Research Article
In vivo Studies on Hepatic Fatty Acids and Cholesterol Pattern in Rabbits
Department of Biochemistry, Faculty of Science, Alexandria University, Alexandria, Egypt
Introduction
The biological effect of pyrolidine or its derivatives on mammalian tissues remains a subject of fundamental interest. Recent advances showed that (1-[ 2-hydroxy-3-octadecan-1-oate]propyl-tetramethyl pyrolidine)N-oxyl-3-carboxylate was used as a potential spin probe for membrane structure studies (Katoch et al., 1999). As well as, N-methyl-N-( 1-methyl-4-pyrolidino-2-butyl)-acetamide was used to release acetyl choline from rat and that derivative appeared to have agonist effect on the cholinergic receptor (Sethy et al., 1988).
In addition, studying the effect of pyrolidine or its copper-complex on rabbit melanocytes tyrosinase revealed that pyrolidine was a noncompetitive inhibitor of tyrosinase enzyme for about 30%, while its copper-complex was an activator for about 4.6 fold (Yacout, 2001).
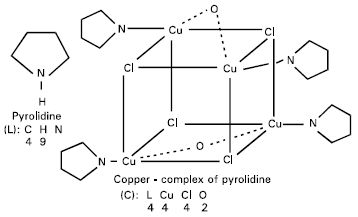 | |
| Fig. 1: | Tested Compounds |
In this research work, studying the (in vivo) effect of pyrolidine alone or its copper-complex, Fig. 1, upon the altered compositions of the lipid fraction in the hepatocytes of rabbit including the fatty acids and cholesterol, since the liver fatty acid composition is a reflection of the biological functions of membranes (Brenner, 1989).
Materials and Methods
nimals: Nine healthy rabbits weighing 80 ± 50 g, each were group housed and maintained under standard laboratory conditions. These were divided into three groups. The first group N served as control, received normal diet and corn oil, second group received normal diet with a daily oral dose of pyrolidine in corn oil (10 mg ml-1 for four days), while the third group was given normal diet with daily oral dose of pyrolidine-copper-complex in corn oil (10 mg ml-1 for four days) .
After the experimental period, animals were sacrificed, liver was removed and analyzed for fatty acid composition as well as cholesterol content. Also, blood samples were collected separately and each obtained serum was ready for assaying both cholesterol and triglycerides.
Separation of fatty acids : An accurate weight of liver 10 g of control, L-treated and C-treated rabbits, each separately were taken and extracted by refluxing in a soxhlet apparatus for 2 hours with about 180 ml of chloroform: methanol (CHCl3: CH3OH) mixture ( 2 : 1, v/v ) according to (Folch et al., 1957). Each lipid extract was evaporated to dryness and each amount of lipid was calculated. A sample from each lipid extract, 200 mg was saponified separately with 25 ml alcoholic KOH (0.5 M ) for 1 hour. Then acidification with concentrated HCl was followed by separation of the fatty acids.
Preparation of methyl esters of the fatty acids: Fatty acids were reacted with methanol (CH3OH) containing H2SO4 at 60 0C for 2 hours in a nitrogen atmosphere. The reaction mixture was mixed with 100 ml saturated salt (NaCl) solution, then 50 ml diethylether (C2H5-O-C2H5) was added. The ether layer was separated and the lower layer was extracted two times with 50 ml portions of ether. The combined ether phases were washed free of acid, dried and evaporated to dryness (Ustun, 1996). The obtained fatty acid methyl ester was then subjected to gas chromatographic (GC) analysis.
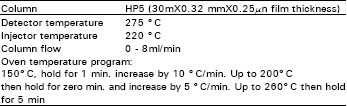
Gas chromatography: A 0.6 FL portion of each methylated sample was injected into the gas chromatograph , capillary column HP5 cross linked 5 % phenyl methyl silicone, using the conditions presented in Table 1.
| Table 1: | Conditions for GC analysis |
 | |
Estimation of cholesterol and triglycerides: Hepatic cholesterol was determined using Liberman-Burchard reagent (Harold, 1988). Cholesterol as standard was provided by Sigma (St. Louis, MO, U. S. A.). Total serum cholesterol as well as total serum triglycerides were measured using kits produced by Boehringer Mannheim, Germany.
Results
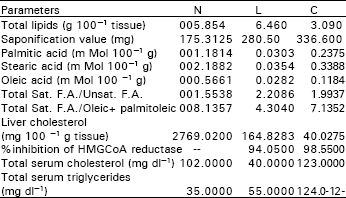
In this investigation, total lipids from the hepatocytes of L-treated and C-treated rabbits compared to control N were estimated. The data (Table 3) showed a marked decrease (3.090g 100-1 tissue) in total lipids of C- treated rabbits, while L- treated rabbits showed a slight increased value (6.460g 100-1 tissue) compared to control (5.854 g 100-1 tissue).
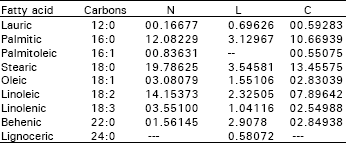
| Table 2: | Percentages of total peak area for fatty acid methyl esters separated from hepatocytes of pyrolidine- treated rabbit (L) and copper-complex-treated rabbit (C), compared to control (N) |
 | |
| Table 3: | Total lipids, fatty acids and cholesterol composition of rabbits treated with pyrolidine (L) or its copper-complex (C) compared to control (N) |
 | |
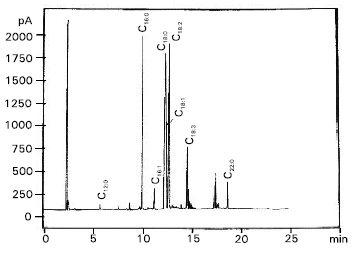 | |
| Fig. 2: | Gas chromatogram of total fatty acid methyl esters separated from hepatocytes of control rabbit (N) |
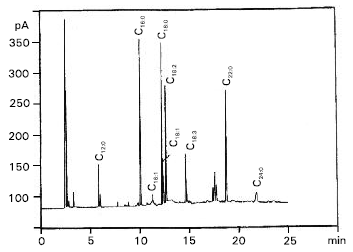 | |
| Fig. 3: | Gas chromatogram of total fatty acid methyl esters separated from hepatocytes of pyrolidine-treated rabbit (L) |
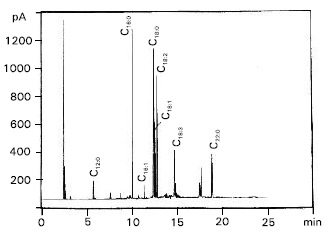 | |
| Fig. 4: | Gas chromatogram of total fatty acid methyl esters separated from hepatocytes of copper-complex-treated rabbit (C) |
Also, estimation of serum triglycerides in both cases compared to control revealed a distinct figures in which serum triglycerides were increased from 35 to 55 and 124 mg dl-1 for L-treated and C-treated rabbits, respectively. The chromatographic peaks, showed in Fig. 2, 3 and 4 were tentatively identified by comparison of obtained retention times with those of standards under the same conditions (Table 2). Total saturated fatty acids / unsaturated fatty acids ratio and total saturated fatty acids / oleic + palmitoleic acids ratio were calculated for each sample separately (Table 3).
The chromatograms illustrated the presence of nine identified fatty acids and traces of three unidentified fatty acids (Table 2). The main differences in fatty acid pattern can be observed (Table 3), the ratio of total saturated fatty acids/unsaturated fatty acids (S/U), changed from 1.5538 to 2.2086 and 1.9937, respectively. As well as, the ratio of total saturated fatty acids/oleic + palmitoleic was changed from 8.1357 to 4.3040 and 7.1352 respectively.
The level of total liver cholesterol was decreased from 2769.02 to 164.8283 and 40.0275 mg 100-1 tissue for L and C-treated ones, respectively. Meanwhile, total serum cholesterol was changed from 102.0 to 40.0 and 123.0 mg dl-1 (Table 3).
Discussion
In continuation to the work on the biological effect of some models of organic compounds, this research work is concerned with the in vivo effect of pyrolidine L and its copper-complex C on the hepatocyte fatty acids and cholesterol pattern. Accordingly, daily administration of L and C separately into rabbits for four days was carried out and the total lipids were extracted from the hepatocytes. A marked decrease was observed in the total lipids content in case of C-treated rabbits, while L-treated rabbits showed a slight increased value compared to control. This result probably due to the lipolytic effect of compound C, which leading to release of short chain fatty acids (seemed to be C12: 0). The obtained result was confirmed by the marked increase in saponification values, 336.600 and 280.500 for C and L–treated rabbits respectively, compared to control, 175.3125 mg.
In addition, estimation of serum triglycerides in both L and C–treated rabbits compared to control revealed a distinct figures in which serum triglycerides levels were increased. This result was in coincidence with many reports, which pointed out that, triglyceride measurement was used, in the diagnosis of disorders, showing that elevated serum triglyceride level was seen in the primary disorder of lipid metabolism (Henry, 1991).
Hepatic fatty acids, which separated from L and C-treated rabbits, beside control were transmethylated and detected using gas chromatography, showing nine identified fatty acids, and traces of three unidentified fatty acids, due to the unavailability of authentic standards. The obtained results would reflect a depression in the palmitic, stearic and oleic acid contents in case of L more than in C–treated rabbits and that seemed to be due to the activation of adenylate cyclase, which increased the level of C-AMP, then stimulate the protein kinase, which in turn activates the lipase by phosphorylating it ,thus tested compounds may induce lipolysis (Stryer, 1995).
As well as, the main differences in fatty acids pattern can be observed in the increased ratio of total S / U, associated with a decrease in S / oleic + palmitoleic ratio. The obtained data showed that pyrolidine and its copper-complex were act as inhibitors of both Δ5 and Δ6-desaturase activities. In contrast, the correlated decrease in S / oleic + palmitoleic ratio may indicated that compound L had a stimulatory effect on the Δ9-desaturase activity more than that of compound C.
The previous data showed that the major product of fatty acid synthase is palmitate, and longer fatty acids were formed by elongation reactions catalyzed by enzymes on the cytosol and the microsomal systems also introduce double bonds into long chain acyl CoA such as conversion of stearoyl CoA into oleoyl CoA by Δ9-desaturase enzyme which contain non heme iron atom (Stryer, 1995).
Therefore, the obtained data suggesting that compound L was an electron donor , apparently increase the conversion of non heme iron atom ( Fe+++) of Δ9-desaturase into the ( Fe++) state, which enables it to interact with O2 and saturated fatty acyl substrate, while compound C showed less Δ9-desaturase activity.
On the other hand, it was known that most of the cholesterol synthesis in vertebrates takes place in the liver. A small fraction of it is incorporated into the membranes of hepatocytes, but most of it is exported to other organs such as adrenal glands and gonads (Lehninger et al., 1993). The primary regulation of cholesterol biosynthesis appears to be centered on the HMGCoA reductase reaction as a rate limiting step (Zubay, 1993). In consequence, the effect of pyrolidine and its copper-complex, administrated in vivo and separately, on hepatic HMGCoA reductase activity four days after dosing was followed as a function of cholesterol concentration, since it is a final product of that biosynthetic process (Lionel et al., 1991). The obtained results revealed that L and C were inhibited HMGCoA reductase catalyzing reaction with % inhibition about 94.05 and 98.55 %, respectively, probably due to the pair or pairs of electrons in compound L and C respectively.
These results are in agreement with data of Ann et al. (1986), who found that some of the cyclohexanol derivatives which contained only a hydroxyl group or were metabolized rapidly to a hydroxyl derivative were inhibitors of HMGCoA reductase by about 59 %, also found that menthol, which is structurally similar to trimethylcyclohexanol inhibited hepatic HMGCoA reductase activity by 70 % when administrated in vivo 17 hours prior to enzyme assay, whereas, cyclopentanol inhibited the enzyme by 37%, but this was not significant suggesting a certain degree of ring size specificity for inhibition by compounds of this nature.
As well as, the effect of L and C as modulator of blood cholesterol level is well established, giving distinct result, compared to control. The obtained result suggesting that inhibition of hepatic cholesterol biosynthesis results in a depression of intracellular cholesterol, which is accompanied by many alterations in cell cholesterol metabolism and may up regulate as well as a delivery of exogenous cholesterol. This result was in agreement with that obtained by Sviridov et al. (1990). In addition, Safonova et al. (1994), suggesting that inhibition of cholesterol biosynthesis results in a dose–dependent stimulation of cholesterol uptake .
Therefore, compound L caused a decrease in cholesterol absorption in rabbits; meanwhile compound C showed an increase in cholesterol uptake by rabbit enterocytes.
It may be concluded that, tested compounds have a distinct influence on the rabbit hepatocytes fatty acids cholesterol as well as serum cholesterol and triglycerides patterns. Hope these compounds could be used in the treatment of patients with hypercholesterolemia.
Acknowledgment
The author is grateful to Prof. Dr. Abd El Rahman M. Professor of inorganic Chemistry, Department of Chemistry, Alexandria University, Egypt, for his kind gift (tested compounds).