifa A. Hassanein, Prof. of Plant Physiology and Head of Botany Department, Faculty of Science, Ain Shams University, for her continuous encouragement and valuable remarks during the work.