ABSTRACT
Intravenous Patient Controlled Analgesia (PCA) with morphine is often used for postoperative analgesia in morbidly obese patients. However, the required doses may increase postoperative respiratory disorders. Adjunction of small doses of ketamine could reduce dose and related side effects. Eighty morbidly obese patients undergoing upper abdominal surgery were randomly assigned to receive (during the postoperative period) either intravenous morphine 1 mg mL-1 (morphine group: M group) or morphine with ketamine 1 mg mL-1 for each (morphine ketamine group: MK group). Morphine consumption was evaluated by cumulative doses every 8 h for 48 h together with Visual Analogue Scale (VAS) scores at rest and at mobilization. Forced Vital Capacity (FVC) and Forced Expiratory Volume in first second (FEV1) were assessed by spirometric evaluation 24 and 48 h after initiation of PCA. Incidence of adverse effects was reported. cumulative morphine consumption was significantly reduced in morphine Ketamine (MK) group. It was 84 (9) mg vs 66(6) mg 48 h after PCA (p<0.05). VAS in both groups were similar at rest and at mobilization. Compared with preoperative values, (FVC) was significantly reduced in M group than MK group: 46% (8) vs 62% (9) (p< 0.05) at 24 h and 58 (7) vs 77% (11) (p<0.05) at 48 h (FEV1) was 60% (9) vs 81% (7) (p<0.05) at 24 h and 62% (6) vs 87% (11) (p<0.05) at 48 h after PCA initiation. PaO2, respiratory rate, and SPO2 were significantly lower in M group than MK group 32 h after PCA initiation (p<0.05). Incidence of nausea and desaturation were significantly higher in M group than MK group (p<0.05). Adding small doses of ketamine to morphine for PCA decreased cumulative morphine consumption, improved postoperative respiratory function and was associated with less adverse effects in morbidly obese patients undergoing upper abdominal surgery.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jms.2008.364.370
URL: https://scialert.net/abstract/?doi=jms.2008.364.370
INTRODUCTION
Incidence of morbid obesity is increasing and so, their need for surgery is also increasing. In this population optimal post-operative pain control is of considerable importance because obesity is an independent risk factor for cardiovascular and respiratory complications after surgery (Derzie et al., 2000). Moreover, morbidity and mortality of morbidly obese patients after upper abdominal procedures were more than two and half times higher than that of their non obese counterparts (Postlethwait and Johnson, 1972). Few clinical trials have evaluated different types of analgesia in morbidly obese patients after abdominal surgery (Roshanak et al., 2003).
The use of epidural analgesia with local anaesthetic and/or opioid provides superior dynamic pain relief (Keblet and Holte, 2001) which in turn improves respiratory function resulting in less postoperative pulmonary complication (Ballantyne et al., 1998).
Nevertheless and related to technique difficulties or patients refusal, morphine analgesia is still widely used despite a potential harmful influence on postoperative functions (Stone et al., 1999).
The large doses needed to provide an optimal analgesic control may be associated with impairment in pulmonary function that could counteract the analgesic benefit (Catley et al., 1985).
Morphine alone is not always successful in this context. Indeed, the nociceptive inputs of patients (especially those in the Surgical Intensive Care Unit (SICU)) have additional sources and severities beyond those created by tissue injuries. Pathological pain states mainly hyperalgesia and allodyna, can be induced. Consequently, morphine may be less effective despite large consumption. This tolerance to morphine is an early process favored by paradoxical nociceptive stimulation (Guillou et al., 2003).
The role of N-Methyl D-Aspartate (NMDA) excitatory glutamate receptors has been established in nociceptive transmission (Petrenko et al., 2003; Cairns et al., 2003). Ketamine is a potent NMDA receptor inhibitor and represents a promising modality in several strategies to prevent pathological pain.
The potential benefit of adding ketamine to morphine for Patient Controlled Analgesia (PCA) has been reported by randomized controlled studies in several types of surgery (Guillou et al., 2003; Petrenko et al., 2003; Cairns et al., 2003; Michelet et al., 2007).
The aim of this double blinded, randomized study was to investigate if the addition of ketamine to intravenous morphine for PCA could result in sparing effect in opioid consumption and improvement in postoperative pain and respiratory disorders in morbidly obese patients after upper abdominal surgery.
MATERIALS AND METHODS
This prospective, randomized and double blinded study was conducted at Kasr Al-Aini Hospital (Cairo University Hospital), from 2005 to 2007. After local ethics committee approval, written informed consent was obtained from 80 morbidly obese patients [Body Mass Index (BMI)>35 kg m-2], aged 20-65 years, (ASA I, II) undergoing upper abdominal surgery.
Exclusion criteria included diagnosed obstructive sleep apnea syndrome, history of drug abuse, significant cardiopulmonary disease, renal impairment (serum creatinine >1.5 mg dL-1) abnormal liver enzymes (transaminases>1.5x normal) and patients who are unable to understand the use of Patient Controlled Analgesia (PCA) were also excluded.
Patients were instructed on the use of Visual Analogue Scale (VAS) in the preanaesthetic visit. They assessed pain using (VAS) ranging from 0 (no pain)to 100 (worst imaginable pain).
Baseline measurements of Forced Vital Capacity (FVC) and forced expiratory volume in first second (FEV1) were measured bed-side (using Zan 300 co-infusion spirometry) before surgery.
All patients received midazolam (3 mg) i.v., 30 min. before induction of anaesthesia.
An anaesthetic management was standardized for all patients, performed with fentanyl 1.5 mg kg-1 and propofol 2 mg kg-1 until loss of consciousness, rocuronium 0.9 mg kg-1 of Ideal Body Weight (IBW) was administered while applying cricoid pressure. Trachea was intubated 60 seconds later.
The lungs were ventilated with a mixture of oxygen/air (FiO2 = 0.5). Tidal volume was set at 10 mL kg-1 (IBW) with peak airway pressure kept below 35 cm H2O. Respiratory rate was adjusted to maintain normocapnia.
Anaesthesia was maintained with isoflurane, fentanyl and rocuronium titrated according to patients needs. Additional analgesia such as nonsteroidal anti-inflammatory drugs, regional or local anaesthetic techniques were not allowed during the operative period.
After the operation, patients were transferred to the Surgical Intensive Care Unit (SICU). Tracheal extubation was postponed until the next morning to ensure adequate ventilation. All patients were mechanically ventilated first with Synchronized Intermittent Mandatory Ventilation (SIMV) followed by Continuous Positive Airway Pressure (CPAP) to allow gradual weaning.
During that time patients received incremental doses of morphine (5 mg) and midazolam (2 mg) to control pain and anxiety.
VAS was assessed hourly before initiation of any pain treatment and recorded by the SICU nurse through the night.
Next morning, patients were assessed for possibility of weaning from mechanical ventilation and extubation. After extubation patients were placed in a 30° back-up Fowler position breathing oxygen through face mask to keep their oxygen saturation up to 95%.
All patients received PCA device (P 5000 IVAC-PCAM) containing either morphine 1 mg mL-1 (morphine group: M group) or morphine with ketamine 1 mg mL-1 each (morphine ketamine group: MK group) as the optimal combination of morphine-ketamine has been reported to be 1:1 (Sveticic et al., 2003). The stability of this solution has been tested with success previously at a wide range of PH values for at least 4 days (Schmid et al., 2002). Patients were randomized according to closed envelope method.
A nurse not involved in the care of the patients prepared the syringes of morphine or morphine ketamine. Patients were instructed to achieve maximal comfort in order to breathe and cough without substantial pain. They were allowed to have bolus doses of morphine or morphine and ketamine at the dosage of 0.015 mL kg-1 IBW at 10 min interval.
If pain control was inadequate, diclofenac 75 mg was administered in order to lower the VAS to less than 40 at mobilization. It was considered as rescue analgesia and recorded as such.
The following variables were recorded and assessed: The 8 h cumulative morphine consumption over 48 h period, the VAS at rest and at mobilization every 8 for 48 h.
Haemodynamic values (Arterial blood pressure, heart rate), respiratory rate and oxygen saturation were continuously monitored and recorded.
Bed-side pulmonary functions (FVC and FEV1) were measured preoperatively and repeated 24 and 48 h after initiation of PCA.
Arterial Blood Gases (ABG) were measured every 8 for 48 h. Ramsay score for sedation was recorded 8 h (Table 1).
The following complications (nausea, pruritis, hallucination, dreams, blurred vision, oral secretions, and desaturation<90%) were reported.
| Table 1: | Modified Ramsay scale for sedation |
 | |
At the end of the study (48 h after initiation of PCA), patients were asked if they were satisfied or not satisfied with regard to the analgesic strategy.
Patients, nurses in charge of postoperative care who monitored the patients, performed analgesia and collected data were blinded to the PCA regimen used.
Statistics: Statistical analysis was performed with Excel and SPSS® statistical package. Data are expressed as mean (SD). Analyses were performed with Mann Whitney U-test for (quantitative variables) and Pearson`s χ2 test for (qualitative variables).
A two way repeated measures Analysis of Variance (ANOVA) followed by a Tukey`s post hoc test were used to evaluate the effect of time and PCA mixture on morphine consumption.
Forty patients were required in each group to detect a 25% difference in the amount of morphine consumption at the 0.05 level of significance with a power of 0.9. A-value < 0.05 was considered significant.
RESULTS
There were no significant difference across the groups with respect to demographic and perioperative data (Table 2).
Adding ketamine to morphine in PCA device resulted in a significant reduction in cumulative morphine consumption as early as 8 h after initiation of the PCA till the end of the study (Fig. 1).
The mean morphine consumption at 48 h was 84 (9) mg in morphine group vs. 66 (6) mg in morphine ketamine group (p<0.05).
This reduction was associated with similar pain scores at rest and at mobilization (Fig. 2, 3).
| Table 2: | Demographic characteristics and perioperative data. Values are expressed as mean±SD |
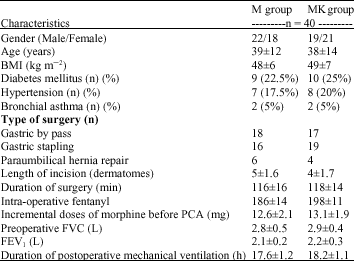 | |
| M = Morphine, MK = Morphine ketamine, BMI = Body mass index, VFC = Forced vital capacity, FEV1 = Forced expiratory volume in first second | |
| Table 3: | Mean arterial pressure, heart rate, respiratory rate and pulse oximetry before and after PCA |
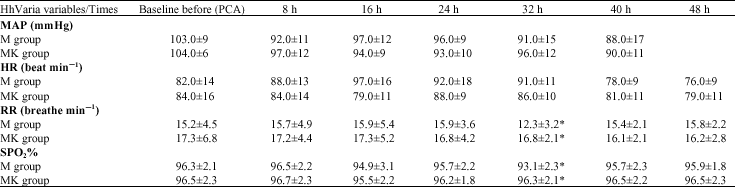 | |
| MAP = Mean arterial pressure, HR = heart rate, RR = respiratory rate, PCA = patient controlled analgesia, * (p<0.05) between groups, Data are mean±SD | |
| Table 4: | Arterial blood gas analysis (patients breathing room air) |
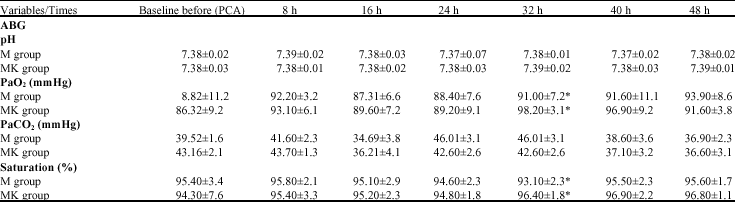 | |
| *(p<0.05) between groups, Data are mean±SD | |
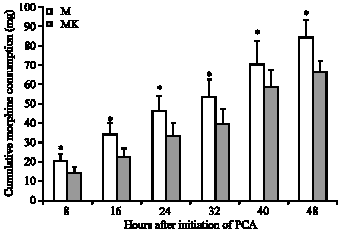 | |
| Fig. 1: | Cumulative morphine consumption, PCA = Patient Controlled Analgesia, *(p<0.05) |
The need for rescue analgesia was more frequent in morphine group (10 patients: 25%) compared with morphine ketamine group (only 2 patients: 5%) (p< 0.05). Haemodynamic data, respiratory rate and pulse oximetry are shown in Table 3. These variables were comparable in the two groups except that respiratory rate was significantly lower in the morphine group 32 h after initiation of PCA :12.3 (3.2) vs. 16.8 (2.1) (p<0.05).
Also SPO2 showed significantly lower values in the morphine group: 93.1 (2.3) vs. 96.3 (2.1) (p< 0.05). Arterial blood gases are shown in Table 4. Values were comparable in both groups except that PaO2 was significantly lower in morphine group 32 h after initiation of PCA: 91 (7.2) vs. 98.2 (3.1) (p<0.05).
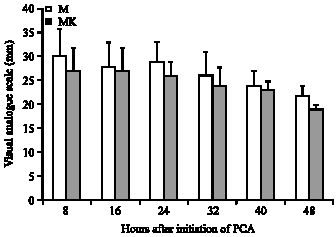 | |
| Fig. 2: | Visual analogue scale score at rest during the use of PCA, PCA = Patient Controlled Analgesia |
Comparing preoperative values, FVC and FEV1 showed significant reduction in both groups. Furthermore, they showed a significant difference between groups with a greater decrease in the morphine group 24 h and 48 h after initiation of PCA (Fig. 4, 5).
FVC at 24 h was 46% (8) in morphine group vs. 62% (9) in morphine ketamine group (p<0.05). At 48 h it was 58% (7) vs. 77% (11) (p<0.05).
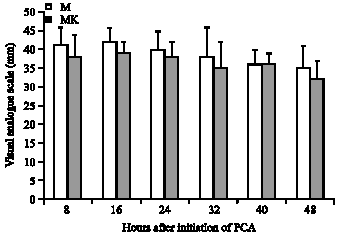 | |
| Fig. 3: | Visual analogue scale score at mobilization during the use of PCA, PCA = Patient Controlled Analgesia |
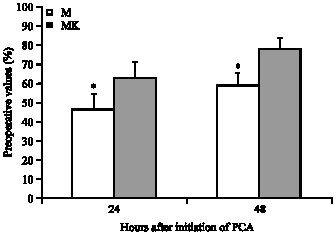 | |
| Fig. 4: | FVC in both groups expressed as a percentage of the preoperative values. Data are mean (SD), PCA = Patient controlled analgesia, *(p<0.05) compared with the preoperative value |
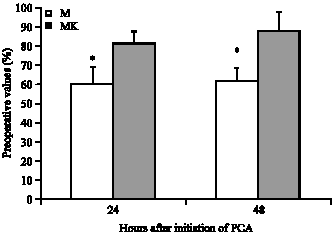 | |
| Fig. 5: | FEV1 in both groups expressed as a percentage of the preoperative values, Data are mean (SD), PCA = Patient controlled analgesia, *(p<0.05) compared with the preoperative value |
| Table 5: | Incidence of complications in the two groups |
 | |
| * p<0.05 | |
FEV1 was 60% (9)vs. 81% (7) at 24 h (p<0.05). At 48 h it was 62% (6) vs. 87% (11)(p<0.05).
Ramsay score showed no difference between the two groups and was maintained between 2 and 3 during the study.
The incidence of side effects was comparable in the two groups expect for nausea and desaturation (Table 5). Incidence of nausea was significantly higher in morphine group than morphine ketamine group (8 vs 2) (p<0.05). Episodes of desaturation (<90%) were significantly more frequent in morphine group (12 vs 5) (p<0.05). Incidence of other side effects (puritis, hallucinations, dreams, blurred vision and oral secretion) were comparable in the two groups.
Regarding patient satisfaction, it was significantly higher in morphine ketamine group than in morphine group (87.5 vs 32.5%) (p<0.05).
DISCUSSION
This prospective double blinded randomized study confirmed the primary hypothesis that adding small doses of ketamine to morphine for PCA allowed a significant reduction in cumulative morphine consumption associated with better post operative analgesia respiratory functions and patients satisfaction.
Several studies have reported a benefit of ketamine use with no difference for cognitive adverse events and lower cumulative analgesic doses for quite better analgesic control (Guillou et al., 2003; Michelet et al., 2007; Adriaenssens et al., 1999). Many factors may account for these beneficial effects. Morbidly obese patients undergoing upper abdominal surgery admitted to SICU experience prolonged noxious stimuli caused by the inflammation reaction of damaged tissues and also created by the monitoring environment, therapeutic devices and nursing care (Guillou et al., 2003). In this context, central sensitization to pain may take place despite the use of adequate doses of opioids.
In addition to inhibition of sensitization in nociceptive pathways, prevention of opiate mediated activation of pronociceptive systems and opiate tolerance may be another mechanism of pain prevention by ketamine (Himmelseher and Durieux, 2005). The development of rapid tolerance and delayed hyperalgesia after intraoperative and postoperative use of different opioids has been reported in surgical patients (Chia et al., 1999; Guignard et al., 2000; Weinbroum, 2003).
In this study, ketamine has been used only for the postoperative period without pre or intra-operative administration. Although there is growing evidence that adjunction of ketamine to general anesthesia with a preemptive effect is beneficial in a variety of surgical procedures (Himmelseher and Durieux, 2005; Bell et al., 2005). This study focused on the postoperative analgesic effect to allow discrimination between a sparing effect on perioperative and postoperative opioid consumption with a specific assessment of influence of ketamine on use of PCA device.
The protocol of the present study included a small dose of ketamine. This choice was supported by the comparison between the studies using ketamine in PCA devices, which indicated that there was no increased morphine sparing effect by increasing ketamine dose to 25-30 mg day-1 (Bell et al., 2005).
As recently highlighted in reviews and studies focusing on selected patients who traditionally require large doses of opioids, the adjunction of morphine in this specific population allowed a decrease in post-operative morphine consumption (Guillou et al., 2003; Ptrenko et al., 2003; Michelet et al., 2007).
However, conflicting results still exist concerning the potential benefit of adding ketamine to morphine for PAC. Indeed, whereas many studies agree with our result (Guillou et al., 2003; Adriaenssens et al., 1999; Lahtinen et al., 2004; Javery et al., 1996). others have failed to confirm them (Reeves et al., 2001).
These discripancies could be explained by differences in methodologies between studies. The lack of sparing effect and adverse cognitive effects reported by Reeves and colleagues could be related to the absence of rescue analgesia and the consecutive higher doses of morphine and ketamine required.
Morbidly obese patients are potentially suffering from restrictive pulmonary dysfunction. Upper abdominal surgery adds to their suffering (Postlethwait and Johnson, 1972). So, it was highly indicated to search for an analgesic regimen that relief their pain without further encroachment on their pulmonary functions.
Although arterial blood gases analysis revealed only limited variation between the study groups, spirometric data confirmed the hypothesis of opioid dependant depression of respiratory function.
This was evident by significantly more reduction in FVC and FEV1 in morphine group 24 and 48 h after PCA when compared with pre-operative values.
This reduction was less marked in morphine ketamine group which expressed significantly higher values than morphine group.
Previous study by Michelet et al. (2007) supported present results. They demonstrated significantly better spirometric data in patients undergoing thoracotomy receiving ketamine with morphine PCA than those receiving morphine PCA.
Previous studies reported that even more powerful analgesic techniques such as epidural analgesia did not result in improved respiratory mechanics (Boissen et al., 2001).
Since high doses of ketamine could be responsible for ventilatory depressive effect (Mildh et al., 1998), it seems that the beneficial effect of ketamine on respiratory function is not related mainly to the influence on respiratory mechanics but it could be due to opioid sparing effect. An objective of this study was to determine whether ketamine could reduce the side effects of opioids particularly nausea.
This study demonstrated significantly lower incidence of nausea. This finding is supported by previous reports which reported similar effects (Bell et al., 2005).
Being administered in small doses, ketamine infusion was not associated with psychomimetic effects or cognitive impairment.
CONCLUSIONS
Adding small doses of ketamine to morphine in PCA devices appears to be an alternative strategy for limitation of postoperative respiratory disorders and for pain control when an intravenous analgesia is scheduled after upper abdominal surgery in morbidly obese patients. Future clinical trials should focus on high risk groups for opioid resistant acute postoperative pain e.g. chronic therapeutic opioid intake, substance abusers and amputations.
ACKNOWLEDGMENTS
I wish to thank all my colleagues, technicians and nurses in the SICU for their great help and cooperation during performing this study.
REFERENCES
- Adriaenssens, G., K. Vermeyen, V. Hoffmann, E. Mertens and H. Adriaensen, 1999. Postoperative analgesia with I.V. patient controlled morphine effect of adding ketamine. Br. J. Anaesth., 83: 393-396.
CrossRefDirect Link - Ballantyne, J.V., D.B. Carr and S. De Ferranti et al., 1998. The comparative effects of postoperative analgesic therapies on pulmonary outcome: Cumulative meta-analysis of randomized controlled trials. Analgesia, 86: 598-612.
PubMedDirect Link - Bell, R., J. Dahl, R. Moore and E. Kalso, 2005. Perioperative ketamine for acute post operative pain: A quantitative and qualitative systematic review. Acta Anaesthesiol. Scand., 49: 1405-1428.
CrossRefDirect Link - Boissen, N., O. Rabary and B. Padovani et al., 2001. Improvement of dynamic analgesia does not decrease atelectasis after thoracotomy. Br. J. Anaesth., 87: 564-569.
CrossRefPubMedDirect Link - Cairns, B.E., P. Svenson, K. Wang, S. Hupfeld, T. Graven-Nielsen, D. Sessler, C.B. Berde and L. Arendt Nielsen, 2003. Activation of peripheral NMDA receptors contributes to human pain and rat afferent discharge evoked by injection of glutamate into the massoger muscle. J. Neurophysiol., 90: 2098-2105.
CrossRefPubMedDirect Link - Catley, D., C. Thornton, C. Jordan, J. Lehane, D. Royston and J. Jones, 1985. Pronounced episodic oxygen desaturation in the postoperative period: Its association with ventilatory pattern analgesic regimen. Anesthesiology, 63: 20-28.
Direct Link - Chia, Y.T., K. Liu, J.J. Wang, M.C. Kuo and S.T. Ho, 1999. Intro-operative high dose fentanyl induces postoperative fentanyl tolerance. Can. J. Anaesth., 46: 872-877.
PubMedDirect Link - Derzie, A.J., F. Silvestri, E. Liriano and P. Beneotti, 2000. Wound closure technique and acute wound complications in gastric surgery for morbird obesity a prospective randomized thrial. J. Am. Coll. Surg., 191: 238-243.
Direct Link - Guignard, B., A.E. Bossard, C. Coste, D.I. Sessler, C. Lebrault, P.Alfonsi, P. Fletcher and M. Chauvin, 2000. Acute opioid tolerance: Intraoperative remifentanyl increases postoperative pain and morphine requirement. Anesthesiology, 93: 409-414.
Direct Link - Guillou, N., M. Tanguy, P. Seguim, B. Beranger, J. Compion and Y. Malledant, 2003. The effect of small dose ketamine on morphine consumption in surgical intensive case unit patients after major abdominal surgery. Anesth. Analg., 97: 843-847.
PubMedDirect Link - Himmelseher, S. and M. Durieux, 2005. Ketamine for perioperative pain management. Anaesthesiology, 102: 211-220.
Direct Link - Javery, K.B., T.W. Ussery, H.G. Steger and G.W. Colclough, 1996. Comparison of morphine and morphine with ketamine for postoperative analgesia. Can. J. Anaesth., 34: 212-215.
CrossRefPubMedDirect Link - Lahtinen, P., H. Kokki, T. Hakala and M.S. Hynynen, 2004. (+) Ketamine as an analgesic adjunct reduces opioid consumption after cardiac surgery. (+) Ketamine as an analgesic adjunct reduces opioid consumption after cardiac surgery. Anesth. Analg., 99: 1295-1301.
PubMedDirect Link - Michelet, P., C. Guervilly, A. Helaine, J.P. Varo, D. Playac, F. Gaillat and T. Dantin, 2007. Adding ketamine to morphine for patient controlled analgesia after thoracic surgery: Influence on morphine consumption, respiratory function, and nocturnal desaturation. Br. J. Anaesth., 99: 396-403.
CrossRefPubMedDirect Link - Mildh, L., M. Taittonen, K. Leino and O. Kirvela, 1998. The effect of low dose ketamine on fentanyl induced respiratory depression. Anaesthesia, 53: 965-970.
CrossRefDirect Link - Petrenko, A.B. T. Yamakura, H. Baba and K. Shimoji, 2003. The role of N-methyl D-aspartate receptors in pain: A review. Anaesth Analg., 97: 1108-1116.
PubMedDirect Link - Postlethwait, R.W. and W.D. Johnson, 1972. Complications following surgery for duodenal ulcer in obese patients. Arch. Surg., 105: 438-440.
Direct Link - Reeves, M., D. Lindholm, P. Myles, H. Fletcher and J. Hunt, 2001. Adding ketamine to morphine for patient controlled analgesia after major abdominal surgery: A double blinded randomized controlled trial. Anaesth. Analg., 93: 116-120.
PubMedDirect Link - Roshanak, C., B. Steven, C. Nicolas and R. Falrice, 2003. Patient controlled iv analgesia is an acceptable pain management strategy in morbidly obese patients undergoing gastric bypass surgery. A retrospective comparison with epidural analgesia. Can. J. Anaesth., 50: 672-678.
PubMedDirect Link - Schmid, R., G. Koren, J. Klin and J. Katz, 2002. 2002The stability of a ketamine morphine solution. Anaesth. Analg., 94: 898-900.
PubMedDirect Link - Stone, J., K. Cozine and A. Wald, 1999. Nocturnal oxygenation during patient controlled analgesia. Anaesth. Analg., 89: 104-110.
PubMedDirect Link - Sveticic, G., A. Gentilini, U. Eichenberger, M. Luginbuhl and M. Curatolo, 2003. Combination of morphine with ketamine for patient controlled analgesia. Anaesthesiology, 98: 1195-1205.
Direct Link - Weinbroum, A.A., 2003. A single small dose of postoperative ketamine provides rapid and sustained improvement in morphine analgesia in presence of morphine-resistant pain. Anaesth Analg., 96: 789-795.
PubMedDirect Link








