Research Article
A Review on Crimean-Congo Haemorrhagic Fever in Asia
Department of Medical Microbiology, Health Research Center, Baqiyatallah (a. s) University of Medical Sciences, Tehran, Islamic Republic of Iran
The disease was first described in the crimea in 1994 and given the name Crimean haemorrhagic fever (Whitehouse, 2004). In 1969 it was recognized that the pathogen causing Crimean haemorrhagic fever was the same as that responsible for an illness identified in 1956 in the Congo and linkage of the 2 place names resulted in the current name for the disease and the virus. The disease is endemic in some countries including South Africa, Congo, Pakistan, Iran and former Yugoslavia (Chinikar et al., 2004) and in fact the disease was reported at least from three continent including Africa, Europe and Asia (Kuljic-Kapulica, 2004; Lvov et al., 2004) and very recently some epidemic form of the diseases were occurred in Kosovo (Kuljic-Kapulica, 2004; Lvov et al., 2004), Iran (Izadi et al., 2004; Chinikar et al., 2004), Pakistan (Jamil et al., 2005), Turkey (Karti et al., 2004; Bakir et al., 2005). South Africa (Fisher-Hoch et al.,1992) and Mauritania (Nabeth et al., 2004) Russia (Lvov et al., 2004) and United Arab Emarat (Chinikar et al., 2004 ). In addition a few nosocomial infections were reported in few countries too (Mehrabi Tavana et al., 2002). This study provides the latest review of Crimean Congo Heamorraghic fever and it’s situation in Asia for preventing the infection.
Epidemiology: CCHF is a severe disease in humans, with a high mortality rate. Fortunately, human illness occurs infrequently, although animal infection may be more common. The geographical distribution of the virus, like that of its tick vector, is widespread (Fig. 1).
CCHF reservoirs: The CCHF virus may infect a wide range of domestic and wild Animals. It has to be said many birds are resistant to the infection however Ostriches are susceptible (Chinikar, 2003). Animal become infected by the bite of infected Ticks (Mehrabi Tavana et al., 2002). A number of tick genera are capable of becoming infected with CCHF virus, but the most efficient and common vectors for CCHF appear to be members of the Hyalomma genus (Flick and Whitehouse, 2005 ).
Trans-ovarial (transmission of the virus from infected female ticks to offspring via eggs) and venereal transmission have been demonstrated amongst some vector species, indicating one mechanism which may contribute to maintaining the circulation of the virus in nature (Mehrabi Tavana et al., 2002 ). However, the most important source for acquisition of the virus by ticks is believed to be infected small vertebrates on which immature Hyalomma ticks feed. Once infected, the tick remains infected through its developmental stages and the mature tick may transmit the infection to large vertebrates, such as livestock (Linthicum and Bailey, 1994). Domestic ruminant animals, such as cattle, sheep and goats, are viraemic (virus circulating in the bloodstream) for around one week after becoming infected (Chinikar, 2003). Humans also who become infected with CCHF acquire the virus from direct contact with blood or other infected tissues from livestock during this time, or they may become infected from a tick bite. Possibly the majority of cases have occurred in those involved with the livestock industry, such as agricultural workers, slaughterhouse workers, veterinarians and medical staff who are caring the patients (Mehrabi Tavana et al., 2002).
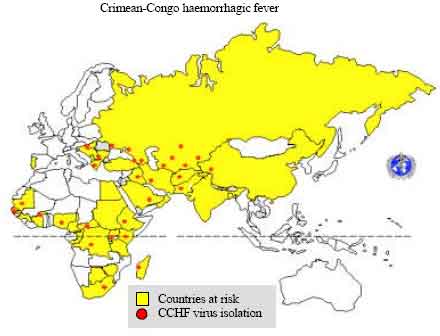 | |
| Fig. 1: | Geographic distribution of Crimean-Congo haemorrhagic fever |
The clinical features: The incubation period following contact with infected blood or tissues is usually five to six days (depends the mode of transmission in fact infection via blood or tissue quicker), with a documented maximum of 13 days. Following infection via tick bite, the incubation period is usually one to three days, with a maximum of nine days. Onset of symptoms is sudden, with fever, myalgia (aching muscles), dizziness, neck pain and stiffness, backache, headache, sore eyes and photophobia (sensitivity to light) (Saijo, 2005 ). The case fatality rate of CCHF ranges from 10-40% (Saijo et al., 2004). Diagnosis: Diagnosis of suspected CCHF is performed in specially-equipped, high biosafety level laboratories (biosafety 2 or 3). IgG and IgM antibodies may be detected in serum by enzyme-linked immunoassay (the ELISA or EIA methods) from about day six of illness. IgM remains detectable for up to four months and IgG levels decline but remain detectable for up to five years. Patients with fatal disease do not usually develop a measurable antibody response and in these individuals, as well as in patients in the first few days of illness, diagnosis is achieved by virus detection in blood or tissue samples. There are several methods for doing this. The virus may be isolated from blood or tissue specimens in the first five days of illness and grown in cell culture. Viral antigens may sometimes be shown in tissue samples using immunofluorescence or EIA. More recently, the Polymerase Chain Reaction (PCR), a molecular method for detecting the viral genome, has been successfully applied in diagnosis (Duh et al., 2006; Ozkurt et al., 2006; Chinikar et al., 2004).
Treatment: General supportive therapy accompanied with the antiviral drug ribavirin has been used in treatment of established CCHF infection. Both oral and intravenous formulations seem to be effective (Mardani et al., 2003).
Control: No vaccine available for human use at present time to prevent the infection, mostly the consideration health measure are very effective to prevent the disease. Although, the tick vectors are numerous and widespread and tick control with acaricides (chemicals intended to kill ticks) is only a realistic option for well-managed livestock production facilities. In addition, the persons who are living in endemic areas should use personal protective measures that include avoidance of areas where tick vectors are abundant and when they are active (Spring to Fall); regular examination of clothing and skin for ticks and their removal and use of repellents is necessary. It should not be forgotten that, persons who work with livestock or other animals in the endemic areas can take practical measures to protect themselves. These include the use of repellents on the skin (e.g., DEET) and clothing (e.g., permethrin) and wearing gloves or other protective clothing to prevent skin contact with infected tissue or blood. High level of take care needed when patients with CCHF are admitted to hospital, there is a risk of nosocomial spread of infection. In the past, serious outbreaks have occurred in this way and it is imperative that adequate infection control measures be observed to prevent this disastrous outcome. Patients with suspected or confirmed CCHF also should be isolated and cared for using barrier nursing techniques. Specimens of blood or tissues taken for diagnostic purposes should be collected and handled using universal precautions. Sharps (needles and other penetrating surgical instruments) and body wastes should be safely disposed of using appropriate decontamination procedures. Health care workers are at risk (Mehrabi Tavana et al., 2002; Chinikar, 2003; Chinikar et al., 2004) they must be considered a high level of care and precaution.
CCHF infection is reported from many countries, in particular in the Persian Gulf region (El-Azazy and Scrimgeour, 1997; Rodriguez et al., 1997; Schwarz et al., 1995; Scrimgeour et al., 1996; Suleiman et al., 1980; Burt et al., 1994; Hassanein et al., 1997; Hoogstraal et al., 1981; Khan et al., 1997). But also from countries on various continents (Hoogstraal, 1979; Duh et al., 2006; Ergonul, 2006). The characteristics of CCHF virus infection in domestic animals are often unapparent, while human infections often result in severe hemorrhagic fevers (Mehrabi Tavana et al., 2002). The incubation period for CCHF virus infection in humans is commonly 1 week, but varies depending on the type of exposure (tick bite, contact with blood, or skin of infected animals). Viruses are only detectable during the first week of illness so analysis of the humoral response is the best mode of detection for the disease (Chinikar, 2003; Chinikar et al., 2004). CCHF in a few cases were seen as nosocominal infection (Mehrabi Tavana et al., 2002). In addition, among different modes of transmission (Jamil et al., 2005; Lvov et al., 2004; Mehrabu Tavana et al., 2002), the made of CCHF virus transmission in Iran is not completely clear, but it seems handling the infected organs of slaughtered livestock may be the main way of contamination in different countries which mentioned above. Highly precaution is needed to prevent the infection and reduce mortality and morbidity as well as economical damages. This situation is very similar in many infected countries.