Research Article
Fumaric Acid Production by Rhizopus oryzae on Corn Distillers` Grains with Solubles
Department of Biology and Microbiology, South Dakota State University, Brookings, 57007 SD, USA
The commercially important specialty chemical fumaric acid has a number of commercial applications including its use in foods, beverages, detergents, animal feeds, paper sizing, printing inks and pharmaceuticals (Bigelis and Tsai, 1995; Tsao et al., 1999; Lee et al., 2004). More than 6 million pounds of fumaric acid are utilized per year in foods alone (Bigelis and Tsai, 1995). Fumaric acid is utilized to regulate the acidic flavor of bakery products, fruit juice drinks, wines, jams, jellies and pie fillings (Dziezak, 1990). Relative to its medical use, fumaric acid esters are used to treat the skin condition psoriasis caused by impaired fumaric acid production (Brewer and Rogers, 2007).
Fumaric acid can be synthesized chemically from the petroleum-based product maleic anhydride using acid-hydrolyzed isomerization (Moresi et al., 1992). Fumaric acid can also be synthesized by submerged fermentation using selected strains of the fungus Rhizopus oryzae. These strains likely accumulate fumaric acid due to the involvement of reductive reactions of the tricarboxylic acid cycle (Kenealy et al., 1986). Metabolic modeling of fumaric production by R. oryzae has also been examined to determine how production can be improved (Gangl et al., 1991). Although R. oryzae is capable of using fermentable sugars to support fumaric acid production (Rhodes et al., 1959, 1962; Cao et al., 1996), the fungus has also been shown to produce fumaric acid on substrates such as corn mash, corn starch, corn steep liquor, potato flour, rice bran and cassava bagasse (Rhodes et al., 1962; Morrin and Ward, 1990; Moresi et al., 1991, 1992; Carta et al., 1999; Moon et al., 2004).
During the corn dry-milling production of ethanol, a primary coproduct is corn distillers’ grains with solubles. Approximately 16 pounds of 90% corn distillers’ grains, with solubles are produced from each bushel of corn processed at ethanol plants (Rausch and Belyea, 2006). Presently, corn distillers’ grains with solubles is used as a protein supplement in animal feeds (Ham et al., 1994). With more than 7 million tons of grains being processed for ethanol production per year, the low value grains could be better used to produce the specialty chemical fumaric acid (Rausch and Belyea, 2006). The remaining fermentable sugars and starch in corn distillers’ grains with solubles could support microbial growth as a carbon source (Moyer, 1953; Moresi et al., 1992). The main supporting benefit of this bioconversion research would be a more economical way to produce fumaric acid from a low cost substrate. In this study, three known fumaric acid-producing strains of R. oryzae were tested for their ability to produce the acid from untreated and treated corn distillers’ grains with solubles by solid-state fermentation. The objectives of this report were to compare fumaric acid production, biomass production and fumaric acid productivity of the strains after growth on the untreated and treated corn distillers’ grains with solubles using solid-state fermentation.
Strains and Growth Conditions
In this study, the fumaric acid-producing strains Rhizopus oryzae ATCC 10260, ATCC 20344 and ATCC 52918 (Rhodes et al., 1959, 1962; Cao et al., 1996), were used. When sterilized by autoclaving, the corn distillers’ grains with solubles was subjected to 121°C at 17 pounds (square inch)-1 of pressure for 20 min. Acid-treated grains were treated with 0.5 to 2.0% H2SO4 and autoclaved for 20 min (Dunning and Lathrop, 1945). Following autoclaving, the pH of the acid-hydrolyzed grains was adjusted to 6.0. A loopful of fungal mycelium of each R. oryzae strain was inoculated into potato dextrose broth (20 mL) and the culture was grown for 72 h at 25°C. The inoculum (500 conidia mL-1) was added to 5 g corn distillers grains (82% moisture) in a sterile 125 mL Erlenmeyer flask and the fungus was grown for 240 h at 25°C. The experiments were conducted (Brookings, SD, USA) from November 2004 through January 2006 and the source of the corn distillers’ grains with solubles was Dakota Ethanol LLC (Wentworth, SD, USA).
Processing of Grains
After 240 h at 25°C, the fumaric acid present in each solid-state fermentation culture was collected using the following procedure. To each culture, sterile water (25 mL) was added. After shaking each culture for 60 min at 25°C, the grains were filtered through a Whatman No. 1 filter. The fungal biomass in each culture was washed with sterile water (10 mL) and also filtered through a Whatman No. 1 filter. Biomass production was determined gravimetrically as previously described (Xie and West, 2006). Biomass levels are expressed as (g) cell weight (g grains with solubles)-1. The filtrates from each culture were combined. To precipitate any protein present in each culture filtrate, ice-cold 0.5 N HClO4 (0.5 mL) was added and the filtrate was stirred. Any protein precipitate present was removed. The filtrate was subsequently neutralized to pH 7.0 with 1 N NaOH. The volume of each culture filtrate was recorded.
Fumaric Acid Assay
The neutralized filtrate was assayed for its fumaric acid content using a modified spectrophotometric assay (Beeckmans and Kanarek, 1977). The assay mix (1 mL) contained 25 mM potassium phosphate buffer pH 7.5, 4.6 units porcine heart fumarase and filtrate. The commercially obtained fumarase (Sigma Chemical Co., St. Louis, MO, USA) had a specific activity of 324 units (mg protein)-1. Fumaric acid standards were also run using the assay. The reaction was monitored at 275 nm by following the decrease in absorbance that is proportional to the concentration of fumaric acid present in the sample. Fumaric acid production was expressed as (g) fumaric acid (kg grains with solubles)-1. Fumaric acid productivity is given as g fumaric acid (kg grains with solubles)-1 h-1. All values represent the mean of three independent determinations involving three separate cultures. The Student’s t-test was used during statistical analysis.
There is considerable interest in producing fumaric acid from a renewable resource such as corn distillers’ grains with solubles (Tsao et al., 1999; Lee et al., 2004). In this investigation, the ability of R. oryzae ATCC 10260, ATCC 20344 or ATCC 52918 to utilize untreated or treated corn distillers’ grains with solubles for fumaric acid production using solid-state fermentation was studied. The fungal strains were grown on untreated corn distillers’ grains with solubles, autoclaved grains or acid-hydrolyzed grains for 240 h at 25°C. The three strains produced fumaric acid on the untreated or treated grains but the treated grains supported a higher level of fumaric acid production by the strains than did the untreated grains (Table 1). Relative to fumaric acid production by ATCC 10260 on the untreated grains, the fungus produced 1.6-fold higher fumaric acid levels on the autoclaved grains (p<0.05) and produced significantly (p<0.01) higher acid concentrations on the acid-hydrolyzed grains (Table 1). Although fumaric acid production by ATCC 20344 was higher on the autoclaved grains, a statistically significant difference in production was not observed (Table 1). Compared to fumaric acid production by ATCC 20344 on the untreated grains, fumaric acid production by the strain was significantly higher on the 0.5% (p<0.05), 1.0% (p<0.01), 1.5% (p<0.01) or the 2.0% (p<0.01) acid-treated grains (Table 1). The highest fumaric acid concentration was produced by ATCC 20344 grown on 1.0% acid-treated grains which was 2.5-fold higher than the level produced by the strain grown on the untreated grains (Table 1). There was a significant difference in fumaric acid production by ATCC 52918 grown on the untreated compared to the treated grains (Table 1). Relative to the untreated grains, fumaric acid production by ATCC 52918 was significantly increased when ATCC 52918 was grown on the autoclaved or acid-treated grains (p<0.01) with the highest level being found after fungal growth on the 1.0% acid-treated grains. A 4.3-fold higher fumaric acid concentration was witnessed after growth on the 1.0% acid-treated grains compared to the untreated grains (Table 1).
Biomass production by the three R. oryzae strains grown on the untreated and treated grains was investigated (Table 2). After 240 h of growth, biomass production by ATCC 10260 was significantly higher for the autoclaved (p<0.05), 0.5% H2SO4-hydrolyzed (p<0.05), 1.0% H2SO4-hydrolyzed (p<0.01) and 1.5% H2SO4-hydrolyzed (p<0.01) grains compared to the untreated grains (Table 2).
| Table 1: | Fumaric acid production by Rhizopus oryzae strains on untreated or treated corn distillers’ grains with solubles as a substrate following 240 h of growth |
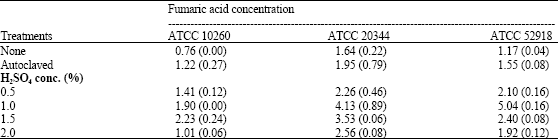 | |
| The results are expressed as g fumaric acid (kg grains with solubles)-1 and represents the mean of three separate trials (standard deviation) | |
| Table 2: | Biomass production by Rhizopus oryzae strains on untreated or treated corn distillers’ grains with solubles as a substrate following 240 h of growth |
 | |
| The results are given as g cell weight (g grains with solubles)-1 and indicates the mean of three separate trials (standard deviation) | |
Biomass production by ATCC 10260 decreased by about half when it was grown on the 2.0% H2SO4-hydrolyzed grains compared to the untreated grains (Table 2). Biomass production by ATCC 20344 grown on the untreated or treated grains was the highest of the three strains examined (Table 2). Biomass production by ATCC 20344 was increased after growth on the autoclaved or acid-treated grains compared to the untreated grains (Table 2). Biomass production by ATCC 20344 was significantly higher on the autoclaved (p<0.01), 0.5% H2SO4 (p<0.05), 1.0% H2SO4 (p<0.01) and 1.5% H2SO4-hydrolyzed (p<0.01) grains relative to the untreated grains. The treatment of the grains affected biomass production by ATCC 52918 very little (Table 2). Only the autoclaving of the grains resulted in a statistically significant (p<0.01) decrease in biomass production by ATCC 52918 relative to its production on the untreated grains (Table 2).
The fumaric acid productivity for all three strains was found to improve following treatment of the grains (Table 3). Autoclaving of the grains had no significant effect upon the fumaric acid productivity of ATCC 10260 or ATCC 20344 compared to the untreated grains (Table 3). In contrast, the fumaric acid productivity of ATCC 52918 grown on the autoclaved grains was twofold higher than its productivity on the untreated grains with the difference being statistically significant (p<0.01). The fumaric acid productivity of each strain grown on 1.0 or 1.5% acid-treated grains was significantly higher (p<0.01) than its productivity on the untreated grains. Treatment of the grains with 0.5% H2SO4 was also found to substantially increase (p<0.01) the fumaric acid productivity of ATCC 10260 and ATCC 52918 while growth of ATCC 20344 on 2.0% H2SO4-hydrolyzed grains significantly elevated (p<0.01) its productivity.
Fumaric acid production by strains of R. oryzae using solid-state fermentation of fermentable sugars or agricultural-based substrates does not appear to have been examined previously. Prior studies have examined the submerged fermentation by Rhizopus species on a variety of substrates. It has been shown that R. oryzae ATCC 10260 or ATCC 52918 produced 38.5 or 43 g L-1 fumaric acid, respectively, after growth on a medium containing 16% glucose and 0.37% urea for 72 h at 33°C (Rhodes et al., 1959). Fumaric acid production by ATCC 52918 was found to be lower at 25.9 g L-1 when the medium contained 10% glucose and 6% urea following growth at 32°C for 96 h (Goldberg and Stieglitz, 1994). It has been shown that the growth morphology of the fungal cells caused by the pH of the cultivation medium affected fumaric acid and biomass production by R. oryzae (Morrin and Ward, 1990; Zhou et al., 2000). ATCC 52918 only produced 1.78 g L-1 fumaric acid in a glucose-peptone medium after 48 h in a fermenter containing clumped fungal cells while biomass production was found to be 2 g L-1 (Morrin and Ward, 1990). Biomass production by ATCC 52918 on a medium containing molasses as a carbon source was noted to be much higher after 72-168 h of growth (Rhodes et al., 1959) than the biomass levels determined for ATCC 52918 after growth on the grains for 240 h (Table 2). This would be expected because fungal cell growth on molasses using submerged fermentation would be more rapid than fungal cell growth on the grains during solid-state fermentation.
| Table 3: | Fumaric acid productivity by Rhizopus oryzae strains grown on untreated or treated corn distillers’ grains with solubles as a substrate for 240 h |
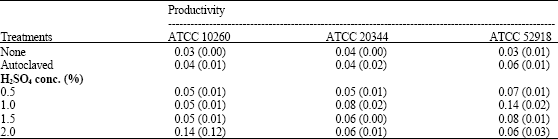 | |
| The results are expressed as g fumaric acid (kg cell weight)-1 h-1 and represents the mean of three independent trials (standard deviation) | |
Some of the agricultural substrates used to support fumaric acid production by Rhizopus species include potato flour, rice bran, enzymatically hydrolyzed cassava bagasse and corn substrates (Rhodes et al., 1962; Morrin and Ward, 1990; Moresi et al., 1991, 1992; Carta et al., 1999; Moon et al., 2004). With respect to corn substrates, it was shown that ATCC 52918 produced 27 g L-1 fumaric acid in shake flask cultures of a corn mash medium containing 2 g L-1 ammonium sulfate after 90 h at 33°C (Rhodes et al., 1962). Corn steep liquor was found to support little fumaric acid production by ATCC 52918 in a medium containing 6 g L-1 glucose but did support a 85-fold higher level of biomass production in a fermenter after 48 h (Morrin and Ward, 1990). Biomass production by ATCC 52918 on the grains for 240 h using solid-state fermentation was much lower than biomass production by the strain grown on the glucose-peptone medium for 48 h using submerged fermentation (Table 2). It was also demonstrated that acid-hydrolyzed or enzymatically treated corn flour supported a high level of fumaric acid production by ATCC 10260 in shake flask cultures after growth for 144 h at 32°C (Moresi et al., 1992). Similar to the finding that acid hydrolysis of corn flour improved its use as a substrate for fumaric acid production by ATCC 10260 (Moresi et al., 1992), the acid hydrolysis of the corn distillers’ grains with solubles improved its ability to support fumaric acid production by the strains selected in this study (Table 1). It is clear from the results of the present study that fumaric acid production by ATCC 10260 or ATCC 52918 was much lower using solid-state fermentation than using submerged fermentation. This is also reflected in fumaric acid productivity by ATCC 52918. When the strain was grown on molasses or potato flour using submerged fermentation (Moresi et al., 1991, 1992), its productivity was about double compared to the highest fumaric acid productivity observed in this study when it was grown on the acid-hydrolyzed grains using solid-state fermentation (Table 3). Although the fumaric acid levels produced by the solid-state fermentation of the grains by the three R. oryzae strains were relatively low compared to fumaric acid production on substrates with a higher sugar content using submerged fermentation, the quantity of grains being produced per year from the dry-milling of corn for ethanol production would still allow a significant amount of fumaric acid to be fermented.
In summary, selected strains of R. oryzae were capable of fumaric acid production whether they were grown on untreated or treated corn distillers’ grains. The acid-treatment of the grains resulted in increased fumaric acid production by the strains compared to the untreated grains. Fumaric acid production was observed to be highest when ATCC 52918 was grown on the 1% acid-treated grains. Biomass production by ATCC 20344 on the untreated or treated grains was higher than the biomass levels produced by ATCC 10260 and ATCC 52918. Fumaric acid specific productivity of all three strains was increased on the treated grains compared to the untreated grains. It appeared that treatment of the grains was necessary to increase fumaric acid production by the strains tested.
Published as paper 3608, Journal Series, South Dakota Agricultural Experiment Station. This study was supported by the South Dakota AES and South Dakota Corn Utilization Council. The results and the mention of brand or firm names does not constitute an endorsement by the South Dakota AES over those mentioned but of a similar nature.