ABSTRACT
The purpose of this study was to assess changes induced in free amino acids in the digestive gland-gonad complex and haemolymph of Indoplanorbis exustus naturally infected with larval digenes. A preliminary thin layer chromatography, followed by HPTLC analysis was used to determine the qualitative status of free amino acids in the uninfected and infected snails. Qualitative analysis showed the presence of seven amino acids, namely, histidine, lysine, alanine, serine, valine, leucine and isoleucine in uninfected DGG and haemolymph, whereas, only lysine was not observed in all infected sample. Alanine was absent in snails infected with Cercariae indicae XVII and Cotylophoron cotylophorom, however, valine also depleted in snails infected with C. cotylophorom. Quantitative analysis of total free amino acids in all infected DGG and haemolymph samples depicted significant decrease (p<0.05) in their concentrations thus clearly indicating depletion of amino acids in parasitized snails.
PDF Abstract XML References Citation
How to cite this article
URL: https://scialert.net/abstract/?doi=jp.2009.87.93
INTRODUCTION
Snail-mediated helminthiasis causes great morbidity in man, birds and other livestock. Snails are essential intermediate host in the life cycle of digenetic trematodes. The family Planorbidae comprises a large group of common fresh water pulmonates snails found in nearly all parts of the world. Many planorbidae are of medical and veterinary importance since they serve as intermediate hosts to various blood, liver and intestinal flukes, may affect man directly or accidentally as parasitic zoonoses. The fresh water snail Indoplanorbis exustus (Mollusca: Gastropoda) is widely distributed in India and act as intermediate host of Schistosoma nasale, Schistosoma spindale and Schistosoma indicum the causative agent of animal schistosomiasis and other trematodal diseases.
Much attention has been paid on the schistosomiasis transmitting snail, Biomphalaria glabrata and major work deals with the study of biochemical changes induced by several molluscicides in different molluscan tissues and haemolymph (Aly et al., 2004; El-Emam and Ebeid, 1989; Mantawy, 2001). It was believed that starvation and schistosome infection caused quantitative and qualitative changes in protein and carbohydrate levels in snails (Iskierko and Gorski, 1974; Becker, 1980; Stanislawski and Becker, 1979; Pinheiro et al., 2001; Mahmoud et al., 2002) and it was concluded that starvation is a physiological condition which resembles trematodal infection (Emara and El-Ghaeb, 1978; Thompson, 1997).
Recent studies on the topic have used HPTLC (High Performance Thin Layer Chromatography) to determine the pathobiochemical effect of Schistosoma monsoni infection on the amino acid (Pachuski et al., 2002) carbohydrate content (Jarusiewicz et al., 2006) carboxylic acids (Massa et al., 2007; Bezerra et al., 1997) and neutral lipids (Fried et al., 2001; Bandstra et al., 2006) in B. glabrata by observing qualitative and quantitative changes in certain analytes as a function of the schistosome infection in the snails in all the aforementioned studies.
The amino acid concentration of three land snails, Limicolaria sp., Archatina archatina and Archachatina marginata, were evaluated. Amino acid results showed that the protein contained nutritionally useful quantities of most of the essential amino acids, including sulphur containing amino acids (Adeyeye and Afolabi, 2004). TLC was used to analyze the free pool amino acid of the digestive gland-gonad complex of B. glabrata infected with larval Echinostoma caproni (Ponder et al., 2004).
However, there is no report available regarding alterations in the free amino acid content in haemolymph and DGG (digestive gland-gonad complex) of Indoplanorbis exustus harboring trematodal infections, therefore, the present study was planned to unravel such changes in haemolymph and DGG of Indoplanorbis exustus naturally infected with digenetic trematodes.
MATERIALS AND METHODS
The planorbid snail Indoplanorbis exustus was collected from irrigated rice fields of Dayalbagh, Agra (27°10’ N and 78° 05’E, a semiarid zone of North India) during July to September 2007. Snails were hand captured and brought to the laboratory where they were maintained in the polypropylene aquaria containing tap water for acclimatization to laboratory conditions. They were fed with spinach ad libitum and water was changed thrice in a week. Dead animals were removed from the aquaria to avoid any contamination. After three days acclimatization, snails were isolated at room temperature (32-35°C) in 50 mL beakers containing tap water. The beakers were kept under the illumination for one hour and examined for the presence of cercaria, if any. Snails shedding cercariae were isolated and treated as infected whereas others considered as controls.
Sample Preparation
The DGG was dissected free of the snail body and checked microscopically whether there is any pre-patent stage of trematodes present in control snails whereas, in the case of infected ones all the intra-molluscan stages were removed prior to homogenization. Homogenate was prepared in 70% ethanol and centrifuged at 3000 rpm for 15 min, the supernatant was then transferred to a beaker and evaporated to dryness. The amino acid residue was then reconstituted with 100 to 200 μL of distilled water and maintained in the freezer until analyzed (Massa et al., 2007). A similar procedure was used to prepare the DGG from the uninfected snails. A total of 32 DGG from infected snails and 40 DGG from the uninfected snails were used. All analyses were based on pools of two to five DGG per sample.
For haemolymph preparation, each infected snail was cracked lightly with a hammer in the bottom of a 6 cm Petri dish to obtain about 50 μL of haemolymph per snail; the haemolymph was allowed to drain into the dish. A pooled sample consisted of 550 μL of haemolymph, obtained from 10-12 snails. The haemolymph was collected with a pippet, placed in an Eppendroff tube with equal amount of 70% ethanol and centrifuged at 3000 rpm for 5 min to separate the supernatant from the hemocytes and cellular debris. The supernatant then transferred to a beaker and evaporated to dryness. The amino acid residue was then reconstituted with 100 to 200 μL or equal amount to that of haemolymph of distilled water and maintained in the freezer until analyzed. A similar procedure was used to collect haemolymph samples from the uninfected snails. A total of 32 snails were used to obtain haemolymph from the infected snails and another 40 were used to obtain haemolymph from the control snails.
Quantitative Analysis of Total Free Amino Acids
Quantitative estimation of total free amino acids was done colorimetrically by ninhydrin method measuring the color at 570 nm (Sadasivam and Manikam, 1996). Standardization curve was drawn at various concentrations of leucine ranging from 10 to 100 μg from which regression equation was calculated. Employing this equation, concentration of total free amino acids in unknown samples was calculated by multiplication of the sample solution concentration interpolated from the calibration curve by original sample volume and division of the product by mass of snail DGG (g) and in the case of haemolymph with volume of haemolymph (μL).
HPTLC Analysis of Amino Acids
Standards of 19 amino acids were prepared as 0.01 mg solutions in 10 mL distilled water. Quantitative analyses of amino acids were performed on 10-20 cm silica gel HPTLC plates with a preadsorbent sample application zone and 19 channels.
Samples and standards were applied with a 2 μL digital micro dispenser. Quantitative amino acid analyses, initial zones were allowed to air dry and plates were developed in a TLC chamber (Camag, TLC Scanner III) containing a saturation pad and n-Butanol-acetic acid-water (3: 1: 1) for the silica gel. Detection of the zones was achieved by spraying the developed and air-dried plates with ninhydrin reagent, briefly, air drying for 30 min and heating for 10 min at 110°C in oven to produce purple zones for all amino acids, except for proline, which had a yellow color.
Statistical Analysis
Results were analysed statistically and quantitative estimation of total free amino acids was done with the help of probit equation. The data are presented as Mean±SEM value. Number of animals per group stated in the figure legends. One way Analysis of Variance (ANOVA) followed by Student- Newman-Keuls test was used to analyze mean differences between experimental groups separately after ascertaining the homogeneity of variance between treatment groups by Bartlett`s test.
RESULTS AND DISCUSSION
Quantitative Analysis of Total Free Amino Acids
For quantitative determination of total free amino acids, standardization curve was drawn at various concentrations of leucine ranging from 10 to 100 μg from which regression equation was calculated as:
Y = 0.0092x
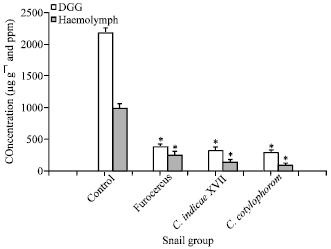 | |
| Fig. 1: | Comparison of total free amino acids in DGG and haemolymph of control and infected Indoplanorbis exustus. Values represent Mean±SD of five samples. *Significant reduction in the concentration of total free amino acids in haemolymph and DGG of infected snails relative to the control (student’s t-test p<0.05) |
| Table 1: | Qualitative data for free amino acid analysis in the haemolymph and DGG of Indoplanorbis exustus |
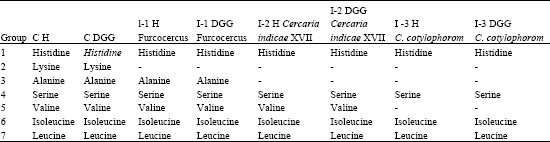 | |
| CH: Control haemolymph, CDGG: Control digestive gland-gonad complex, I: Infected | |
Significant differences between amino acid concentration in infected and uninfected sample were evaluated using Student’s t-test with p<0.05 (Fig. 1). It is evident from the Fig. 1 that a significant decrease occur in concentration of total free amino acids in DGG and haemolymph of various larval digenes in infected snails as compared to the DGG and haemolymph of controls. The values were found to be statistically significant (p<0.05).
Qualitative Analysis of Free Amino Acids through TLC and HPTLC
For qualitative determination of free amino acids in DGG and haemolymph of control and infected snails, initially TLC was done in different ratios of n-Butanol: Acetic acid: Water. The work was further extended to HPTLC whereby all standards and samples were analysed on preparative silica gel plates utilizing 10 μL microdispensers and after drying plates were developed in 3:1:1 ratio of n-Butanol, acetic acid and water. This was followed by scanning of the plates through Camag TLC scanner III utilizing CATS software.
Results are reported in Table 1 which shows the presence of seven amino acids in haemolymph and DGG namely, histidine, lysine, alanine, serine, valine, leucine and isoleucine in uninfected DGG and haemolymph, whereas, only lysine was not observed in all infected samples. Alanine was absent in snail infected with cercaria indicae XVII and Cotylophoron cotylophorom cercariae, however valine also depleted in snail infected with Cotylophoron cotylophorom cercariae (Fig. 2).
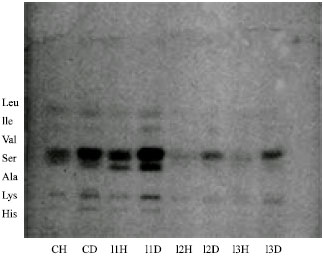 | |
| Fig. 2: | HPTLC analysis of infected and uninfected Indoplanorbis exustus. CH: Uninfected haemolymph, CD: Uninfected DGG, I1: Furcocercus cercariae, I2: Cercariae indicae XVII, I3: C. cotylophorom cercariae |
By comparison with the migration of standards, histidine, lysine, alanine, serine, valine, leucine and isoleucine were identified as amino acids found in both the infected and uninfected DGG. Our most interesting finding was the significant reduction of all of the amino acids in the DGG and haemolymph of infected snails relative to the uninfected controls which is evident from Fig. 2. These larval stages inhabit the intertubular spaces of the DGG and cause mechanical and lytic damage to the digestive gland cells of the hepatopancreas (Thompson, 1997). The damage probably resulted in a leakage of the amino acids, which are in turn utilized by the larval stages. Moreover, increased metabolic activity associated with the presence of larval trematodes may accelerate the use of the amino acids by host cells in the digestive gland as well. As documented in several studies (Thompson, 1997), the site of active intermediary metabolism in these snails is associated with the mitochondria of the digestive gland cells.
Few studies have been done on the amino acid content of Biomphalaria glabrata snails infected with the intramolluscan stages of Echinostoma caproni as determined by thin layer chromatography (Ponder et al., 2004). Study reported a reduction in free amino acid levels in the DGG tissues only of B. glabrata. Ponder and his coworkers found the presence of histidine, lysine, serine, alanine, valine, isoleucine and leucine qualitatively however no statistical difference was observed quantitatively in uninfected and infected snails. On the other hand, amino acids in adult E. caproni revealed the presence of histidine, lysine, alanine and proline only (Ponder et al., 2003). There are few reports available on the quantitative and qualitative characterization of neutral lipids, sugars, organic acid and carboxylic acids (Bezerra et al., 1999; Bandstra et al., 2006; Jarusiewicz et al., 2006; Massa et al., 2007). There are no studies that examined the effects of trematodal infection on free amino acids in Indoplanorbis exustus snails by thin layer chromatography.
Our findings show the presence of similar seven amino acids in haemolymph and DGG as observed by Ponder et al. (2003) namely histidine, lysine, alanine, serine, valine, leucine and isoleucine in uninfected DGG and haemolymph, whereas, only lysine was not observed in all infected samples. This is in conformity with the work of Pachuski et al. (2002). Lysine is probably essential amino acids for the developing intramolluscan stages, i.e., sporocysts, redia and cercariae, of the parasitic trematodes. Since, this amino acid cannot be synthesized de novo, it is provided by the snail host Indoplanorbis exustus.
In the present study, Alanine was absent in snails infected with Cercariae indicae XVII and Cotylophoron cotylophorom cercariae, however valine also depleted in snail infected with Cotylophoron cotylophorom cercariae. Absence of alanine and valine has been observed for the first time in such infections. We can correlate depletion of these amino acids with the size and species of cercariae, which were larger than the furcocercus species. Further, quantitative data for amino acids confirms this qualitative depletion of amino acids in infected snails. Presumably, the reduction in these acids reflected their possible use as metabolites by the developing sporocysts, redia and cercariae of digenetic trematodes which reproduce asexually and exhaust the amino acid reservoir of host. The significant decline in total free amino acids in infected snails reflects the parasite’s ability to obtain these amino acids from its host. This is a first report on Indoplanorbis exustus, vector of a number of treamtodal diseases including schistosomiasis, which demonstrated signinificant altereations in the free amino acids content in the host snails infected with three different cercarial infections.
ACKNOWLEDGMENTS
The authors are thankful to Department of Zoology, Dayalbagh Educational Institute, Dayalbagh, Agra for providing necessary facilities and infrastructure to carry out this research and gratefully acknowledge the technical assistance of Mr. Gursaran Singh in collecting snails.
REFERENCES
- Adeyeye, E.I. and E.O. Afolabi, 2004. Amino acid composition of three different types of land snails consumed in Nigeria. Food Chem., 85: 535-539.
CrossRefDirect Link - Aly, S.A., H.F. Aly, N. Saba-el-Rigal and E.M. Sammour, 2004. Induced changes in biochemical parameters of the molluscan tissues non-infected using two potent plants molluscicides. J. Egypt Soc. Parasitol., 34: 527-542.
PubMed - Bandstra, S.R., B. Fried and J. Sherma, 2006. High performance thin layer chromatographic analysis of neutral lipids and phospholipids in Biomphalaria glabrata patently infected with Echinostoma caproni. Para Res., 99: 414-418.
CrossRef - Bezerra, J.C.B., W. Becker and U.E. Zelck, 1997. A comparative study of the organic acid content of the haemolymph of Schistosoma mansoni resistant and susceptible strains of Biomphalaria glabrata. Memories Inst. Oswaldo. Cruz, 92: 421-425.
Direct Link - Bezerra, J.C., A. Kemper and W. Becker, 1999. Profile of organic acid concentrations in the digestive gland and hemolymph of Biomphalaria glabrata under estivation. Memorias Inst. Oswaldo Cruz, 94: 779-784.
PubMed - Becker, W., 1980. Metabolic interrelationship of parasitic trematodes and molluscs specially Schistosoma mansoni in Biomphalaria glabrata. Z. Parasitol., 63: 101-111.
CrossRef - El-Emam, M.A. and F.A. Ebeid, 1989. Effect of Schistosoma mansoni infection, starvation and molluscicides on acid phosphate, transaminases and total protein in tissues and haemolymph of B. glabrata. J. Egypt Soc. Parasitol., 19: 139-147.
PubMed - Emara, S.H. and F.M. El-Ghaeb, 1978. Total protein free amino acids in haemolymph of snails Biomphalaria alaxandrina and Helisoma duryi exposed to Schistosoma monsoni miracidia. Ind. J. Exp. Biol., 16: 378-381.
PubMed - Fried, B., E.E. Muller, A. Broadway and J. Sherma, 2001. Effects on the development of Schistosoma mansoni in Biomphalaria glabrata and on the neutral lipid content of the digestive gland-gonad complex of the snail. J. Parasitol., 87: 223-225.
CrossRef - Jarusiewicz, J.A., J. Sherma and B. Fried, 2006. Thin layer chromatographic analysis of glucose and maltose in estivated Biomphalaria glabrata snails and those infected with Schistosoma mansoni. Comp. Biochem. Physiol. B. Biochem. Mol. Biol., 145: 346-349.
PubMed - Iskierko, J. and A. Gorski, 1974. Effect of starvation and low temperature on the content of free amino acids in the leg tissue and haemolymph of Helix pomatia. Acta. Physiol. Polonica, 25: 371-375.
PubMed - Mantawy, M.I., 2001. Potential effect of Allium cepa and Allium sativum on haemolymph of Biomphalaria alexandrina, the intermediate host of Schistosoma mansoni. J. Egypt Soc. Parasitol., 31: 711-723.
PubMed - Mahmoud, M.B., S.M. El-Dafrawy, K.A. El-Sayed and A.T. El-Din, 2002. Effect of Echinostoma liei infection on alterations of protein content and some enzymes in Biomphalaria alexandrina snails. J. Egypt Soc. Parasitol., 32: 361-372.
PubMed - Massa, D.R., J. Michael, C.B. Fried and J. Sherma, 2007. HPLC Analysis of Selected carboxylic acid in B. glabrata patently infected with Schistosoma mansoni. Para Res., 101: 925-928.
CrossRef - Pachuski, J., B. Fried and J. Sherma, 2002. HPTLC analysis of amino acids in Biomphalaria glabrata infected with Schistosoma mansoni. J. Liquid Chromat. Releted Tech., 25: 2345-2349.
Direct Link - Ponder, E.L., B. Fried and J. Sherma, 2003. Effects of hypotonicity on amino acid release in adult Echinostoma caproni as determined by thin layer chromatography. Parasitol. Res., 89: 242-244.
CrossRef - Ponder, E.L., B. Fried and J. Shermal, 2004. Free-pool amino acids in Biomphalaria glabrata infected with Echinostoma caproni as determined by thin-layer chromatography. J. Parasitol., 90: 665-666.
Direct Link - Pinheiro, J., E.M. Gomes and G.M. Changes, 2001. Aminotransferases activity in the haemolymph of Bradybaena similaris (Gastropoda Xanthonychidae) under starvation. Memories Inst. Oswaldlo Cruz, 96: 1161-1164.
CrossRef - Stanislawski, E. and W. Becker, 1979. Alterations of the free amino acid content in the hemolymph of Biomphalaria glabrata (pulmonata) in starvation and after infection with Schistosoma mansoni (trematoda). Comp. Biochem. Physiol., 63: 477-482.
PubMed








