ABSTRACT
Ketorolac (Non-steroidal anti-inflammatory drugs) is associated with gastric ulceration and irritation due to presence of free carboxylic group. The current investigation reports mutual amide prodrug synthesis of ketorolac with Glucosamine, a nutrional supplement for arthritis. The current investigation reports synthesis of mutual amide prodrug of ketorolac by masking free carboxylic group with Glucosamine, a nutrional supplement for treatment of arthritis. Confirmation and characterization of the structure of the synthesized prodrug done by elemental and spectroscopy analysis, m.p., determination of migration parameters (Rf, RM and Rt) by using TLC and HPLC, respectively. Partition coefficient and solubility study confirms its lipophilic character so can be suitable candidate for controlled release delivery. In vitro hydrolytic studies of prodrug confirms good rate of hydrolysis in blood plasma, fecal matter and simulated intestinal fluid while stable in gastric simulated fluid. In vivo pharmacological screening performed on animals. Prodrug with respect to etodolac shows good analgesic, antiinflammatory and antiarthritic activity. The prodrug was assessed for their probable damaging effects by ulcerogeniticity and histopathological analysis. Less ulceration in the gastric region during histopathological study was found when treated with prodrug. The data proves better action of prodrug as compared to ketorolac and are profitable in having less gastrointestinal side effects.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jms.2013.36.42
URL: https://scialert.net/abstract/?doi=jms.2013.36.42
INTRODUCTION
Non-steroidal Anti-inflammatory Drugs (NSAIDs) are widely employed for the treatment of chronic inflammatory diseases, such as arthritis. Long term clinical use of most of the available acidic NSAIDs is strongly limited by their side effects notably gastric irritation, ulceration, bleeding, perforation and in some cases may develop from relatively mild to more serious and potentially life threatening states (Champion et al., 1997; Schoen and Vender, 1989). These gastroenteropathic side effects are generally believed to result from the direct contact effect, local irritation due to the effect of free carboxylic group in the moiety causing local blockage of prostaglandin biosynthesis in the gastrointestinal tract (Cioli et al., 1979). By the reason of presence of free carboxylic group in NSAID’s could not be used as up to its latent (Mishra et al., 2008a; Abdel-Azeem et al., 2009). Couple of literatures suggests to overcome gastric problems associated with NSAIDs by derivatization of carboxylic function into ester and amide prodrugs (Bonina et al., 2002; Halen et al., 2007; Uludag et al., 2011; Shanbhag et al., 1992; Mishra et al., 2008b; Lohade et al., 2009).
Ketorolac (KC) chemically is pyrrolizine carboxylic acid provided as a recemic mixture, is administered systemically for the control of mild to moderate pain as well as of some postoperative and cancer related pain (Brocks and Jamali, 1992). KC like other NSAIDs produces gastrointestinal side effects (Liang and Hsu, 2003). So it is desired to develop a mutual prodrug of KC of the need to overcome related GI side effects. Glucosamine (GLU) is an amino sugar which is body’s basic starting material for production of natural joint component like critical joint lubricant and shock absorber. It has been widely touted as being an effective arthritis treatment and wound healing property (Rubin et al., 2001; Ghodeswar et al., 2003).
Numerous reports of development of mutual amide prodrug using GLU with NSAID’s other than KC were reported (Ghodeswar et al., 2004; Neha et al., 2005). Yet no attempts were made to develop mutual amide prodrug using GLU with KC for possible use in management of arthritis with less or no gastric side effects. Thus present work aims to synthesize mutual amide prodrug of KC by temporarily masking of free carboxylic group of KC with GLU to stabilize prodrug (KC-GLU) in gastric region and further aimed to characterize on chemical and pharmacological basis. Various proteolytic enzymes especially amidase will help in release of KC and GLU by hydrolysis of peptide linkage of KC-GLU in basic pH of intestinal specifically colonic region. So as to produce minimized GIT disturbances while producing synergistic anti-inflammatory, analgesic and antiarthritic activities.
MATERIAL AND METHODS
KC, GLU was procured from Windlas Biotech Limited, Dehradun India. Dicyclohexylcarbodiimide (DCC) was purchased from Himedia lab pvt. Ltd., Mumbai, India. Triethyl amine, benzene, methanol, dimethyl sulfoxide (DMSO), sodium bicarbonate was purchased from SD fine chem., Mumbai, India. Diethylether, HPLC-grade methanol, acetonitrile and water was procured from Merck, Mumbai, India. Distilled water was used throughout the study.
Synthesis of mutual prodrug KC-GLU: Initially activation of carboxylic acid functional group of KC (0.01 mol L-1) was accomplished by using DCC (0.01 mol L-1) in ethanol at 0-5°C for 20 min, resulting a reactive intermediate ketorolac-Oacylisourea. 0.01 mol L-1 Glucosamine solution was prepared with DMSO as cosalvent (4 mL), 2 mL of triethyl amine was added dropwise at 0°C and volume was made upto 20 mL with methanol. Equimolar amount of both prepared solutions of GLU and ketorolac o-acylisourea was mixed with proper stirring at 0°C for 24 h. The white precipitate formed as side product in reaction mixture was filtered off, remaining washed with 10% NaHCO3 and distilled water to remove other impurities. Further extraction with ether was done and crude product was obtained after vaccum evaporation of ether layer. The product was recrystallized with methanol and dried under vacuum. Figure 1 illustrates scheme of synthesis.
Characterization and preformulation studies of the synthesized prodrug: Physical properties of synthesized prodrug, viz., yields, color, odor, aqueous solubility, Rf, Rm and m.p. were noted. Solubility of 10 mg of prodrug was checked in methanol, dichloromethane, ethanol and ether at 37±10°C in glass test tubes. Test tube is gently shaken and solubility was observed. In case of any observed insoluble fraction, the known amount of solvent was further added to ascertain the solubility of the compound. To check confirmation and purity of product the thin layer chromatography was used. The plates of silica gel G were dried and activated. The solvent system methanol: dichloromethane: benzene (4:1.5:0.5) with visualizing agent ninhydrin was used. A dark pink spot on heating at 121°C for 2-3 min in day light observed for Rf and Rm value calculation. Further confirmation and purity of compound checked on Qualisil Gold C18 column injecting on HPLC (LC 2010HT liquid chromatograph, Shimadzu, Japan). The melting point of the drug and synthesized prodrug was determined by capillary fusion method, by using calibrated thermometer and melting point apparatus (Jindal, S.M. Scientific Instruments Pvt. Ltd., New Delhi).
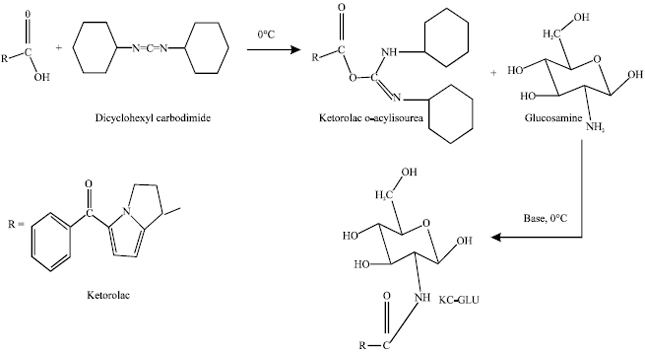 | |
| Fig. 1: | Scheme of synthesis of mutual prodrug from Ketorolac and Glucosamine |
The λmax was determined at double beam UV-Vis spectrophotometer (UV-1700 Pharmspec, Shimadzu, Japan) by using software UV probe ver 2.33. Partition coefficient was determined in n-octanol/phosphate buffer of pH 7.4. The elemental analysis of KC-GLU was performed in Central Drug Research Institute, Lucknow, India, using Carlo-Erba Model 1108 Analyzer. It was carried out to find the percentage of C, H and N in the prodrug. The IR spectra of the compounds were obtained on Spectrum two (FTIR spectormeter, Perkin Elmer, USA) by using software spectrum ver 10.3.02. The 1HNMR and mass analysis of the KC and KC-GLU was done on NMR spectrophotometer (JEOL) at 300MHz using CDCl3 as solvent and on FAB mass spectrometer (JEOL SX 102/DA-6000 mass spectrometer) (at IIT, Kanpur, India).
Protein binding study: Protein binding study of KC-GLU is performed by using cellophane membrane. 100 μg mL-1 prodrug solution was made in phosphate buffer (PBS) at pH 7.4. A 100 mL of this solution was taken in a beaker. The cellophane membrane was washed with distilled water fallowed by buffer solution. Cellophane membrane was tied at the opening end of dialysis tube containing (6%) egg albumin and dipped into the drug solution. The whole assembly was placed on a magnetic stirrer with 100 rpm and at 37°C. After every one hour, 2 mL of the PBS containing drug solution was replaced of the beaker with fresh 2 mL of PBS. Withdrawn sample was diluted further with appropriate amount of phosphate buffer and monitored on UV spectroscopy at 323 nm.
In vitro hydrolysis study: Hydrolysis of prodrug was studied in simulated gastric fluid (SGF, pH 1.2), simulated intestinal fluid (SIF, pH 7.4), PBS (pH 6.8) containing fresh rat faecal content (20% w/v, to provide amidase) and 80% human plasma (pH 7.4). 100 μg mL-1 prodrug solution was prepared in SGF at 37°C. 5 mL solution was withdrawn from the prodrug solution at 15 min. time interval (0-90 min) fallowed by addition of methanol to make up the volume. Upon hydrolysis sample was shaken for 5 min with equal amount of methanol to extract free KC. After centrifugation at 2500 rpm for 5 min all clear supernatant obtained for different time interval was injected into column of HPLC. Mobile phase methanol: water 80:20 was used with flow rate of 0.8 mL min-1, UV detector (D2 lamp) for the amount of free KC at 323 nm. The same procedure was fallowed for SIF and 80% human plasma for hydrolysis of EC-GLU. For reversion study in rat faeces, sample was processed by protein precipitation technique using appropriate amount of acetonitrile.
Pharmacological screening: For all in vivo screening Sprague dawley rats (100-200 g) and Swiss albino mice (20-25 g) were randomly divided into six groups each of 6 rat’s three males and three females, including a control and a two standard group for KC and KC-GLU. The selected animals were housed in acrylic cages at standard environmental conditions at 25±2°C, relative humidity of 45-55%, in a well ventilated room, fed with standard rodent diet and water ad libitum. All the animals were acclimatized for a week before experiment. All the experiments were carried out under the guidelines and approval of Institutional Animal Ethics Committee (IAEC/PSIT/1273/ac/09), Pranveer singh Institute of technology, Kanpur, India.
Anti-inflammatory activity: In vivo anti inflammatory action of prodrug was determined by hind paw oedema method using carrageenan (0.1 mL, 1% w/v) as phlogistic agent (Brodie et al., 1970). The initial volume of right hind paw of rat was measured by plethysmometer without administration of drug. A 1% sodium carboxy methyl cellulose (CMC) suspension containing KC and KC-GLU (equivalent to 1.5 mg kg-1 body weight) was administered orally to the standard groups. Control group administers only 1% CMC suspension. After 30 min of administration of the drug and prodrug, carrageenan solution in normal saline was injected into the planter surface of right hind paw of each animal. The volume of swelling of right hind paw of each rat was measured after 0.5, 1, 2, 4 and 6 h. The mean increase in the volume of the right hind paw of rat was compared with control and standard. The percent inhibition of paw oedema was calculated as:
Percentage inhibition = (1-Vt/Vc)x100 |
where, Vt is mean relative change in paw oedema volume in test group and Vc is mean relative change in paw oedema volume in control group.
Analgesic activity: The analgesic activity of the KC and KC-GLU was determined by acetic acid induced writhing method in mice (Ghosh, 1984). Swiss albino mice were divided into three groups (n = 6 in each group). Dose equivalent to 1.5 mg kg-1 body weight was administered to control and standard groups. Three hours after treatment, 0.6% (v/v) acetic acid solution (10 mL kg-1) was injected to mice intraperitoneally. Total number of writhes i.e., constriction of abdomen, turning of trunk and extension of hind limbs was counted for 20 min. The analgesic effect was expressed as percent reduction of writhes in comparison with the control.
Antiarthritic activity: Arthritis was induced in the left hind paw of rats by the intraplantar injection of 0.1 mL of Complete Freund’s Adjuvant (Vogel and Vogel, 1997). The adjuvant contained heat killed Mycobacterium tuberculosis in sterile paraffin oil (10 mg mL-1). The paw volume of all the animal groups was measured by plethysmograph at 0, 7, 14 and 21 days after the injection of Freund’s complete adjuvant.
Ulcerogenic activity: Gastrointestinal toxicity of the KC-GLU was observed and compared with the parent drug by measuring mean ulcer index (Rainsford, 1977). The rats were divided into six groups, each comprising six animals, including a control and standard group. The control group was administered orally by 1% CMC. Standard KC and KC-GLU was administered orally (dose equivalent to 5 mg kg-1 body weight) as a suspension with 1% CMC daily for 5 days. The rats were fasted after the administration of last dose, were sacrificed by decapitation and the stomach was removed. Further the stomach was opened and washed using distilled water. A binocular magnifier was used to count the lesions on the gastric mucosa. Ulcers found greater than 0.5 mm were recorded. The size of ulcers was measured by Microimage process software (DA1-180M v 2.01; Sunny International United Co., Ltd, Zhejiang, China), using an Olympus SP 350 camera (Olympus, Tokyo, Japan). Ulcers were scored as: 0 for normal-coloured stomach; 0.5 for red colouration; 1 for spot ulcers; 1.5 for haemorrhagic streaks; 2 for ulcers of 3 mm up to 5 mm and 3 for ulcers of 5 mm and greater (Kulkarni, 2000) and mean ulcer index calculated as:
UI = 1x(number of lesions of grade 1)+2x(number of lesions of grade 2)+3x(number of lesions of grade 3)/10 |
Histopathological study: The histopathological studies of stomach of rats were carried out using haemotoxylin and eosin stain. The removed stomach tissues of rats were fixed in 10% normal saline solution for at least 48 h. Further processed routinely and the tissues were surrounded in paraffin wax. Histological sections were cut at 5-6 μm and stained with routine haematoxylin and eosin, observed for necrosis and ulcer.
RESULTS
Characterization and preformulation studies of the synthesized prodrug: Single step synthesis of mutual amide prodrug of KC and GLU was performed resulting more than 89%. Initial characterization of KC-GLU was performed by TLC and HPLC. By the physiochemical properties, elemental and spectral analysis (Table 1) of KC-GLU confirms synthesis, molecular structure and molecular formula of KC-GLU. The synthesized prodrug of KC was subjected to solubility, partition coefficient and hydrolytic studies. KC-GLU was freely soluble in methanol, dichloromethane, soluble in ethanol and less soluble in ether. Rt value for KC and KC-GLU was found 4.11 and 11.1 min, respectively. Partition coefficient value of KC-GLU is 1.10. Protein binding of prodrug was found 11% less in comparison to pure KC. The Hydrolytic release of free KC after hydrolysis of KC-GLU was observed in SGF, SIF, PBS (pH 6.8) containing fresh rat fecal content and in 80% human plasma. The hydrolytic release is 13, 63, 72 and 79%, respectively. The t1/2 value of KC-GLU in SIF, PBS with rat feces and 80% human plasma is 139, 101 and 67 min, respectively.
Pharmacological screening: The synthesized prodrug along with KC was evaluated for analgesic, anti-inflammatory, ulcerogenic, antiarthritic and histopathology study. Table 2 reports data of pharmacological screenings for oral dose of control, KC and KC-GLU. In analgesic study, the KC-GLU has better pain reduction after 3 h in comparison to KC. The anti-inflammatory activity is reported after 6 h and is better than KC. In antiarthritic activity on 21st day percentage Inhibition of edema for KC and KC-GLU is 28.8 and 46.9%, respectively. The ulcerogenic index observed for prodrug is more than KC. The stomach walls of rat in ulcerogenic study are presented in Fig. 2. The animals treated with only KC have deep hemorrhagic spots and necrotic cells while animals treated with KC-GLU have small red spots but no necrosis. On comparing histopathology of the stomachs of control rats (Fig. 3a) and those treated with KC (Fig. 3b) and KC-GLU (Fig. 3c), more severe ulcers and necrosis were evident in the drug group than the prodrug group.
| Table 1: | Physicochemical properties and spectral data of synthesized mutual prodrug of Ketorolac |
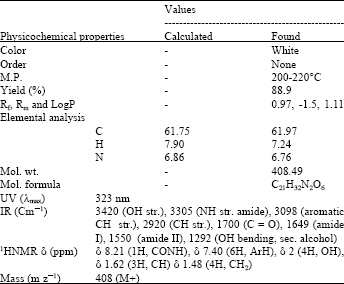 | |
DISCUSSION
Coupling reagent based amide synthesis provides direct activation of carboxylic group of KC to generate its active form which can react with amine group of GLU. The single step synthesis results high yield. The different migration parameter (Rf, Rm) of product than KC by TLC confirms synthesis of new product. Further by spectral analysis molecular structure of new compound is confirmed as KC-GLU. The high solubility of prodrug in comparison to standard drug is might be due to presence of amide linkage. Due to this transcellular absorption by lipid membrane permeation might be limited in gastric region. This would facilitate delivery of intact prodrug to lower intestinal region. C18 column used as the stationary phase had more affinity for nonpolar drugs and higher RM, logP and Rt value for KC-GLU with respects to parent drug supports it’s more lipophilic nature. Lesser protein binding for KC-GLU indicates more availability of the KC-GLU for hydrolysis in plasma and the required dose will be less so as to minimize dose related effect on prostaglandin formation. In SGF negligible amount of prodrug hydrolysis observed while in SIF and in PBS at colonic pH with rat’s fresh fecal content indicates its better hydrolysis. It may be due to presence of basic pH and amidase enzyme. The lower t1/2 value of KC-GLU in 80% human plasma is due to presence of different active proteolytic enzyme to break amide linkage.
Pharmacological screening: In analgesic study, significant reduction in number of writhes is observed after treatment with KC and KC-GLU. It indicates initially after 2 h of administration advantage of KC over KC-GLU but after 3 h equimolar dose of KC-GLU to KC has better analgesic activity. It may be due to stability of prodrug in gastric pH. Percentage inhibition of edema after 6 h and antiarthritic activity on 21st is found better for prodrug with respect to drug. GLU has anti inflammatory property apart from antiarthritic activity (Yomogida et al., 2008) which is responsible for this synergistic effect on inflammation and arthritis. The minimized ulcerogenic index of prodrug and less hemorrhagic spots, necrosis in histopathology study might be resulting due to inhibition of direct contact effect of free carboxyl group of the ketorolac to the gastric mucosa. It is also due to negligible hydrolysis of KC-GLU in stomach (pH 1.2) region as well.
| Table 2: | Data for In vivo study |
 | |
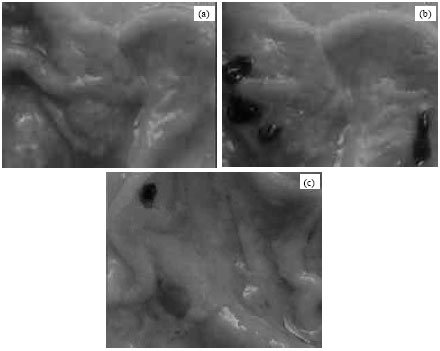 | |
| Fig. 2(a-c): | Evaluation of ulcerogenic activity in the stomach of rats treated with (a) Drug vehicle 1% w/v CMC, (b) KC (5 mg kg-1 body weight) and (c) Mutual prodrug KC-GLU (equivalent to 5 mg kg-1 body weight) |
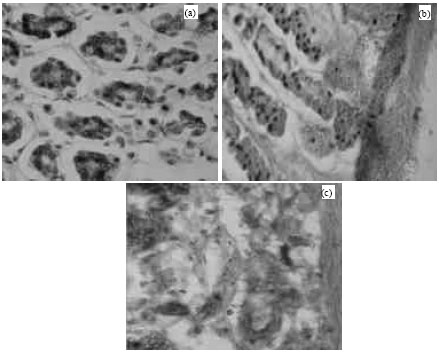 | |
| Fig. 3(a-c): | Histopathology of Sprague dawley rat stomach after 14 days treated with (a) 1% w/v CMC, (b) KC (5 mg kg-1 body weight) and (c) Mutual prodrug (equivalent to 5 mg kg-1 body weight) |
CONCLUSION
In conclusion the synthesized prodrug had increased solubility, synergistic antiinflammatory and antiarthritic activity with lower toxicity and less ulcerogenic activity than the parent drug KC. Thus, this prodrug approach solves not only the KC related gastric adverse effects but also synergizes the pharmacological activities. Moreover, lipophilic nature of prodrug makes it a suitable candidate for controlled release delivery.
REFERENCES
- Abdel-Azeem, A.Z., A.A. Abdel-Hafez, G.S. El-Karamany and H.H. Farag, 2009. Chlorzoxazone esters of some non-steroidal anti-inflammatory (NSAI) carboxylic acids as mutual prodrugs: Design, synthesis, pharmacological investigations and docking studies. Bio. Med. Chem., 17: 3665-3670.
CrossRefDirect Link - Bonina, F., C. Puglia, N.A. Santagati, A. Saija, A. Tomaino and B. Tita, 2002. Oligoethylene ester derivatives of ketoprofen, naproxen and diclofenac as oral prodrugs: A pharmacological evaluation. Pharmazie, 57: 552-555.
PubMedDirect Link - Brodie, D.A., P.G. Cook, B.J. Bauer and G.E. Dagle, 1970. Indomethacin-induced intestinal lesions in rats. Toxicol. Pharmacol., 17: 615-624.
Direct Link - Champion, G.D., P.H. Feng, T. Azuma, D.E. Caughey and K.H. Chan et al., 1997. NSAID induced gastrointestinal damage. Epidemiology risk and prevention, with an evaluation of the role of Misoprostol: An Asia-Pacific perspective and consensus. Drugs, 53: 6-19.
PubMedDirect Link - Cioli, V., S. Putzolu, V. Rossi, P. Scorza Barcellona and C. Carrandino, 1979. The role of direct tissue contact in the production of gastrointestinal ulcers by antiinflammatory drugs in rats. Toxicol. Applied Pharmacol., 50: 283-289.
CrossRefDirect Link - Brocks, D.R. and F. Jamali, 1992. Clinical pharmacokinetics of ketorolac tromethamine. Clin. Pharmacokin., 23: 415-427.
PubMedDirect Link - Ghodeswar, B.C., M.R. Bhojani and S.S. Dhaneshwar, 2003. Synthesis and biological evaluation of glucosamine conjugate prodrug of flurbiprofen. Ind. Drugs, 40: 156-159.
Direct Link - Ghodeswar, B.C., R.N. Pophalikar, M.R. Bhojani, D. Nagpal and S.S. Dhaneshwar, 2004. Synthesis and pharmacological evaluation of mutual prodrugs of some nonsteroidal antiinflammatory drugs with glucosamine. Ind. J. Pharm. Sci., 66: 773-777.
Direct Link - Halen, P.K., K.K. Chagti, R. Giridhar and M.R. Yadav, 2007. Substituted aminoalcohol ester analogs of indomethacin with reduced toxic effects. Med. Chem. Res., 16: 101-111.
CrossRefDirect Link - Liang, T.H. and P.N. Hsu, 2003. Double-blind, randomised, comparative trial of etodolac SR versus diclofenac in the treatment of osteoarthritis of the knee. Curr. Med. Res. Opin., 19: 336-341.
Direct Link - Uludag, M.O., B.C. Ergun, D.A. Alkan, N. Ercan, G.Y. Ozkan and E. Banoglu, 2011. Stable ester and amide conjugates of some NSAIDs as analgesic and antiinflammatory compounds with improved biological activity Turk. J. Chem., 35: 427-439.
CrossRef - Mishra, A., R. Veerasamy, P.K. Jain, V.K. Dixit and R.K. Agrawal, 2008. Synthesis, characterization and pharmacological evaluation of amide prodrugs of ketorolac. Eur. J. Med. Chem., 43: 2464-2472.
CrossRefDirect Link - Neha, G., N. Deepika, S.D. Suneela, S.R. Dhaneshwar and S.C. Chaturvedi, 2005. Synthesis, hydrolysis, kinetics and pharmacodynamic profiles of novel prodrugs of flubiprofen. Ind. J. Pharm. Sci., 67: 369-373.
Direct Link - Rainsford, K.D., 1977. Comparative Studies of Gastric Ulcerogenesis by Non-steroid Anti-inflammatory Drugs. Proc. R. Soc. Med., 70: 4-10.
Direct Link - Mishra, A., V. Ravichandran, R.K. Agrawal, V.K. Dixit and R.K. Agrawal, 2008. Synthesis, characterization and pharmacological evaluation of amide prodrugs of flubiprofen. J. Braz. Chem. Soc., 19: 89-100.
Direct Link - Rubin, B.R., J.M. Talent, P. Kongtawelert, R.M. Pertusi, M.D. Forman and R.W. Gracy, 2001. Oral polymeric N-acetyl-D-glucosamine and osteoarthritis. J. Am. Osteopath Assoc., 101: 339-344.
Direct Link - Schoen, R.T. and R.J. Vender, 1989. Mechanisms of nonsteroidal anti-inflammatory drug-induced gastric damage. Am. J. Med., 86: 449-458.
CrossRefDirect Link - Shanbhag, V.R., A.M. Crider, R. Gokhale, Harpalani and R.M. Dick, 1992. Ester and amide prodrugs of ibuprofen and naproxen: Synthesis, anti-inflammatory activity and gastrointestinal toxicity. J. Pharm. Sci., 81: 149-154.
Direct Link - Yomogida, S., J. Hua, K. Sakamoto and I. Nagaoka, 2008. Glucosamine suppresses interleukin-8 production and ICAM-1 expression by TNF-alpha-stimulated human colonic epithelial HT-29 cells. Int. J. Mol. Med., 22: 205-211.
PubMedDirect Link








