Research Article
Molecular Modelling Analysis of the Metabolism of Hydrazine
School of Biomedical Sciences, Faculty of Health Sciences, (242, The University of Sydney, P.O. Box 170, Lidcombe, NSW 1 825, Australia
Hydrazine is a simple nitrogen compound, used extensively in the manufacture of several important industrial products including plastic blowing agents, growth retardants and pharmaceuticals and in rocket propellants (Preece et al., 1991). It is also a metabolite of two important drugs namely isoniazid (Noda et al., 1978; Blair et al., 1985), CNS disturbances (Roe et al., 1979) and hydralazine (Timbrell and Harland, 1979). The compound has also been used in the treatment of weight loss in terminal cancer patients (Gold, 1987).
Anhydrous hydrazine is a fuming colourless liquid with boiling point 114°C. It is remarkably stable even though it has large (positive) heat of formation. It is a toxic compound causing fatty liver due to triglyceride accumulation (Amenta and Johnston, 1962) and in some cases liver necrosis (Patrick and Back, 1965), CNS disturbances (Roe et al., 1979) and tumours in various organs (Roe et al., 1979; Balo, 1979). Hydrazine also causes depletion of ATP and reduced glutathione (Jenner and Timbrell, 1994). It is found that in experimental animals hepatotoxicity of hydrazine can be modulated by induction or inhibition of cytochrome P450.
As nitrogen has no stable radioactive isotope, hydrazine metabolism cannot be studied by conventional analytical methodologies. Recently NMR spectroscopy has been applied to study metabolism of hydrazine. Figure 1 shows the important metabolites of hydrazine. It can be seen that hydrazine is acetylated to produce monoacteylhydrazine and diacetylhydrazine. But these compounds constitute only a small proportion of the absorbed dose (<5%). About 10% is excreted unchanged. A significant proportion (25%) is metabolized to nitrogen that is detected in expired air (Springer et al., 1980; Nelson and Gordon, 1982). Other metabolites of hydrazine include 1,4,5,6-tetrahydro-6-oxo-3-pyridazine carboxylic acid (THOPC) formed from conjugation between hydrazine and 2-oxoglutarate, pyruvate hydrazone and urea (Preece et al., 1991).
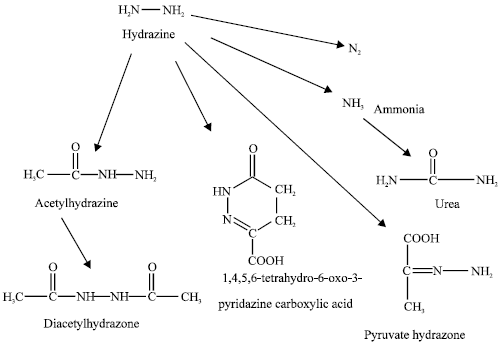 | |
| Fig. 1: | Metabolic pathways for hydrazine (Preece et al., 1991) |
In this study, molecular modelling analyses have been carried out using the programs HyperChem 7.0 (HyperChem, 2002) and Spartan ’02 (Spartan, 2002) to investigate the relative stability of hydrazine and its metabolites.
The geometries of hydrazine, acetylhydrazine, diacetylhydrazine, 1,4,5,6-tetrahydro-6-oxo-3-pyridazine carboxylic acid (THOPC), pyruvate hydrazone, ammonia and nitrogen have been optimized based on molecular mechanics, semi-empirical and DFT calculations, using the molecular modelling programs Spartan ’02 and HyperChem 7.0. Molecular mechanics calculations were carried out using MM+ force field. Semi-empirical calculations were carried out using the routine PM3. DFT calculations were carried using the program Spartan ’02 at B3LYP/6-31G* level. For the optimized structures, single point calculations were carried to give heat of formation, enthalpy, entropy, free energy, dipole moment and solvation energy, HOMO and LUMO.
Table 1 gives the total energy, heat of formation as per PM3 calculation, enthalpy, entropy, free energy, dipole moment, energies of HOMO and LUMO as per both PM3 and DFT calculations for hydrazine, acetylhydrazine, diacetylhydrazine, 1,4,5,6-tetrahydro-6-oxo-3-pyridazine carboxylic acid (THOPC), pyruvate hydrazone, ammonia, nitrogen, oxygen and water. Figure 2 gives the optimized structures of hydrazine, acetylhydrazine, diacetylhydrazine, THOPC, pyruvate hydrazone, urea, ammonia and nitrogen as per PM3 calculations using the program HyperChem 7.0. The structures also give 2D contours of total electrostatic potential. The contours of total electrostatic potential show the concentration of negative charges around the carbonyl oxygen in all the compounds, indicating that the positions may be subject to electrophilic attack.
| Table 1: | Calculated thermodynamic and other parameters for hydrazine and its metabolites [‘DM’ stands for dipole moment] |
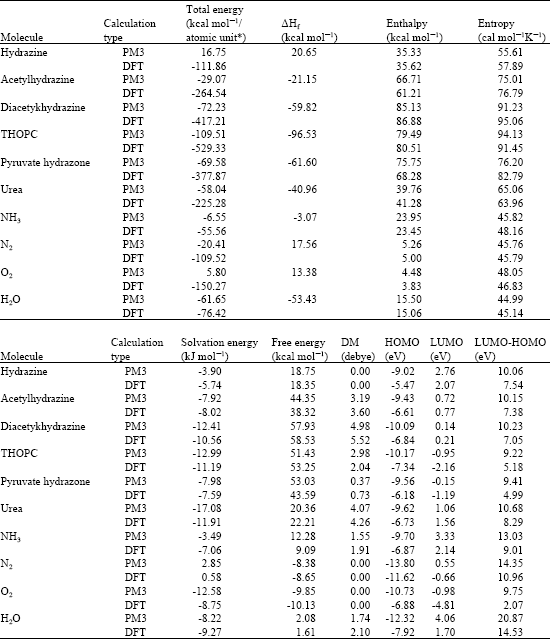 | |
| * in atomic units from DFT calculations | |
The solvation energies of hydrazine, acetylhydrazine, diacetylhydrazine, THOPC, pyruvate hydrazone, ammonia and nitrogen from PM3 calculations in kcal mol-1 are respectively -3.90, -7.92, -12.41, -12.99, -7.98, -17.08, -3.49 and 2.85, respectively indicating that all the metabolites of hydrazine except nitrogen are more soluble in water and hence can be more easily excreted. Although nitrogen has very low solubility in water, being a gas it can be easily excreted in breath. Pyruvate hydrazone and THOPC are found to have lower LUMO-HOMO energy differences indicating that two metabolites would be most kinetically labile. Relatively large LUMO-HOMO energy difference (7.54 eV as per DFT calculations) suggests that hydrazine would have unusually high stability. Urea also is found to have a large value for LUMO-HOMO energy difference indicating its relative kinetic inertness.
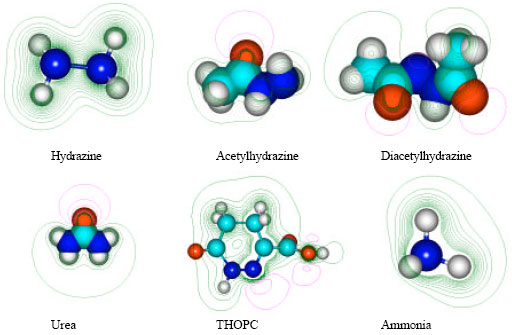 | |
| Fig. 2: | Structures of hydrazine, acetylhydrazine, diacetylhydrazine, urea, THOPC and ammonia giving 2D contours of total electrostatic potential showing concentration of negative charges around oxygen centres in acetylhydrazine, diacetylhydrazine, urea, oxygen and nitrogen centres in the case of THOPC |
 | |
| Fig. 3: | Structures of hydrazine, urea, diacetylhydrazine and THOPC giving surface electric charges where red indicates negative, blue indicates positive and green indicates neutral |
The surfaces of hydrazine, diacetylhydrazine, urea and THOPC appear to be negatively charged, the most negative being that of urea (Fig. 3). The concentration of negative charges on the surface gives further support to the idea that the molecules would be subject to electrophilic attack.
Although the oxidation of hydrazine to nitrogen and water occurs spontaneously, hydrazine molecule has a high stability. Molecular modelling analyses show that the apparent high stability of hydrazine is due to its kinetic inertness. The metabolites THOPC and pyruvate hydrazone have relatively low LUMO-HOMO differences indicating that the two compounds would be the most reactive metabolites of hydrazine. The presence of negatively charged regions indicates that hydrazine and its metabolites may be subject to electrophilic attack.