ABSTRACT
Ethanolic extracts of two controversial medicinal plants Andrographis paniculata (ApE) and Swertia chirayita (ScE) were evaluated for its potential to enhance the repair of radiation-induced DNA damage. Post-irradiation administration of these extracts to mice resulted in faster cellular DNA repair, as revealed from the increased Cellular Repair Index (CRI) of blood leukocytes. Exposure of mice to 2 Gy whole-body gamma radiation resulted in the formation of strand breaks of cellular DNA of bone marrow cells and the unrepaired strand breaks eventually lead to the formation of micronuclei in the reticulocytes. Administration of ApE and ScE under post-irradiation conditions showed a decrease in the number of micronucleated reticulocytes. Administration of these extracts also aided in the regeneration of the immune system as revealed from the induction of endogenous spleen colony formation in mice exposed to whole body gamma radiation. Thus, the studies revealed that ethanolic extracts of Andrographis paniculata and Swertia chirayita abetted the revival of immune system following radiation exposure and the induction of faster and legitimate DNA repair can be the underlying mechanism.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jpt.2012.20.28
URL: https://scialert.net/abstract/?doi=jpt.2012.20.28
INTRODUCTION
The widespread use of radiation in diagnosis, therapy, industry, energy sector and inadvertent exposure during air and space travel and the possibility of nuclear accidents and nuclear terrorism increased the fear of exposure of living beings to radiation and radiation induced damages. Exposure to ionizing radiation results in the disruption of the delicate pro-oxidant/antioxidant balance in tissues by Reactive Oxygen Species (ROS), generated during radiolysis of water (Nair et al., 2001). This leads to protein, lipid and DNA oxidation (Kehrer, 1993; Tan et al., 1988) causing alterations in gene expression, cell proliferation and apoptosis that finally results in reproductive death, interphase death, division delay, chromosome aberrations or mutations (Karbownik and Reiter, 2000). These harmful effects of radiation could be effectively kept under check by the proper use of radioprotectors which are compounds that act as free radical scavengers or enhance the repair of damaged cellular machinery.
Amifostine is the only clinically accepted radioprotector but its radioprotective property is short lived and is associated with many side effects at clinically effective doses. The effectiveness of Amifostine as a radioprotector under post radiation exposure scenario is also not promising (Moulder, 2002; Stone et al., 2004; Hoffmann et al., 2001). The development of an effective radioprotector is always associated with inherent toxicity. Thus medicinal plants that are used in traditional systems of medicine are now being extensively screened to be identified as a prospective radioprotector (Shukla and Gupta, 2010). A. paniculata (Family: Acanthaceae) and S. chirayita (Family: Gentianaceae) are two controversial medicinal plants that are used in the traditional Ayurvedic system of medicine (Nair, 2004). These two plants have several similar pharmacological activities such as anti-inflammatory, hepatoprotective, antidiarrhoeal, antiviral and antimalarial activity (Joshi and Dhawan, 2005; Mishra et al., 2009; Reen et al., 2001; Gupta et al., 1990; Kumar et al., 2004; Wiart et al., 2005). A. paniculata is commonly seen in southern regions of India and S. chirayita in Himalayan parts of India, Nepal and Bhutan. A. paniculata is a substitute of S. chirayita in southeastern region.
In this study, we present compelling evidence to demonstrate enhancement of cellular DNA repair following oral administration of the extract of these plants in whole body gamma irradiated mice and the results indicate potential of these extracts as therapeutics under post radiation exposure scenario.
MATERIALS AND METHODS
Chemicals: Acridine orange, high melting point agarose and low melting point agarose were obtained from Sigma Chemical Company Inc., St Louis, MO, USA. EDTA (Ethylene Diamine Tetra Acetic acid), silver nitrate, ammonium nitrate, zinc sulphate hepta hydrate and tungstosilicic acid were from Merck Specialities Pvt. Ltd., Mumbai, India. All the other chemicals and reagents used in this study were of analytical grade.
Plant materials: A. paniculata (Batch No: B2NIN of KAPL, Athani) and S. chirayita (Batch No: B2N2N of KAPL, Athani) were obtained from Kerala Ayurveda Pvt. Ltd., Athani, Kerala, India. The plants were taxonomically identified as ‘A. paniculata (Burm. f.) Wallich ex Nees and S. chirayita (Roxb. ex Flem) H. Karst’ by Dr. P.N. Ganesh, Associate Professor of Botany, Sree Krishna College, Guruvayur, Kerala, India.
Preparation of extracts: Shade dried aerial parts of both plants were subjected for size reduction to coarse powder. The powders were extracted with 70% ethanol at 70°C using Soxhlet apparatus for 48 h. The ethanolic extracts of both plants were concentrated in water bath and lyophilisation was carried out at -55°C to get solid crude extracts. The yield for A. paniculata was 5.8% w/w and that for S. chirayita was 1.4% w/w. The HPLC and HPTLC fingerprint profiles of the extracts are presented in Fig. 1 and 2. Ethanolic extracts of A. paniculata and S. chirayita were termed as ApE and ScE, respectively.
Animals: Swiss albino mice of 8-10 weeks old, weighing 22-25 g was obtained from the Small Animal Breeding Section (SABS), Kerala Agricultural University, Mannuthy, Thrissur. Kerala. They were kept under standard conditions of temperature and humidity in the Centre’s Animal House Facility. The animals were provided with standard mouse chow (Sai Durga Feeds and Foods, Bangalore, India) and water ad libitum.
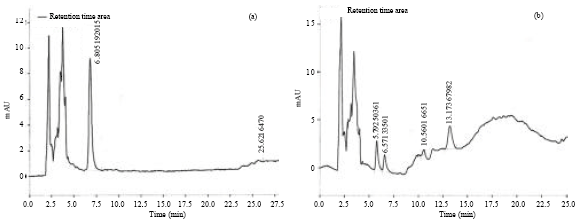 | |
| Fig. 1(a-b): | HPLC of (a) Andrographis paniculata and (b) Swertia chirayita extract |
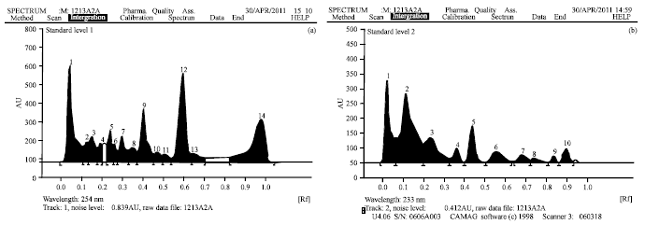 | |
| Fig. 2(a-b): | HPTLC of (a) Andrographis paniculata extract showing 14 peaks and (b) Swertia chirayita extract showing 10 peaks |
All animal experiments in this study were carried out with the prior approval of the Institutional Animal Ethics Committee (IAEC) and were conducted strictly adhering to the guidelines of committee for the purpose of control and supervision of experiments on animals (CPCSEA) constituted by the Animal Welfare Division of Government of India.
Irradiation: Irradiation was carried out using a 60Co-Theratron Phoenix Teletherapy unit (Atomic Energy Ltd., Ottawa, Canada) at a dose rate of 1.88 Gy per min.
For all experiments Swiss albino mice were divided into 4 groups and treated as follows:
| • | Group 1:Sham irradiation+distilled water |
| • | Group 2:2, 4 or 6 Gy 60Co-γ-rays+distilled water |
| • | Group 3:2, 4 or 6 Gy 60Co-γ-rays+ApE (200 mg kg-1) |
| • | Group 4:2, 4 or 6 Gy 60Co-γ-rays+ScE (200 mg kg-1) |
Animals of group 1 and 2 received distilled water, group 1 was kept as untreated control and group 2 was exposed to radiation and was kept as control. Animals were administered with ApE or ScE (200 mg kg-1) to the animals in group 3 or 4, respectively immediately after exposure to ionizing radiation.
Effect of ApE and ScE on repair of cellular DNA damage during exposure to gamma radiation: Two animals from each group exposed to 4 Gy gamma radiation were sacrificed to obtain blood at different time intervals (0, 30, 60 and 90 min). Comet assay was performed on these cells.
Alkaline single cell gel electrophoresis (Comet assay): Alkaline single cell gel electrophoresis was performed using the method given by Singh (2000), with minor modifications (Chandrasekharan et al., 2009). Microscopic slides were coated with normal melting point agarose (1% in PBS) and kept at 4°C till the agarose was solidified. On these slides, 200 μL of 0.8% low melting point agarose containing 50 μL of treated cells were added. After solidification of the low melting agarose, the slides were immersed in pre chilled Lysing solution containing 2.5 M NaCl, 100 mM EDTA, 10 mM Tris-HCl, pH-10, 1% DMSO, 1% Triton X-100 and kept for 1 h at 4°C for lysis of the cells. After lysis, the slides were drained properly and placed in a horizontal electrophoretic apparatus filled with freshly prepared electrophoresis buffer containing 300 mM NaOH, 1 mM EDTA, 0.2% DMSO, pH = 13. The slides were equilibrated in buffer for 20 min and electrophoresis was carried out for 30 min at 20 V. After electrophoresis the slides were washed gently with 0.4 mM Tris-HCl buffer, pH-7.4 to remove alkali. The slides were again washed with distilled water, kept at 37°C for 2 h to dry the gel and silver staining was carried out. The comets were visualized under a binocular microscope and more than 50 images were captured and analyzed using the software ‘CASP’ to find out the extent of DNA damage measured in terms of different comet parameters such as % DNA in tail, tail length, tail moment and olive tail moment (Konca et al., 2003). The parameter Tail Moment (TM) is the product of tail length and % DNA in tail and Olive Tail Moment (OTM) is the product of the distance between the centre of gravity of the head and the centre of gravity of the tail and % DNA in tail. Results are Cellular Repair Index (CRI) which is the percentage decrease from the initial value due to repair (Menon and Nair, 2011).
Effect of ApE and ScE on induction of micronucleus: The micronucleus assay with mouse peripheral blood reticulocytes as reported by Hayashi et al. (1990) using Acridine Orange (AO) coated was carried out to evaluate the chromosomal damage. The animals exposed to 2 Gy gamma radiation was used for this study, since micronucleus induction was found to be maximum at this dose.
Five microliter of peripheral blood was collected from the tail without any anticoagulant at 24th and 48th hour of irradiation onto acridine orange-coated slides, covered immediately with cover glass and these slides were allowed to stand for a few h or overnight in refrigerator to allow cells to settle and to maximize staining. The slides were observed under a blue excitation (488 nm) and a yellow to orange barrier filter (515 nm). The slides were observed for 2000 reticulocytes of peripheral blood (identified by their reticulum structure with red fluorescence) and % of micronucleated (round in shape with a strong yellow-green fluorescence) reticulocytes were scored.
Effect of ApE and ScE on endogenous spleen colony formation in mice exposed to gamma radiation: Three animals from each group was sacrificed on the 12th day post-irradiation by cervical dislocation and the spleen was excised out and fixed in Bouin’s solution containing 1.2% saturated picric acid, 30-40% formalin and glacial acetic acid in the ratio 15:5:1 and the spleens were analyzed for colony formations (Till and Culloch, 1961). The result is obtained from the average of each group and presented as number of spleen colony per spleen.
Statistical analysis: The results are presented as Mean±standard deviation of the studied groups. The statistical analyses of the results were performed using ANOVA with Tukey-Kramer multiple comparisons test.
RESULTS
Effect of ApE and ScE on repair of gamma radiation-induced cellular DNA damage: Exposure of animals to 4 Gy whole body gamma radiation resulted in damage to the cellular DNA as can be evidenced from increased comet parameters such as % DNA in tail, tail length, tail moment and Olive tail moment of cellular DNA of peripheral blood leukocytes following radiation exposure (Fig. 3a-d). These parameters decreased with time after radiation exposure of the animals due to repair of the radiation induced lesions in DNA. The rate of decrease of comet parameters relate to the rate of repair.
To quantify the efficiency of the cells to repair and rejoin strand breaks in DNA, a relation based on the comet parameters of the cellular DNA named Cellular DNA repair index or CRI is used. CRI for a particular comet parameter at any time point is defined as the percentage decrease from the initial value due to repair:
The effect of ApE and ScE on repair of cellular DNA damage was determined by assessing various comet parameters of peripheral blood leukocytes at various time intervals. The results indicate that administration of ApE or ScE (200 mg kg-1) enhanced the repair of cellular DNA.
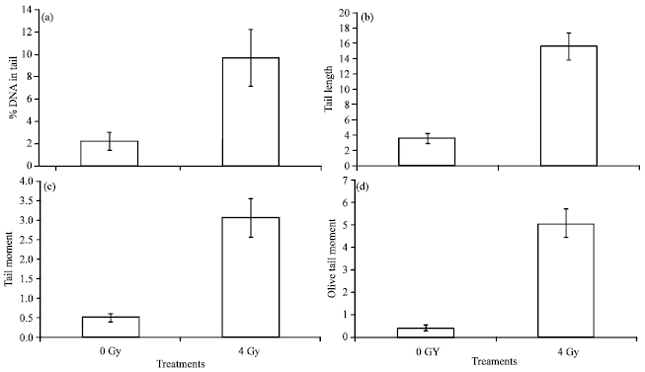 | |
| Fig. 3(a-d): | DNA damage expressed as various comet parameters of mice peripheral blood leukocytes exposed to whole body 4 Gy gamma radiation. Comet assay performed immediately after radiation exposure |
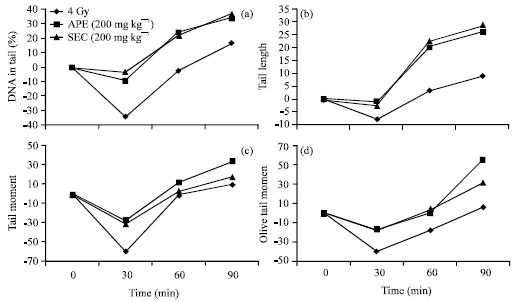 | |
| Fig. 4(a-d): | Effect of APE (200 mg kg-1) and SCE (200 mg kg-1) on repair of cellular DNA of mouse leukocytes from damages induced by exposure to 4 Gy gamma radiation expressed as cellular DNA repair index (CRI) |
| Table 1: | Effect of Administration of ApE (200 mg kg-1) and ScE (200 mg kg-1) extract on induction of micronucleus in animals exposed to 2 Gy gamma-radiation |
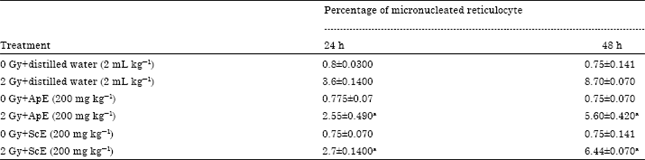 | |
| a: Indicates p<0.001 when compared with respective control | |
The repair efficiency was assessed from comet parameters like % DNA in tail, tail length, tail moment and Olive tail moment which was found to be increased from 16.65, 8.97, 9.85 and 6.05-33.96%, 25.71, 33.23 and 55.12% in the case of ApE administration; 36.47, 28.94, 17.25 and 31.22% in the case of ScE administration at 90 min post irradiation as shown in Fig. 4a-d.
Effect of ApE and ScE on induction of micronucleus: Whole body exposure to 2 Gy gamma radiation in albino mice resulted in significant increase in micro-nucleated reticulocytes. Administration of ApE (200 mg kg-1) and ScE (200 mg kg-1) did not have any effect on the induction of micronuclei. Increase in the induction of micronuclei following irradiation was found to be time dependant, as in the 24 h period the percentage of micronucleated reticulocyte was 3.6±0.14 and at 48 h it increased to 8.4±0.07. Administration of ApE (200 mg kg-1) and ScE (200 mg kg-1) immediately after exposure to 2 Gy radiation resulted in significant decrease in micronucleus induction as evident from Table 1.
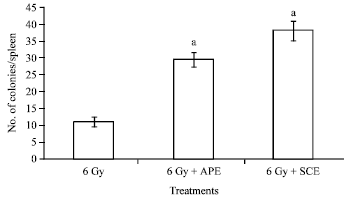 | |
| Fig. 5: | Endogenous spleen colony formation in animals exposed to 6 Gy gamma radiation and administered with APE (200 mg kg-1) or SCE (200 mg kg-1). a: Indicates p<0.001 when compared with respective control |
Effect of ApE and ScE on endogenous spleen colony formation in mice exposed to gamma radiation: From the result presented in Fig. 5, it can be seen that, administration of both ApE and ScE (200 mg kg-1) to 6 Gy gamma irradiated mice resulted in enhanced spleen colony formation. Administration of ApE or ScE without exposing the animals to radiation did not induce induction of spleen colony formation. The control irradiated animals developed on an average of 11±1.41 colonies whereas ApE administration resulted in an increase to 29.5±2.12 colonies and ScE administration increased the number of spleen colonies to 38±2.83.
DISCUSSION
Radiation is one of the most severe causes of oxidative damage arbitrated by free radical flux. Low levels of ionizing radiation exposure cause damage to the hematopoietic system consequently resulting in hematopoietic syndrome (Mettler and Voelz, 2002). Since ionizing radiation is finding application in varied fields, the anthropogenic radiation dose also adds on to the already existing natural background radiation to which humans are constantly being exposed (Wakeford, 2004). Low level increase in background radiation can be a cause of cellular damage to the population. In this present scenario, a nontoxic radioprotector which can be used on a regular basis as a dietary supplement is of significant importance.
Compounds with antioxidant properties have been shown to prevent the deleterious effects of ionizing radiations in living systems and bio-molecules due to their ability to scavenge free radicals (Mathew et al., 2007). Our previous study has confirmed the antioxidant activity of A. paniculata and S. chirayita (Nagalekshmi et al., 2011).
In the present study, the administration of alcoholic extracts of A. paniculata and S. chirayita to mice post irradiation resulted in the rapid repair of cellular DNA as confirmed by the reduction in comet parameters. Decrease in comet parameters is taken as an indication of repair of cellular DNA (Schmezer et al., 2001). Unrepaired DNA fragments at the last sampling time may be due to the slower repair process or defective repair machinery. When repair process is defective, the unrepaired DNA fragment will be expressed as micronucleus in the dividing cells (Torous et al., 2003). Administration of A. paniculata and S. chirayita extracts to irradiated mice have shown a decreased micronucleus count, reflecting the ability of the plants in aiding legitimate repair of the damaged genomic DNA.
Formation of endogenous spleen colonies is an index of hematopoietic stem cell proliferation. Spleen colony forming cells have been identified as the common precursor of tissue mast cells and granulocytes (Kitamura et al., 1981). For the present study, the ability of the extracts of A. paniculata and S. chirayita in assisting the recovery of the hematopoietic system from radiation induced damage is ascertained.
REFERENCES
- Chandrasekharan, D.K., T.V. Kagiya and C.K. Nair, 2009. Radiation protection by 6-palmitoyl ascorbic acid-2-glucoside: Studies on DNA damage in vitro, ex vivo, in vivo and oxidative stress in vivo. J. Radiat. Res., 50: 203-212.
PubMed - Gupta, S., M.A. Choudhry, J.N.S. Yadava, V. Srivastava and J.S. Tandon, 1990. Antidiarrhoeal activity of diterpenes of Andrographis paniculata (Kal-Megh) against Escherichia coli enterotoxin in in vivo models. Pharm. Biol., 28: 273-283.
CrossRefDirect Link - Hayashi, M., T. Morita, Y. Kodama, T. Sofuni and M. Ishidate, 1990. The micronucleus assay with mouse peripheral blood reticulocytes using acridine orange-coated slides. Mutat. Res., 245: 245-249.
PubMed - Hoffmann, G.R., J. Buccola and M.S. Merz, 2001. Structure-activity analysis of the potentiation by aminothiols of the chromosome-damaging effect of bleomycin in G0 human lymphocytes. Environ. Mol. Mut., 37: 117-127.
CrossRef - Karbownik, M. and R.J. Reiter, 2000. Antioxidative effects of melatonin in protection against cellular damage caused by ionizing radiation. Proc. Soc. Exp. Biol. Med., 225: 9-22.
PubMed - Kehrer, J.P., 1993. Free radicals as mediators of tissue injury and disease. Crit. Rev. Toxicol., 23: 21-48.
CrossRefPubMedDirect Link - Kitamura, Y., M. Yokoyama, H. Matsuda, T. Ohno and K.J. Mori, 1981. Spleen colony-forming cell as common precursor for tissue mast cells and granulocytes. Nature, 291: 159-160.
CrossRef - Konca, K., A. Lankoff, A. Banasik, H. Lisowka and T. Kuszewsk et al., 2003. A cross-platform public domain PC image-analysis program for the comet assay. Mutat. Res., 534: 15-20.
PubMed - Kumar, R.A., K. Sridevi, N.V. Kumar, S. Nanduri and S. Rajagopal, 2004. Anticancer and immunostimulatory compounds from Andrographis paniculata. J. Ethnopharmcol., 92: 291-295.
CrossRefDirect Link - Mathew, D., C.K. Nair, J.A. Jacob, N. Biswas, T. Mukherjee, S. Kapoor and T.V. Kagiya, 2007. Ascorbic acid monoglucoside as antioxidant and radioprotector. J. Radiat. Res., 48: 369-376.
PubMed - Menon, A. and C.K.K. Nair, 2011. POLY MVA: A dietary supplement containing α-lipoic acid palladium complex, enhances cellular DNA repair. Int. J. Low Radiat., 8: 42-54.
Direct Link - Mettler, Jr. F.A. and G.L. Voelz, 2002. Major radiation exposure-What to expect and how to respond. N. Engl. J. Med., 346: 1554-1561.
PubMed - Mishra, K., A.P. Dash, B.K. Swain and N. Dey, 2009. Anti-malarial activities of Andrographis paniculata and Hedyotis corymbosa extracts and their combination with curcumin. Malar. J., Vol. 8.
CrossRefDirect Link - Moulder, J.E., 2002. Report on an interagency workshop on the radiobiology of nuclear terrorism. Molecular and cellular biology of moderate dose (1-10 Sv) radiation and potential mechanisms of radiation protection. Radiat. Res., 158: 118-124.
Direct Link - Nagalekshmi, R., A. Menon, D.K. Chandrasekharan and C.K.K. Nair, 2011. Hepatoprotective activity of Andrographis paniculata and Swertia chirayita. Food. Chem. Toxicol., 49: 3367-3373.
CrossRefDirect Link - Nair, C., D. Parida and T. Nomura, 2001. Radioprotectors in radiotherapy. J. Radiat. Res., 42: 21-37.
PubMed - Nair, R.V., 2004. Controversial Drug Plants. Universities Press, India, ISBN: 9788173714696, Pages: 257.
Direct Link - Reen, R.K, M. Karan, K. Singh, V. Karan, R.K. Johri and J. Singh, 2001. Screening of various Swertia species extracts in primary monolayer cultures of rat hepatocytes against carbon tetrachloride and paracetamol-induced toxicity. J. Ethnopharmacol., 75: 239-247.
PubMedDirect Link - Schmezer, P., N.R. Behbahani, A.R.S. Thiel, W. Rittgen and P. Drings et al., 2001. Rapid screening assay for mutagen sensitivity and DNA repair capacity in human peripheral blood lymphocytes. Mutagenesis, 16: 25-30.
CrossRef - Shukla, S.K and M.L. Gupta, 2010. Approach towards development of a radioprotector using herbal source against lethal irradiation. Int. Rec. J. Plant. Sci., 1: 118-125.
Direct Link - Singh, N.P., 2000. Microgels for estimation of DNA strand breaks, DNA protein crosslinks and apoptosis. Mutat. Res., 455: 111-127.
PubMedDirect Link - Stone, H.B., J.E. Moulder, C.N. Coleman, K.K. Ang and M.S. Anscher et al., 2004. Models for evaluating agents intended for the prophylaxis, mitigation and treatment of radiation injuries report of an NCI workshop. Radiat. Res., 162: 711-728.
PubMed - Tan, K.H., D.J. Meyer, N. Gillies and B. Ketterer, 1988. Detoxification of DNA hydroperoxide by glutathione transferases and the purification and characterization of glutathione transferases of the rat liver nucleus. Biochem. J., 254: 841-845.
PubMed - Till, J.E. and E.A.M. Culloch, 1961. A direct measurement of the radiation sensitivity of normal mouse bone marrow cells. Radiat. Res., 14: 213-222.
PubMed - Torous, D.K., N.E. Hall, F.G. Murante, S.E. Gleason, C.R. Tometsko and S.D. Dertinger, 2003. Comparative scoring of micronucleated reticulocytes in rat peripheral blood by flow cytometry and microscopy. Toxicol. Sci., 74: 309-314.
CrossRef - Wiart, C., K. Kumar, M.Y. Yusof, H. Hamimah, Z.M. Fauzi and M. Sulaiman, 2005. Antiviral properties of ent-labdene diterpenes of Andrographis paniculata Nees, inhibitors of herpes simplex virus type 1. Phytother. Res., 19: 1069-1070.
CrossRef








