ABSTRACT
Sixty seven female patients who had underwent axillary lymph node dissection of at least level II, for invasive breast cancer were included in this study to evaluate incidence and risk factors (age, tumor size, surgical procedure, No. of axillary nodes removed and No. of positive nodes removed) for breast cancer-related arm lymph edema. Fifteen patients (22.4%) developed clinical lymph edema (grade 2-4) at one year postoperative. The age of the patients at time of treatment constitute one of significant risk factors for developing lymph edema (33.3% of patients older than 55 years developed lymph edema vs. 13.5% in patients younger than 55). The extent of axillary dissection as defined by the No. of nodes dissected is another risk factor (where lymph edema development was 11.8% in patients with <10 lymph nodes removed, it was 23.7% in patients with 10-15 lymph nodes removed and 33.3% in patients with >15 lymph nodes) also No. of positive lymph nodes removed (while 28.6% of patients with 1-3 positive lymph nodes developed lymph edema, 36.4% of patients with more than 3 positive nodes shows postoperative lymph edema). So, age of the patients, No. of lymph nodes dissected and No. of positive nodes was statistically significant factors leading to the development of arm edema. While tumor size and type of surgical procedure, were not significantly related to post mastectomy lymph edema. So, these risk factors should be taken into account in clinical practice to reduce the incidence of postmastectomy lymph edema.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jms.2008.498.502
URL: https://scialert.net/abstract/?doi=jms.2008.498.502
INTRODUCTION
Lymph edema is a relatively common, potentially serious and unpleased complication after axillary lymph node dissection (ALND) for breast cancer. It may be associated with functional, esthetic and psychological problems, thereby affecting the Quality-of-Life (QOL) of breast cancer survivors (Sakorafas et al., 2006).
Breast cancer-related lymph edema (BCRL) occurs as a consequence of surgery and/or radiotherapy to axillary lymph nodes. However, its pathophysiology is poorly understood and likely to be complex and multifactorial (Purushotham et al., 2007).
Lymph edema is a chronic condition with estimates of incidence ranging from 6 to 83%. Caused by the abnormal accumulation of protein-rich fluid in the interstitial space due to inadequate lymphatic drainage. Clinical manifestations include swelling, fibrosis and hardening of affected tissues, leading to decreased joint mobility, pain and discomfort. The static protein-rich environment promotes bacteria, increasing the risk of infection (Clark et al., 2005). Although it is clear that surgical removal of lymph nodes from the axilla is the single most important event in BCRL, the pathophysiological mechanisms that cause edema are poorly understood and would need to account for a No. of clinical observations, such as an onset that is often delayed for months or even years and the phenomenon of sparing, in which parts of an otherwise swollen arm (often the hand) remain unaffected. Several pathophysiological observations are also unexplained, such as the finding of a protein concentration in the interstitial fluid of the epifascia of the swollen arm that is lower than would be expected in edema of reduced lymph flow (Simon et al., 2005).
The aim of this study was to examine incidence and risk factors (age, tumor size, surgical procedure, No. of axillary nodes removed and No. of positive nodes removed] for breast cancer-related arm lymph edema.
MATERIALS AND METHODS
From October 2002 to April 2007, 67 female patients who had undergone axillary lymph node dissection of at least level II, for invasive breast cancer in Tadawi general hospital were included in this study. Exclusion criteria was loco regional recurrence, if both breasts were treated, if the patient had not participated in follow up for more than six months and if radiotherapy given to the axilla. Patient characteristics are listed in Table 1.
Mean age was 55.8 years (range 29-73 years).
| Table 1: | Patient characteristics in this study |
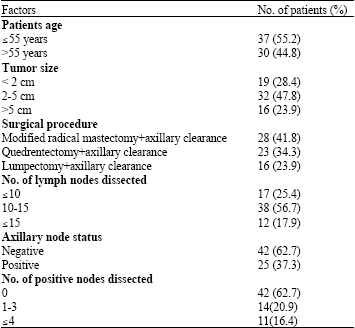 | |
Surgical procedures: Modified radical mastectomy plus axillary clearance 28 patients (41.8%), quadrentectomy with axillary clearance 23 patients (34.3%), lumpectomy with axillary clearance 16 patients (23.9%).
The circumference of the arm was measured 15 cm above the olecranon. Process and 10 cm below the olecranon process. Measurements more than 1 cm different. From the measurement recorded at the previous examination were confirmed by a second observer. Measurements were scored as edema if the circumference of either the upper arm or the lower arm was more than 1 cm greater than the measurement at the last examination. Edema was defined as follows: grade 1, difference from previous measurement greater than 1 cm in the nondominant arm or greater than 1.5 cm in the dominant arm; grade 2, difference of 2 cm or more between arms; grade 3, symptomatic edema necessitates intervention with compression and grade 4, edema causes loss of arm function (Kiel and Rademacker, 1996). Hand edema was not scored in this study unless it was accompanied by changes in arm circumference. Results were reported when clinically significant edema (grades 2-4) is present. Several instances of grade 2 edema resolved to grade 1 edema after treatment (with antibiotics, compression stockings or compression pumps). No patient experienced complete persistent resolution of edema.
Measurements were undertaken 1, 6 and 12 months postoperatively. Baseline arm measurements were taken during surgical pre-assessment. Data for age, axillary node status, No. of axillary nodes removed, type of surgery and radiotherapy were extracted from routinely collected clinical information. None of the 67 patients studied were treated with radiotherapy to the axillary lymph nodes. Actuarial incidence was determined with Kaplan-Meier product limit curves, curves were compared between subgroups by means of the log-rank test (SAS statistical system, SAS (statistical analysis software) Institute, Cary, NC).
RESULTS
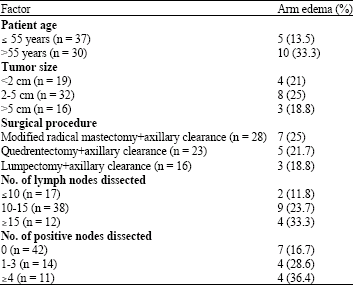
During the follow-up period, 15 patients (22.4%) developed clinical lymph edema (grade 2-4) at one year postoperative, of this 6(40%) were developed lymph edema by 6 months postoperatively and 9 (60%) were developed lymph edema by 12 months. Table 2 shows incidence of lymph edema in relation to patient`s factors.
The mean age of the patients was 55.8 years range from 29 to 73 years and the median follow-up period was 15 months. The mean age of the patients in the lymph edema group at the time of surgery was 59 years, compared with 57 years for those patients who were free of lymph edema. Thirty seven patients were 55 years or less at time of treatment (55.2%), while 30 patients were more than 55 years (44.8%). The age of the patients at time of treatment was statistically significant factor leading to the development of arm edema, 10 women older than 55 years develop arm edema; the incidence of arm edema in this group was 33.3% and 5 women younger than 55 years developed arm edema 13.5% (p = 0.0005). The extent of axillary dissection as defined by the No. of nodes dissected has influenced the probability of arm edema as edema increases as long as No. of lymph nodes removed increase.
| Table 2: | Incidence of lymph edema in relation to patient`s factors |
 | |
| Table 3: | Factors affecting arm edema |
 | |
Also No. of positive lymph nodes removed has great influence on occurrence of lymph edema. While type of surgery shows no association on edema incidence. Also tumor size shows no significant relation to arm edema.
Univariate analysis was performed on factors that led to edema (Table 3) age greater than 55 years, positive nodes and extent of axillary dissection were the three leading factors that led to arm edema. While tumor size and type of surgical procedure shows no significant effect on arm edema.
DISCUSSION
In this study 15 patients had developed lymph edema (22.4%) at one year after surgery, incidence shows wide variations in literature, Clark et al. (2005) found incidence of lymph edema in a series of 183 patients followed for 3 years to be 20.7% of this 80% developed lymph edema at one year.
Kwan et al. (2002) found that 12.5% of randomly selected patients had demonstrated lymph edema. In addition, Sarah et al. (2002) in a retrospective cohort study, of 151 women surgically treated for early-stage breast cancer (stages 0-II) reported that lymph edema was present in 42 women (27.8%): 28.2% of women treated with mastectomy and 27.5% of women treated conservatively, all with axillary lymph node dissection.
Kissin et al. (1986) reported that lymph edema (measured by limb volume) was present in 25% of the members of a cohort of 200 patients after a variety of surgical treatments for breast cancer overall and in 38% of patients receiving axillary node dissection plus radiation therapy. Virginia et al. (2001) summarizes reports of 10 studies of lymph edema incidence following a variety of surgical procedures and adjuvant therapies and reported that the overall incidence of arm edema was 26%, with a range from 0% with partial or total mastectomy and sentinel node biopsy to 56% 2 years after surgery (modified radical mastectomy or breast-conserving surgery with maxillary radiation therapy) and axillary dissection, with overall prevalence of lymph edema after modern breast cancer treatment at 10-20%.
Also incidence of lymph edema increase over time in this study 40% of lymph edema appears at 6 months and 60% developed at one year. In a longitudinal study of 93 patients after breast cancer surgery, Tasmuth et al. (1996) found the prevalence of arm edema to increase from 22% at 1 month after surgery to 36% at 1 year. The timing of onset of lymph edema following breast cancer treatment varies. Guedes Neto 1997 found that 73% of 142 patients with arm edema had developed the edema within 1 year of treatment for breast cancer. Werner et al. (1991) found that the mean time from treatment to development of lymph edema was 14 months (range, 2-92 months).
In this study the age of the patients, extent of axillary dissection (as reflected by the No. of nodes in the axillary dissection specimen) and No. of positive nodes dissected were the only significant factors affecting arm edema. Present results add further support to previous reports correlating lymph edema with physical and pathological risk factors, Kiel and Rademacker (1996) found that the actuarial probability of edema was predicted by age, No. of lymph nodes dissected and No. of positive lymph nodes dissected in a cohort of 183 patients treated with breast-conserving surgery and breast radiation therapy. Liljegren and Holmberg (1997) found that only age and No. of lymph nodes excised predicted No. of arm problems (edema or subjective arm symptoms) in a multivariate model: The relative risk of arm problems was 0.93 per year of increasing age (95% CI = 0.91 to 0.97) and 1.11 per lymph node excision (95% CI = 1.05 to 1.18).
Borup and Lundgren (1989) in a study of 100 patients who had undergone partial or total mastectomy, found that arm edema was associated with degree of axillary surgery (30% of 47 patients with axillary dissection developed arm edema versus none of the 48 patients with axillary sampling).
Present results were contradictory to results of Clark et al. (2005) who found that Hospital skin puncture (vs. none), mastectomy (vs. wide local excision or lumpectomy) and body mass index 26 (vs. BMI 19-26) were the only significant risk factors, he stated that other features previously postulated as risk factors (surgery on dominant side, age, axillary node status, No. of axillary nodes removed, radiotherapy) were not associated with lymph edema in his study.
The results of Purushotham et al. (2007) are counterintuitive to present results and No. of retrospective studies, their results suggests that positive node status was significantly inversely associated with arm volume. Furthermore, the No. of positive nodes also correlated inversely with arm volume. These results are counterintuitive to the conventional understanding of the pathophysiology of breast cancer related lymph edema.
In this study stage of breast cancer and type of surgical procedure were not relevant to postoperative lymph edema. These results are supported by Gerber et al. (1992) who reported no significant difference in the extent of arm edema among 237 patients randomly assigned to receive either modified radical mastectomy or local excision, axillary dissection and radiation therapy.
Sarah et al. (2002) found lymph edema (an arm volume difference 200 cm3) was measured in 42 women (27.8%). Mastectomy or conservative surgery patients had similar lymph edema rates.
From univariate analysis done by Kasse et al. (1999), he found 7 factors associated with lymph edema: The big size of the tumor (p = 0.005), clinically involved axillary lymph nodes (p = 0.001), metastatic disease (p = 0.0046), traditional or inadequate surgery out of the Institute (p = 0.001), lack of post-operative chemotherapy (p = 0.002), postoperative external beam radiations (p = 0.005), relapse (p = 0.002).
Also Herd-Smith et al. (2001), reported that radiotherapy, the No. of lymph nodes removed and the size of the tumor were identified as being significant prognostic factors that appear to increase the risk of lymph edema of the arm in patients who undergo dissection of the axillary lymph nodes.
Soran et al. (2006) investigated lymph edema related risk factors (occupation/hobby (hand use), TNM stage, No. of dissected nodes, No. of positive nodes, tumor size, infection, allergy, diabetes mellitus, hypertension, hypothyroidism, chronic obstructive pulmonary disease and body mass index) and concluded that the risk and severity of lymph edema was statistically related to infection, BMI and level of hand use.
CONCLUSION
According to this study the risk of lymph edema after breast cancer surgery is related to age of the patient, No. of lymph nodes removed and No. of positive nodes removed. These risk factors should be taken into account in clinical practice.
REFERENCES
- Borup C.S. and E. Lundgren, 1989. Sequelae of axillary dissection vs. axillary sampling with or without irradiation for breast cancer. A randomized trial. Acta Chir Scand, 155: 515-519.
Direct Link - Clark, B., J. Sitzia and W. Harlow, 2005. Incidence and risk of arm edema following treatment for breast cancer: A three-year follow –up study. Q.J.M., 98: 343-348.
Direct Link - Gerber, L., M. Lampert, C. Wood, M. Duncan and T. D'Angelo et al., 1992. Comparison of pain, motion, and edema after modified radical mastectomy vs. local excision with axillary dissection and radiation. Breast Cancer Res. Treat, 21: 139-145.
CrossRef - Herd-Smith A., A. Russo, M.G. Muraca, M.R.D. Turco and G. Cardona, 2001. Prognostic factors for lymph edema after primary treatment of breast carcinoma. Cancer, 92: 1783-1787.
Direct Link - Kasse A.A., M. Diop, M. Dieng, A. Deme and D. Ndaw et al., 1999. Risk factors for lymph edema of the arm after mastectomy for breast cancer. Dakar Med., 44: 32-35.
Direct Link - Kiel, K.D. and A.W. Rademacker, 1996. Early-stage breast cancer: Arm edema after wide excision and breast irradiation. Radiology, 198: 279-283.
Direct Link - Kissin, M.W., G.Q. Della Rovere, D. Easton and G. Westbury, 1986. Risk of lymph edema following the treatment of breast cancer. Br. J. Surg., 73: 580-584.
CrossRef - Sakorafas G.H., G. Peros, L. Cataliotti and G. Vlastos, 2006. Lymph edema following axillary lymph node dissection for breast cancer. Surg. Oncol., 5: 153-165.
Direct Link - Sarah M.B., A.M. Lindsay, E.S. Thayer, W.L. Wayne and T.K. Maureen, 2002. Lymph edema and quality of life in survivors of early-stage. Breast Cancer Arch. Surg., 137: 1253-1257.
Direct Link - Simon J.P., W.B. Robert, K.S. Chandra, R.B. James and T.B. Britton, et al., 2005. Short-term effects of axillary lymph node clearance surgery on lymphatic physiology of the arm in breast cancer. J. Appl. Physiol., 99: 2345-2351.
Direct Link - Soran, A., G. D'Angelo, M. Begovic, F. Ardic and A. Harlak et al., 2006. Breast cancer-related lymph edema-what are the significant predictors and how they affect the severity of lymph edema. Breast J., 12: 536-543.
CrossRef - Tasmuth T., K.V. Smitten and E.P. Kalso, 1996. Symptoms during the first year after radical and conservative surgery for breast cancer. Br. J. Cancer, 74: 2024-2031.
Direct Link - Virginia S.E., L.P. Marjorie, A.G. Patricia, J. Adams and K.L. Kahn, 2001. Arm Edema in Breast Cancer Patients. J. Natl. Cancer Inst., 93: 96-111.
Direct Link - Werner R.S., B. McCormick, J. Petrek, L. Cox and C. Cirrincione et al., 1991. Arm edema in conservatively managed breast cancer: Obesity is a major predictive factor. Radiology, 180: 177-184.
Direct Link - Kwan W., J. Jeremy, M. Lorana, C. Dingee and G. McGregor et al., 2002. Chronic arm morbidity after curative breast cancer treatment: Prevalence and impact on quality of life. J. Clin. Oncol., 20: 4242-4248.
Direct Link








