Research Article
Thermotolerant Coliform and Escherichia coli Detection and Enumeration Through Multiple Tube Fermentation
Water Pollution Research, National Research Center, Asian Network for Scientific Information Dokki, Cairo, Egypt
Faecal contamination in water sources must be identified in order to adequately address water criteria problems to protect public health. Water quality indicators levels are critical parameters that drive management decision (Carson et al., 2001; Mclellan et al., 2003). Currently, coliforms and E. coli are of great importance among bacterial indicators used in water quality definition and health risk assessments. (Ho and Tam, 1997; Baudisova, 1997; Alonso et al., 1999; WHO, 2001, 2004; Giannoulis et al., 2005).
Escherichia coli is considered as a more specific indicator of faecal pollution than other faecal colifoms (Borrego and Figueras, 1997; Leclerc et al., 2000 and Sueiro et al., 2001).While faecal coliforms have been found in ambient waters in the absence of apparent faecal pollution though, may establish viable populations especially when high levels of carbohydrates are available as nutrient sources (Toranzos and Mcfeters, 1997; Mclellan et al., 2001), the Escherichia coli could not multiply outside warm blooded animals (Leclerc et al., 2000; Meays et al., 2004). In addition, Byamukama et al. (2000) attributed the discriminatory power of E. coli as an indicator organism is done to its weak ability to replicate in the natural environment. Moreover, Baudisova (1997) supports the use of E. coli assessment as main indicator of faecal pollution than many other biological indicators. Although E. coli is EPA recommended indicator of faecal pollution, Faecal Coliforms (FC) continue to be widely accepted for monitoring of recreational waters according to data reported by U.S EPA (1986, 2000). Determination of faecal coliforms and E. coli using Most-Probable Number (MPN) by multiple tube technique are based on lactose fermentation within 24 h at 44.5±0.2°C.
Furthermore, E. coli is detected by the same method but often by using different media formulations and a test for indole production in the same multiple-tube. The aim of the present study is to clear the efficiency of last mentioned statement for accurate detection of faecal coliforms and E. coli by a simple, rapid and economic technique with supporting acid and gas production at 44.5°C by the addition of mannitol sugar.
MATERIAL AND METHODS
Comparison between standard and modified media for faecal coliforms and Escherichia coli detection: Enumeration of Faecal Coliform (FC) and Escherichia coli by most probable number (MPN) technique were carried out using two procedures according to APHA (1998).
| • | The first procedure was applied using four standard media : m-7hfc broth (polypeptone, 5 g; yeast extract, 3 g; lactose, 10 g; d-mannitol, 5 g; sodium chloride, 7.5 g; sodium deoxycholate, 0.1 g; Bromocresol purple, 0.35 g; phenol red, 0.3 g 1 L Distilled water (D.W)), A-1 broth (lactose, 5 g; Tryptone, 20 g; Sodium chloride, 5 g; Triton X-100, 1 mL 1 L D.W), EC broth (Tryptone, 20 g; Lactose, 5 g; Bile salt No.3, 1.5 g; K2HPO4, 4 g; KH2PO4, 1.5 g; Sodium chloride, 5 g/1 L D.W) and MacConky broth (Peptone, 20 g; Lactose, 10 g; Sodium chloride, 5 g; Bile salt, 5 g; Bromocresol purple, 0.01 g/1 L D.W). |
| • | The second procedure was applied using the previously maintained standard media with some additives or reduction in the same compounds of their contents. These media were modified by adding 1% tryptophane, 0.1% d-mannitol and the quantity of lactose halved in three of the mentioned media (EC, A-1 and MacConky media). The fourth medium (m-7h-fc) was modified by adding 1% tryptophane and reducing the quantity of lactose to half while it contains d-mannitol originally. |
These comparisons between standard and modified for FC and E. coli detection was carried out via testing 12 different samples represent (4, sewage; 4, Nile water and 4, ground water). Faecal coliform-MPN was estimated according to APHA1998. All inoculated tubes representing different media and different samples sources ware incubated at 44.5°C in a circulating water bath for 24 h while incubation period was reduced for 7 h in case of m-7hfc according to APHA (1998). Acid and gas production in incubated tubes were recorded as a positive test for FC in both standard and modified media.
For E. coli enumeration using the modified media, in the same tubes which showing gas production of the faecal coliform (positive test), a few drops of Kovac's reagent was added to each faecal coliform positive tube, any shade of pink in the reaction layer of the indol test are considered as a positive results for E. coli presence test. On the other hand, enumeration of E. coli with the standard media, one inoculum from each all positive tubes (FC positive test) was inoculated into a tryptophan broth 1% (TW).
These inoculated tubes were incubated at 44.5°C for 18 h at least to detect indol production. A few drops of Kovac's reagent was added to each tube of (TW). Development of any shade of pink in the reaction layer of the indol test, are considered a positive E. coli test (Smith and Rockliff, 1982).
MPN of FC or E. coli in each test was estimated from MPN tables (APHA, 1998).
Application of Modified (M) media (M-m7h fc, M-A-1, M-MacConky and M-EC media): Water samples (56) were collected from different environmental sources as ground water (10, Nile water (16), Sewage (10), treated sewage (septic tank effluent) (10) and sludge (10) the collected samples were analyzed within 4-6 h from their collection for detecting and enumerating FC and E. coli using modified media as described previously.
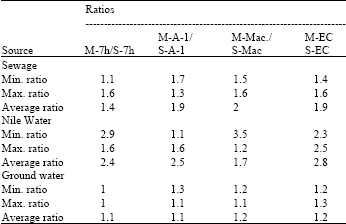
Water sources representing natural water bearing different loads of contaminants as inorganic and organic matter whether it is dead or viable from different sources. The most important marker in our concept is the organic dead or viable load. Table 1 and 2 show results of MPN-ratios between Modified (M) and standard (S) media (M/S media) for faecal coliforms and E. coli in different aquatic environments for raw MPN data represented by Minimum, Maximum and average values (Table 1 and 2).
From these results, it can be observed that recoveries of faecal coliforms and E. coli mostly in all cases using all modified media were more over values of that detected by using standard media i.e., showing more higher MPN values.
MPN-ratio for faecal coliforms was more than one time in all different sources of water samples (sewage, Nile or ground waters). Highest average MPN-ratios were 2.8 with M-EC in Nile water, 2 with M-MacConky broth in sewage and 1.2 with each M-MacConky or M-EC in groundwater for faecal coliforms. On the other hand, the lowest average MPN-ratios were 1.7 with M-MacConky broth in Nile water 1.4 with M-m-7hfc broth in sewage and 1.1 with M-A-1 broth or M-m-7h fc broth in ground water for faecal coliforms. In sewage, MPN-ratios for FC ranged form 1.1 to 1.6, 1.7 to 1.3, 1.5 to 1.6 and from 1.4 to 1.6 with M-m-7h fc broth, M-A-1, M-MacConky and M-EC, respectively. In Nile water, MPN-ratios of FC ranged from 2.9 to 1.6, 1.1 to 1.6, 1.2 to 3.5 and 2.3 to 2.5 with M-m-7hfc, M-A-1, M-MacConky and M-EC media, respectively. MPN-ratios using M-m-7hfc showed no differences between modified and standard media in groundwater. However other media MPN-ratios ranged from 1.3 to 1.1, 1.2 to 1.1 and 1.2 to 1.3 with M-A1, M-MacConky and M-EC media, respectively for faecal coliforms.
According to results in Table 2 it can observed that MPN-ratio for E. coli with all modified media was 1.1 times greater than with standard media. The average MPN-ratios of E. coli were 5 with M-A-1 or 5.1 with other modified media in sewage, 1.3 with M-MacConky and 1.2 with other modified media in groundwater (Table 2).
| Table 1: | MPN-ratios using modified and standard media for fecal coliform enumeration |
 | |
| Table 2: | MPN-ratios using modified and standard media for Escherichia coli enumeration |
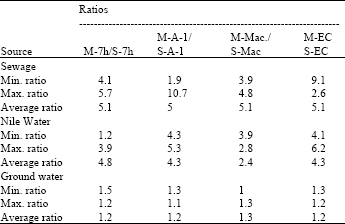 | |
In Nile water, highest average MPN-ratio was 4.8 with M-m-7hfc.
MPN-ratios for E. coli ranged from 4.1 to 5.7, 1.9 to 10.7, 3.9 to 4.8 and 9.1 to 2.6 in sewage, while reaching 1.2 to 3.9, 4.3 to 5.3, 3.9 to 2.8 and 4.1 to 6.2 in Nile water. Groundwater showing ratio ranged from 1.5 to 1.2, 1.3 to 1.1, 1 to 1.3 and 1.3 to 1.2 with M-m-7hfc, M-A-1, M-MacConky and M-EC media, respectively (Table 2). Generally the modified media gave higher recoveries of FC and E. coli than standard media in different aquatic environment bearing lower or higher numbers of any or both of the two used parameters (Table 1 and 2).
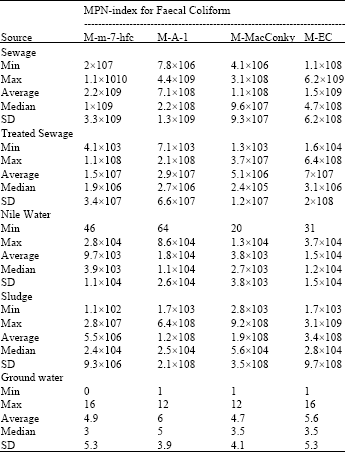
From Table 3 it can be noted that the highest average recoveries of (FC) were 109 using M-m-7hfc or M-EC media in sewage, 104 using M-A-1 or MEC in Nile water and 6 using M-A-1 in ground water. In addition to that, there is no difference with the average densities of FC in treated sewage 107 (MPN/100 mL) and sludge 108 (MPN 1 /g) using all modified media except M-MacConky broth 106 in treated sewage and M-m7hfc 106 in sludge samples.
| Table 3: | comparison of FC density obtained by MPN procedure using different media |
 | |
| SD: Standard Deviation | |
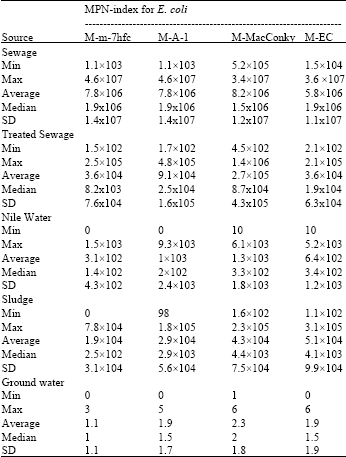
Results in Table 4 showed that the densities of E. coli were 106 and 104 in sewage and sludge samples with all modified media. Furthermore the densities of E. coli were 104 with all modified Media except M-MacConky broth 105 in treated sewage 103 with M-A-1 or M-MacConky and 102 with M-m-7hfc or M-EC media in Nile water. Highest average recovery of E. coli in ground water was 2.3 using M-MacConky broth. Generally highest recoveries of E. coli were observed with M-MacConky broth from different aquatic environments (Table 4). The best overall performance of E. coli detection was obtained using M-MacConky or M-A-1 media.
Water quality assessment is depending upon the detection of the most specific, sensitive, rapid and easily enumerated bacterial indicators in the tested or examined water source. Baudisova (1997) and WHO guidelines (2001, 2004) recommended the use of E. coli as the main indicator for detection of faecal pollution. Thus the evaluation of the specificity and sensitivity of a new detection method for E. coli which is highly recommended.
| Table 4: | Comparison of E. coli density obtained by MPN procedure using different media |
 | |
| Note: All sources (MPN index/100 mL except sludge (MPN index/1 g) | |
Escherichia coli assessment bring advantages when compared with coliform group because it survive in water for a shorter period than other coliforms including faecal coliforms. Significant differences appear especially in summer when elevated water temperature allows development of non-faecal bacteria (Baudisova, 1997).
Various modified media have been described in which both gas and indole formation can be demonstrated in a single tube at the same step. There is also a close relation between the fermentation of lactose and mannitol by E. coli, indeed occasional some of non lactose fermenting strains of E. coli are known to ferment mannitol (Kasweck and Filiermans, 1978; Wright, 1982).
Lactose fermentation i.e. acid and gas production at elevated temperature served to differentiate faecal from non faecal coliforms, as far as it will known that, E. coli produces acid and gas from lactose fermentation at temperature (44-46°C) (Dockins and Mcfeters , 1978; El-Abagy et al., 1980; Ashbolt et al., 2001; Mclellan et al., 2001; Rompre et al., 2002; WHO, 2004). On the other hand, it was reported that only 90% of E. coli isolates produce acid and gas from lactose under these conditions (Mechman and Romero, 1982). Another distinguishing criteria is their ability to produce indole at elevated temperature (44-46°C). This criteria has been suggested and applied as a specific test for E. coli detection where 95% of E. coli were indole positive within (6-18 h) at 44.5°C (Anderson et al., 1980; Smith and Rockliff, 1982; Humphrey and Gawler, 1986; Wang et al., 2001; WHO, 2001, 2004).
Smith and Rockliff (1982) noted that, at elevated temperature, the use of mannitol in place of lactose leads to false positive results (acid and gas formation) while indole production is reduced in the absence of lactose. They managed to use tryptophan for seek of indole production reducing mannitol concentration for the production of acid and gas quantity correlated to exact E. coli density.
During the present work four of traditional MPN methods using four of the commonly used standard media in the field of the environmental microbiology specially water sanitation. These four media were m-7h-fc broth, A-1, MacConky and EC media. The proposed modification during the present application is based on the use of lactose and mannitol 1 g as a fermentable sugar and 10 g tryptophane and other content of each corresponding standard media/1 L. In case of M-m-7hfc broth, it contains 5 g lactose, 5 g mannitol and 10 g tryptophane and other content. These modifications were applied for the purpose of acid and gas production, indole formation and withstanding at 44.5°C for the present organisms).
Observed results, showed that MPN-ratio between modified and standard methods media were higher than one time for both FC and E. coli. Thus MPN-technique using different modified media for FC and E. coli enumeration was evaluated during this work. Comparison between obtained results based on determination of upper and lower counting limit as well as recovery comparability between modified media seems to offer advantage for detection and enumeration of FC and E. coli in the same tube of MPN-technique at the same time of enumeration. It can be safely said that M-EC and M-A-1 media were the best for FC enumeration while M-MacConky and M-A1 were the best for E. coli detection.
From this study, the final conclusion is that this new technique is more suitable for detection and enumeration of FC and E. coli at the same time under the same conditions. Generally, this method needs further studies for developing the mentioned technique application in the field of environmental quality control and quality assurance.