Research Article
Biochemical Studies on Rabbit Treated with Di-2-Pyridylglyoxal
Department of Biochemistry, Faculty of Science, Alexandria University, Alexandria, Egypt
Glyoxal and some of its derivatives have a wide range of biological effects. For instance, glyoxal reacts with single-strand DNA and permit blocking of essentially all guanine residues (Birnboim and Mitchel, 1978). Also, glyoxal was used as a probe for micro determination of the structure of unidentified nucleic acid components (Shapiro et al., 1986). It may induce lung epithelial cell apoptosis (Kasper et al., 2000; Roehlecke et al., 2000).
Likewise, methylglyoxal was found to promote phosphorylation of certain kinases and induced degradation of certain polymerases (Akhand et al., 2001). Also, methylglyoxal inhibits the growth of E.coli that accompanied by the inhibition of protein synthesis and initiation of replication of DNA (Farval and McBrien, 1980).
Meanwhile, glyoxalbis (guanyl hydrazone) which was used as antileukemic drug and its analog ethylmethylglyoxalbis (guanyl hydrazone), were found to be competitive inhibitors of adenosyl methionine decarboxylase, diamine oxidase and ornithine decarboxylase (Seppanen et al., 1984). On the other hand, phenylglyoxal was used to determine the role of arginyl residues in the alpha subunit of tryptophane synthase (Eun and Miles, 1984).
Therefore, in this study we focus on the in vivo effect of di-2-pyridylglyoxal upon the biochemical changes that may appear in rabbit with respect to liver alcohol dehydrogenase (ADH;Alcohol:NAD oxidoreductase; E.C.1.1.1.1). This enzyme catalyzes the reversible oxidation of primary and secondary alcohols to their corresponding aldehydes.
The present research work is continuation of our previous research (Yacout, 2000; Yacout, 2001; Balbaa et al., 2001).
Materials and Methods
Chemicals: Di-2-pyridylglyoxal (Fig. 1) and orcinol were purchased from Aldrich Chemical Co. Ltd., Gillingham, England. NAD and cholesterol were purchased from Sigma Chemical Co. (St. Louis, MO, USA). Folin-Ciocalteu's phenol reagent and diphenylamine were purchased from Merk, Hohenburnn. Bovine serum albumin was purchased from Fisher Biotech (Fisher Scientific Fair Lawn, N.J., USA). t-RNA from E.coli and DNA sodium salt from calf thymus were purchased from BDH Chemicals Ltd. (Poole, England).
 | |
| Fig. 1: | Di-2-pyridylglyoxal |
Animals: Eight healthy rabbits weighing 800±50 g each were divided into four groups. The first group (N) served as control, received normal diet and corn oil. The second group (G1) received normal diet with daily oral dose of di-2-pyridylglyoxal in corn oil (5 mg ml-1 for four days). The third group (G2) was received 10 mg ml-1 and the fourth group (G3) was received 25 mg ml-1, each for four days. Blood was collected separately, left to clot and each obtained serum was ready for clinical analysis. Incorporation of di-2-pyridylglyoxal using (G2 ) was effective but with an overall low degree of systemic toxicity, compared to (G1) which was ineffective and (G3) which was more toxic. So, liver was removed from group (G2) as well as control (N) then analyzed for alcohol dehydrogenase activity, cholesterol, RNA and DNA content.
Serum clinical analysis: The following serum parameters: aspartate aminotransferase (AST), alanine aminotransferase (ALT), total protein, albumin, bilirubin, urea, creatinine, cholesterol, triglycerides, insulin, glucose, serum creatine kinase, alkaline phosphatase and acid phosphatase were measured in control group as well as in (G1), (G2) and (G3) groups, using kits produced by Boehringer Mannheim, Germany.
Isolation of liver alcohol dehydrogenase: According to the method of Nabeeh and Charles (1967) fresh liver (15 g) from control rabbit and G2-exposed rabbit were ground and homogenized with distilled water at 40C for 10 min using Tekmar tissumizer, each separately. Each homogenate was filtered off; fractionated with solid ammonium sulfate. Centrifugation was carried out at 7000 g for 60 min at 40C using Hettich Zentrifugen EBA 12 R cooling centrifuge. The precipitated enzyme was removed by tris-HCl buffer (0.1 M, pH 8.5) and dialyzed against the same buffer, at 40C for overnight. A sample from each fraction was assayed for alcohol dehydrogenase activity and protein content.
Estimation of protein: The protein content was estimated according to the method as reported by Lowry et al. (1951) using bovine serum albumin as standard.
Enzyme assay: Alcohol dehydrogenase activity was measured spectrophotometrically by recording the rate of production of NADH at 340 nm based on a modified method of Biochemica, Cat. No 15420EAAR by adding 2.6 ml of 0.1 M tris-HCl buffer, pH 8.5 to 0.1 ml of 0.2 M ethanol as substrate, 0.05 ml of 0.1 M NAD in 0.01 HCl and 0.1 ml of semicarbazide-HCl 0.05M. Incubate the above assay mixture at 300C for 3-4 min and then at zero time, 0.1 ml of enzyme preparation was added to start the reaction after making up the volume with water to 3 ml. Determine the rate of absorbency resulting from reduction of NAD and then specific activity of control and (G2) group was calculated according to Jurgen and Marianne (1983).
Estimation of hepatic cholesterol: 2.1 g of liver were removed from G2-treated rabbit and control rabbit each separately and then extracted with chloroform: methanol mixture (2:1,v/v) according to Folch et al. (1957), using soxhlet apparatus for two hours. A sample from each lipid extract (70 mg) was saponified separately with 25 ml alcoholic KOH (0.5 M), for one hour. Each unsaponified layer was evaporated to dryness and then dissolved in 2 ml chloroform and the extracted cholesterol was determined according to Harold (1988).
Estimation of hepatic RNA and DNA: One g of liver from (G2 )-treated rabbit and control rabbit (N) were homogenized in 10 ml of 0.25 M sucrose, separately. Each obtained homogenate was fractionated separately for RNA and DNA as described by Keleti and Lederer (1974), yielding fraction (I) for RNA and fraction (II) for DNA measurements. 0.5 ml of fraction (I) was mixed with 1.5 ml of orcinol reagent in a test tube covered with marble, placed in boiling water bath for 25 min and then cooled. The developed colour was measured at 665 nm against blank, containing 0.5 ml of 0.3 M KOH-0.5 M perchloric acid mixture (3: 5,v/v) and then treated as test sample.
0.9 ml of fraction (II) was mixed with 1.8 ml diphenylamine reagent, in a test tube covered with marble and placed in a boiling water bath for 12 min and then cooled. The developed colour was estimated at 596 nm against blank solution, containing 0.9 ml of 0.5 M perchloric acid and treated as test sample. Standard curves of RNA and DNA were used.
This work was carried out in the Biochemistry Department, Faculty of Science, Alexandria University, Alexandria, Egypt.
The obtained results (Table 1) revealed that AST/ALT ratio of the exposed groups G1 (0.69), G2 (0.36) and G3 (0.70) were found to be near the control (0.74), whereas the A/G ratio was found to be (1.28), (2.21) and (1.49) respectively compared to control (3.08) and urea/creatinine ratio was (101.56), (77.14) and (120.69) respectively compared to control (82.86). Moreover, estimation of serum bilirubin, cholesterol and triglycerides showed a remarkable decrease, compared to control. Estimation of serum insulin and glucose of the examined groups were found to be almost the same with control.
| Table 1: | Serum parameters of (G1), (G2) and (G3)-treated rabbits, compared to control (N) group |
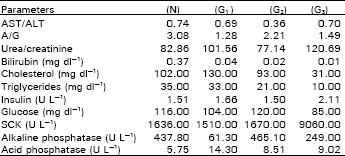 | |
| AST/ALT: Aspartate aminotransferase/alanine aminotransferase | |
| A/G: Albumin/globulin, SCK: Serum creatine kinase | |
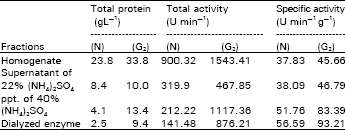
| Table 2: | Purification steps of alcohol dehydrogenase from G2-treated rabbit, compared to control (N) group |
 | |
| Table 3: | Lipids, cholesterol, RNA and DNA content in liver of G2-treated rabbit, compared to control (N) group |
 | |
Measurement of alkaline phosphatase activity revealed a result nearly the same with control. Meanwhile, the activity of acid phosphatase showed a slight increased value. Estimation of SCK of G-treated rabbits was increased with dose compared to control. Present results were agreed with Szent-Gyorgi and McLaughlin (1975) who demonstrated that glyoxal or methylglyoxal interact with biogenic amines and form biologically active free radicals which in relation to muscular dystrophy.
Partially purified alcohol dehydrogenase enzymes which isolated from G2-treated rabbit and control were assayed for their specific activities. The data (Table 2) emphasize that di-2-pyridylglyoxal was reacted with the enzyme with a rapid activation, reaches 64.71% of that of the native enzyme.
Moreover, previous studies carried out by Bryce (1970, 1973) have indicated that the reaction of NAD+ and ethanol was depend on about six amino groups at the active site of the dimeric enzyme, which involves imidazole of histidine and the amino groups of argenine as well as lysine residues.
It would appear that di-2-pyridylglyoxal was probably formed Schiff base via the oxygen atom of the carbonyl (s) group of the examined compound and the nitrogen of the imidazole, arginine or lysine residues on the active site, and that was agreed with Louis et al. (1975), or may be attributed to the reaction of the tested compound with some amino groups which are not essential for activity but just close enough to the active site. So that their substitution may interfere with binding of coenzyme and promotes the dissociation of enzyme-coenzyme complex, which is the rate limiting in the catalytic reaction, as reported by David and Bryce (1975).
Also, present results (Table 2) were in agreement with the data obtained by Canella and Sodini (1975), who suggested that the inactivated enzyme by glyoxal only had 11 argenine and 11 lysine modified residues per mole, reaches 10% inhibition. Meanwhile, 4 argenine and 3 lysine residues per mole only were modified in the enzyme at the maximum of activation.
On the other hand, estimation of total lipids in the liver of G2-treated rabbit (33.33 mg g-1) compared to control (58.54 mg g-1) would reflect a depression in lipid content, that about 43% (Table 3). These results were probably due to the lipolytic effect of the examined compound on hepatic cells. Consistently, liver cholesterol content generally parallels the total lipids, in which liver cholesterol of (G2) group was (1.49 mg g-1) compared to control group (27.69 mg g-1). It has been stated that, hepatic HMGCoA-reductase (rate limiting step enzyme) activity, was followed as a function of cholesterol concentration, since it is the final product of that biosynthetic process, as pointed out by Lionel et al. (1991).
Therefore, the obtained data demonstrated that di-2-pyridylglyoxal was an inhibitor to HMGCoA-reductase for about 94.63% inhibition, may be owing to the presence of pairs of electrons in the examined compound or its metabolic form. That result was in agreement with data reported by Ann et al. (1986), who found that some of the cyclohexanol derivatives which contained a hydroxyl group (OH) or were metabolized rapidly to a hydroxyl derivative were inhibitors of HMGCoA-reductase.
Furthermore, study of the hepatic DNA content, elaborated that the examined compound has a powerful negative effect on the replication of DNA to about 50% (0.12 mg g-1) compared to control (0.25 mg g-1). Meanwhile, estimation of hepatic RNA content revealed a slight decreased in value (10.15 mg g-1), compared to control (10.56 mg g-1). That was in agreement with Furihata et al. (1989) who demonstrated that glyoxal at doses of 50-550 mg kg-1 body weight induced DNA damage in the pyloric mucosa of rat stomach, two hours after its administration.
In conclusion, di-2-pyridylglyoxal was shown to be a powerful inhibitor of cholesterol biosynthesis, induced DNA damage that leads to the inhibitory action upon protein and RNA synthesis. As well as, this compound was found to be a powerful activator of alcohol dehydrogenase enzyme with a low degree of toxicity.