Research Article
Sesame Oil: Potential Interaction with P450 Isozymes
Department of Pharmacology and Toxicology, Faculty of Veterinary Medicine, University of Adnan Menderes, Isikli Koyu 09100, Aydin, Turkey
Sesame, Sesamum indicum L., is an annual herb native to the tropics and grown primarily for its oil-rich seeds. This highly aromatic oil ranges in hue from golden to brown and is extensively used in Asian cuisine. The oil is sometimes used as cooking oil, but most often is used as a seasoning accent in stir-fries, dressings, sauces and marinades. In addition, sesame oil is also used in the some injectable drug formulations in human and veterinary medicine.
Sesame lignans (a non-fat constituent) or antioxidants such as sesamin, episesamin, sesaminol and sesamolin are major constituents of sesame oil and all have chemically methylenedioxyphenyl group (Kamal-Eldin and Appelqvist, 1994; Kamal-Eldin et al., 1994) (Fig. 1). Sesame seed lignans were reported to be responsible for many unique chemical and physiological properties of sesame oil (Kamal-Eldin, 1995). Sesamin, one of the lignans present most abundantly in sesame seed and found in various medicinal plants. Sesamin increases the detoxification capability of liver, reduces the incidence of chemically induced tumors and protects neuronal cells against oxidative stress and exhibits anti-hypertensive, anti-inflammatory and anti-allergic effect (Hirose et al., 1992; Hou et al., 2003; Jeng and Hou, 2005).
The synergists that have a methylenedioxyphenyl group were firstly introduced in 1940 to increase the effectiveness of pyrethrum. Since, then many compounds have appeared, but only a few are still marketed. These synergists are piperonyl butoxide, sesamin, sesamolin and sesamex (Fig. 1). Piperonyl butoxide, a semisynthetic derivative of safrole, is the most widely used synthetic pyrethrin synergist and there are no reports available on toxic effects on humans resulting from the exposure to it (Breathnach, 1998).
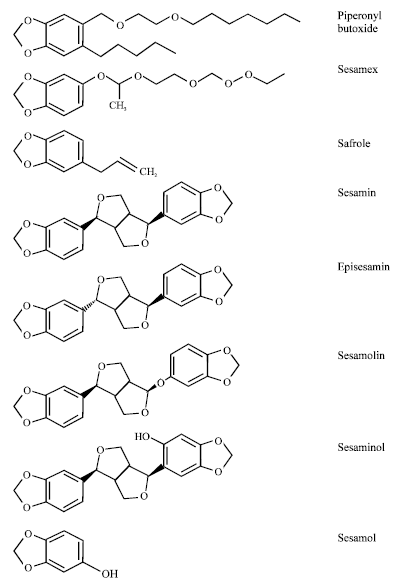 | |
| Fig. 1: | Chemical structure of methylenedioxyphenyl compounds |
It was found in sesame oil and later named sesamine (Ware and Whitacre, 2004). Piperonyl butoxide does not have any pesticidal activity. However, when combined with insecticides, such as pyrethrin, pyrethroid and carbamate insecticides, their potency is increased considerably (Maklakov et al., 2001). It was clearly shown that PB interacts (induce or inhibit) with some P450 isozymes both in insects and mammalian species (Hodgson and Philpot, 1974; Adams et al., 1993a, b). This family of enzymes acts as the principal detoxification pathway for many pesticides. Piperonyl butoxide is a potent cytochrome P450 inhibitor and this inhibition of the detoxification pathway allows higher unchanged systemic concentrations of the active insecticide to remain within the target animal for a longer period. It was demonstrated that the major route of metabolism of piperonyl butoxide involved the opening of the methylenedioxy ring followed by loss of the methylene group into the endogenous metabolic pool (Cockburn and Needham, 1998). This is also believed to be the basis of the initial inhibition of the cytochrome P450 enzyme system, which is essential for the compound’s efficacy as a synergist. The previous studies have been also shown that piperonyl butoxide reduced the oxidative metabolic inactivation of benzimidazole anthelmintics significantly in animals (McKellar et al., 2002; Gokbulut, 2000; Benchaoui and McKellar, 1996). More recently, it was demonstrated that the plasma concentration profiles of fenbendazole, a metabolite generated from oxfendazole, were significantly lower after the treatment with oxfendazole alone compared to those obtained after the oxfendazole+piperony butoxide treatment. The enhanced pharmacokinetic profiles correlated with increased anthelmintic efficacy (Sanchez Bruni et al., 2005). Moreover, it was shown that the methylenedioxyphenyl compounds inactivated human CYP1A1, CYP2C9, CYP2D6 and CYP3A4 (Nakajima et al., 1999). It was also indicated that isosafrole, another methylenedioxyphenyl compound, interacted with the cythochrome P450 enzymes and more recently it was reported that safrole inhibited human cythochrome P450 enzymes (CYP1A2, CYP2E1 and CYP2A6) (Ueng et al., 2005).
The consumption of sesame oil by human is increasing because of their health benefits mentioned above. According to my hypothesis sesame seed or oil could interact with the P450 isozymes and affect the drug metabolisms in human, due to having a methylenedioxyphenyl group I suggest that the methylenedioxyphenyl compounds in sesame oil are potent inhibitors or inactivators of some CYP isoforms. The degree of interaction could be more related with the amount of lignans in sesame seed or oil and their consumption amount by human. Nevertheless, there is no data available on the literature on the interaction between sesame seed, sesame oil or the lignans and drugs. Obviously this hypothesis will require careful confirmation in animal and human trials focused on the determination of the interaction and identification of the P450 isozymes interacted with the lignans.