ABSTRACT
In this study Daphnia magna enriched for three different time period (3, 6 and 9 h) and non-enriched Daphnia magna were fed to the whitefish fry (average weight 54.7±0.5 mg) during 14 days. A significant growth difference between fry fed with enriched and non-enriched Daphnia was observed (p<0.05), while survival rate did not significantly differ among the treatments (p<0.05). Furthermore, the highest pH stress resistance was found in those larvae fed with Daphnia enriched for 9 and 6 h (p<0.05). We observed that feeding of the whitefish larvae with HUFA-enriched Daphnia resulted in growth improvement compared to non-enriched Daphnia. Therefore, to achieve higher production, nutrition optimization of the whitefish larvae especially by using HUFA in daily foods and transferring it to fish body through live food is recommended for whitefish hatcheries. The result of this study proved that feeding the whitefish with live food containing high n-3 HUFA content increased larval resistance to pH stress.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jfas.2010.49.55
URL: https://scialert.net/abstract/?doi=jfas.2010.49.55
INTRODUCTION
Rearing of larval fish is the most critical stage in the production cycle for many species. The primary problem in rearing relates to the transitional period from endogenous to exogenous food resources and thus to adequate feed supply (Abi and Kestemont, 1994). A readily available diet which has a high nutritional quality and is easily accepted and digested by the larval fish is essential to success (Kim et al., 1996). Dietary lipids play an important role in fish nutrition for provision of both Essential Fatty Acids (EFA) and energy. Dietary lipids also assist in the absorption of fat-soluble nutrients (Sargent et al., 1999). Live prey organisms, especially zooplankton, are generally used as initial larval food for certain species of fish (Legar et al., 1986). Bering naturally low in EPA, live foods commonly used for first feeding of larvae, such as Rotifer and Artemia, have to be enriched with lipids rich in EPA prior feeding (Copeman et al., 2002). Daphnia sp., however poor in EPA, is one of the most important starter live food in whitefish in Iran. Therefore, Daphnia EFA-enrichment as a live food is needed to meet the requirements of sturgeon for EFA.
Some methods have significantly enhanced EFA level in Daphnia sp. (Ravet et al., 2003; Von Elbert, 2002), but no study exists to test the effects of EFA-enriched Daphnia on the performance of whitefish larvae. This study presents a specific approach to enrich Daphnia with cod liver oil. The study also aims at evaluating the role of HUFA in the first feeding of the whitefish larvae and their effects on the growth performance and body composition of the larvae.
MATERIALS AND METHODS
Whitefish larvae (10 days post hatch) were obtained from the Shahid Beheshtee Center in Anzali, Iran from January to April 2008.
The larvae were fed with Artemia nauplii (Instar 1) for two days before collection. The larvae with initial wet weight of 54.7±0.5 mg and total length of 22.3±0.3 mm were randomly distributed in groups of 150 individuals per tank into 12 rectangular elliptic fiberglass tanks of 15 L each (10 larvae L-1). Each tank was supplied with water via 1 inch PVC pipe at a flow rate of 3 L min-1. Water was continuously aerated (compressed air) to keep oxygen levels close to 5.8±0.3 mg L-1 (n = 56). The tank outlet and inlet was protected by a 250 μm net screen. Water quality was checked periodically; pH was about 7.8±0.02 (n = 28), temperature was 19±1°C (n = 56) and photoperiod was 12 L: 12 D.
Daphnia magna of the mean length size 1.6±0.15 mm (n = 30) were collected from an earthen pond in Shahid Beheshtee Hatchery Center (Anzali, Iran). Enrichment solution was prepared with cod liver-oil (Seven Seas), polysorbate (Tween 80, Merck) and freshwater according to Ako et al. (1994). In this method, first, 5 mL polysorbate was added to 50 mL freshwater and mixed carefully, then 50 mL cod liver oil was added to the solution and mixed; 0.3-0.5 mL of the final solution as an enrichment solution was used for 1 L of the incubator. Daphina magna at a density of 10,000 ind. L-1 were enriched in an incubator at a temperature of about 20°C according to methods described by Von Elert (2002) and three different enrichment exposure times were employed (3, 6 and 9 h). The solutions were prepared daily in order to maintain the quality at comparable level throughout the experiment.
Four different Daphnia enrichment treatments were tested (analysis was performed in triplicate): (1) Non-enrichment Daphnia, (2) enriched Daphnia with Cod Liver Oil (CLO) in 3 h, (3) enriched Daphnia with CLO in 6 h and (4) enriched Daphnia with CLO in 9 h. Larvae were fed Daphnia ad libitum (Kolkovski et al., 2000) four times per day.
The survival rate in each treatment was calculated, based on counting the number of dead larvae. The wet weight of larvae was calculated, based on counting the number of dead larvae. The wet weight of larvae was measured by randomly sampling of 10 larvae in each replicate on 3rd, 5th, 12th and 14th days. Growth index of fish calculated based on following equations (Lee et al., 2003).
| (1) |
| (2) |
After 14 days of larval rearing, the resistance of larvae against extreme pH values was determined at pH levels of 4.5 and 11, respectively. First, 10 L larval rearing water was poured into a 60 L aquarium and 12 small baskets were placed inside the aquarium. The designated pH of the water was adjusted with 0.1 N HCL and 1 N NaOH. Then, 30 larvae were taken randomly from each replicate and transferred to the small baskets inside the aquarium. The larval survival was calculated by counting of the dead larvae after 30 min pH stress test. Artificial sea water (25 g L-1) was made by adding marine salt into the water in order to evaluate larvae resistance against salinity stress (Kolkovski et al., 2000). From each tank, 30 larvae were collected and transferred into the small baskets inside the aquarium. The larval survival was computed by counting of the dead larvae after 15 min salinity stress test.
Enough samples (100 samples) of Daphnia and 10 larvae from each replicate were collected and kept in a freezer before analyzing. The extraction of lipid from Daphnia and fish larvae was done through saponification with 2 g NaOH in 100 mL methanol according to Folch et al. (1957). Fatty acid methyl esters were prepared by transesterification with borontrifluoride (BF3) in methanol (Metcalfe and Schmitz, 1961). Fatty acids methyl esters were measured in Unicam 4600 gas chromatograph (USA) with flame ionization detector. The column was BPX 70 with a capillary column of 30 m length and ID of 0.25 mm. The carrier gas was helium at a flow rate of 30 mL sec-1. Detector and injector temperature were 250 and 240°C, respectively. The thermal gradient was 170°C for 5 min, then increased by 3°C min-1 -200°C and held this temperature for 20 min. The fatty acids were quantified by comparing areas of their peaks with a peak of an internal standard, (C18: 0). The peaks of fatty acids were carried out by connecting the GC with personal computer and utilizing Millennium software. Peak identification was performed by means of standard sample.
To analyze statistical results, one-way ANOVA was applied and the mean comparison was done through LSD test at reliability level of 5%. All variances were checked for normality and homogeneity. Data analysis was done in SPSS software (release 12.0).
RESULTS
Eicosapentaenoic acid (EPA, 22:5 n-3) content in non-enriched Daphnia was about 0.06 mg g-1 DW and docosahexanoic acid (DHA, 22:6 n-3) was not detected. After enrichment with cod liver-oil for 3, 6 and 9 h, EPA content increased to about 0.13, 0.43 and 0.69 mg g-1 DW of Daphnia, respectively; however, DHA content was only detected in Daphnia enriched for 3 and 6 h. The highest n-3 HuFA concentration observed for 9 h enriched Daphnia (0.69 mg g-1 DW) (Table 1).
Content of n-3 HUFA in larvae fed the enriched Daphnia was improved accordingly with increments of the enrichment times. This means that larvae with the highest n-3 HUFA (2.39 mg g-1 DW) were those that were fed with fed with the Daphnia enriched diet exposed at 9 h (Table 2).
We t weight of the whitefish after the 1st, 3rd, 5th, 12th and 14th day of rearing are shown in Fig. 1. Growth of those larvae fed the 9 h enriched Daphnia was better than in all the other treatments. The lowest growth was observed in larvae fed with non-enriched Daphnia.
| Table 1: | Average fattyacid content in Daphnia magna before and after enrichment in three different times during 3, 6 and 9 h (in mg/day/g of Daphnia)* |
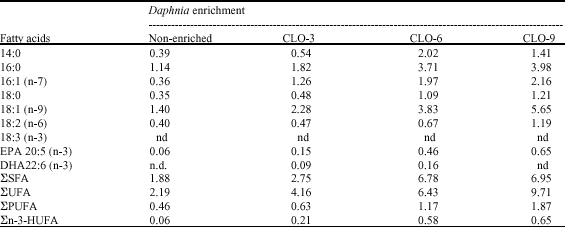 | |
| *Values are expressed the means from two replicate. ΣSFA: Total saturated fatty acid; ΣUFA: Total unsaturated fatty acid; ΣPUFA: Total polyunsaturated fatty acid; Σ n-3 HUFA: Total n-3 highly unsaturated fatty acid; nd: Not detected; EPA: Eicosapentaenoic acid; DHA: Docosahexanoic acid; CLO: Cod liver-oil | |
| Table 2: | Average fatty acid content in the whitefish larvae at the end of the experiment (in mg per dry gram)* |
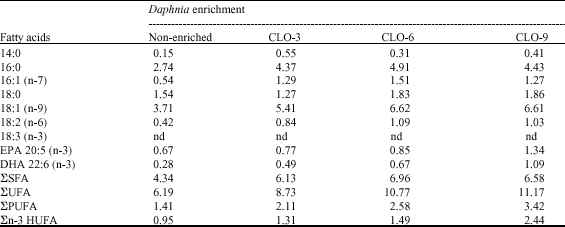 | |
| *Values are expressed the means from two replicate. nd: Not detected; ΣSFA = Total saturated fatty acid; ΣUFA: Total unsaturated fatty acid; ΣPUFA: Total polyunsaturated fatty acid; Σn-3 HUFA: Total n-3 highly unsaturated fatty acid; nd: Not detected; EPA: Eicosapentaenoic acid; DHA: Docosahexanoic acid; CLO: Cod liver-oil | |
| Table 3: | Average growth and survival of the whitefish larvae fed with Daphnia enriched at various levels of time* |
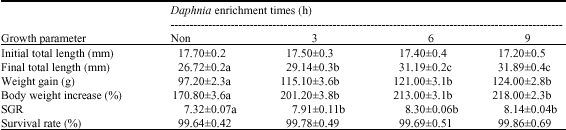 | |
| *Mean±SD of three replicates. Numbers within the same row with different letter(s) are significantly different (p<0.05) | |
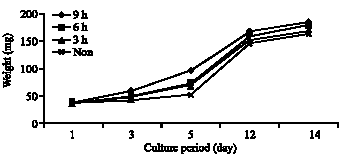 | |
| Fig. 1: | Average weight of the whitefish larvea during 14 days, fed non enriched Daphnia and 3, 6 and 9 h enriched Daphnia. Each data point represents mean weight of three replicates (n = 30) |
Table 3 shows significant differences between growth of the fish larvae fed with non-enriched and enriched Daphnia (p<0.05), but no significant differences are observed among the enriched treatments (p>0.05). The highest weight gain, SGR and final total length were observed in the larvae that fed with 9 h enriched Daphnia. Survival of fish fed enriched Daphnia was not significantly different from that fed non enriched Daphnia (p>0.05).
The highest resistance was observed in those larvae fed with enriched Daphnia in 9 h. Although, significant differences between enriched and non-enriched treatments were evident (p>0.05), the difference between 6 and 9 h enrichment was not significant (p>0.05). In addition, results of high pH condition showed significant differences among the treatments (p<0.05). The highest and lowest survivals were observed in 9 h enriched and non enriched Daphnia, respectively. Results of salinity stress showed that there were not any significant differences among the treatments; survival was very low in all groups, as it is shown in Fig. 2.
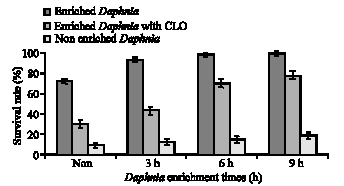 | |
| Fig. 2: | Average survival rate (%) of the whitefish larvae in pH (30 min exposure) and salinity stress tests (15 min). Each data point represent mean weight and standard deviation of three replicates (n = 90) |
DISCUSSION
Many different methods have been used for enrichment of live foods (Coutteau and Sorgeloos, 1997; Weers and Gulati, 1997; Von Elert, 2002; Ravet et al., 2003). The enrichment method of Daphnia boosted up considerably its fatty acids content in comparison to similar studies conducted with different oil sources for enrichment of Artemia (Kraul et al., 1993; Ako et al., 1994).
The EPA of Daphnia was increased with the increment of enrichment times. However, DHA was detected in the 6 h of enriched Daphnia and not detected in 9 h (Table 1). It maybe related to Daphnia ability to convert α-LA and DHA to EPA, which was also supported by Sundbom and Vrede (1997), Von Elert (2002) and Ravet et al. (2003). Von Elert (2002) suggested additional EPA in food of Daphnia galeata only increased in EPA content. He also showed that EPA was not converted to other fatty acids but DHA could be converted to EPA. This conversion is well-known in Artemia franciscana and Brachionus plicatilis that have been shown to catabolise DHA selectively during starvation (Von Elvert, 2002). This reason might be due to fast filtration of Daphnia that they use DHA to obviate their starvation in 9 h enrichment.
It is worth stating that both of EPA and DHA play a critical role in growth and evolution of fish larvae specially sea fishes (Watanabe et al., 1983; Kraul et al., 1993). It was also observed that feeding of the whitefish larvae with HUFA-enriched Daphnia resulted in growth improvement compared to non-enriched Daphnia. Since enriched Daphnia provides necessary energy, larvae can pass better through external nutrition and as a result production will increase. Therefore, to achieve higher production, nutrition optimization of the whitefish larvae especially by using HUFA in daily foods and transferring it to fish body through live food is recommended for whitefish hatcheries.
Survival rate whitefish larvae was not affected by enrichment, which was in agreement with Kolkovski et al. (2000), who did not find any relationship between HUFA enrichment and walleye (Stizostedion vitreum) larval survival.
The result of this study proved that feeding the whitefish with live food containing high n-3 HUFA content increased larval resistance to pH stress (Fig. 2). The competitive interactions between EPA and AA are important in the formation of eicosanoids. Ecosanoids are a group of biologically active molecules, once known as local hormones, which include prostaglandins, thromboxanes and leukotrienes (Sargent et al., 1999). Several studies have demonstrated that prostaglandins (PGs) are involved in the control of osmoregulatory processes and the regulation of the stress included Hypothalamus-Pituitary-Interrenal (HPI) axis, which facilitates the release of cortisol, the main corticosteroid in teleost fish (Gupta et al., 1985; Wales, 1988). Relationship between feeding of larvae with HUFA-enriched Artemia and enhancing resistance against environmental stress has been reported in other species of fishes (Ako et al., 1994). According to the results of this, Daphnia HUFA level was enhanced by increasing the time and, therefore, resulting in a better larval development, stress resistance and production enhancement.
REFERENCES
- Abi, A. and P. Kestemont, 1994. Compartion of nutritional status of goldfish (Carassius auratus) larvae fed with live, mixed, or diet. Aquaculture, 128: 163-176.
Direct Link - Ako, H., C.S. Tamaru, P. Bass and C.S. Lee, 1994. Enhancing the resistance to physical stress in larvae of Mugil cephalus by the feeding enriched Artemia nauplii. Aquaculture, 122: 81-90.
CrossRef - Copeman, L.A., C.C. Parrish, J.A. Brown and M. Harel, 2002. Effects of docosahexaenoic, eicosapentaenoic and arachidonic acids on the early growth, survival, lipid composition and pigmentation of yellowtail flounder (Limanda ferruginea): A live food enrichment experiment. Aquaculture, 210: 285-304.
CrossRefDirect Link - Coutteau, P. and P. Sorgeloos, 1997. Manipulation of dietary lipids fatty acids and vitamins in zooplankton cultures. Freshwater Biol., 38: 501-512.
CrossRef - Folch, J., M. Lees and G.H.S. Stanley, 1957. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem., 226: 497-509.
CrossRefPubMedDirect Link - Kim, J., K.C. Masse and R.W. Hardy, 1996. Adult Artemia as food for first feeding coho salmon (Oncorhynchus kisutch). Aquaculture, 144: 217-226.
CrossRef - Kolkovski, S., S. Czesny, C. Yackey, R. Moreau and F. Cihla et al., 2000. The effect of vitamin C and E in (n-3) highly unsaturated fatty acid-enriched Artemia nauplii on growth, survival and stress resistance of freshwater walleye (Stizostedion vitreum) larvae. Aquacul. Nutr., 6: 199-200.
CrossRef - Kraul, S.H., K. Brittain, R. Cantrell and T. Nagao, 1993. Nutritional factors affecting stress resistance in the larval mahimahi, Coryphaena hippurus. J. World Aquacul. Soc., 24: 186-193.
CrossRef - Lee, S.M., J.H. Lee and K.D. Kim, 2003. Effect of dietary essential fatty acids on growth, body composition, and blood chemistry of juvenile starry flounder (Platichthys stellatus). Aquaculture, 225: 269-281.
CrossRef - Leger, P., D.A. Bengtson, K.L. Simpson and P. Sorgeloos, 1986. The use and nutritional value of Artemia as a food source. Oceanogr. Mar. Biol., 24: 521-623.
Direct Link - Ravet, J.L., M.T. Brett and D.C. Muller-Navarra, 2003. A test of the role of polyunsaturated fatty acids in phytoplankton food quality for Daphnia using liposome supplementation. Limnol. Oceanogr., 48: 1938-1947.
Direct Link - Sundbom, M. and T. Vrede, 1997. Effects of fatty acid and phosphorus content of food on growth, survival and reproduction of Daphnia. Freshwater Biol., 38: 665-674.
CrossRef - Von Elert, E., 2002. Determination of limiting polyunsaturated fatty acids in Daphnia galeata using a new method enrich food algae with single fatty acids. Limnol. Oceanogr., 46: 1764-1773.
Direct Link - Wales, N.A.M., 1988. Hormone studies in Myxine glutinosa: Effects of the eicosanoids arachidonic acid, prostaglandin E1, E2, A2, F2a, thromboxane B2 and of indomethacin on plasma cortisol, blood pressure, urine flow and electrolytr balance. J. Comp. Phy. B Biochemical Systemic Environ. Physiol., 158: 621-626.
CrossRef - Watanabe, T., C. Kitajima and S. Fujita, 1983. Nutritional values of live organisms used in Japan for mass propagation of fish: A review. Aquacult., 34: 115-143.
CrossRef - Weers, P.M.M. and R.D. Gulati, 1997. Effect of the addition of polyunsaturated fatty acids to the diet on the growth and fecundity of Daphnia galeata. Freshwater Biol., 38: 721-729.
CrossRefDirect Link - Metcalfe, L.D. and A.A. Schmitz, 1961. The rapid preparation of fatty acid esters for gas chromatographic analysis. Anal. Chem., 33: 363-364.
CrossRefDirect Link - Sargent, J.R., L.A. McEvoy and J.G. Bell, 1997. Requirements, presentation and sources of polyunsaturated fatty acids in marine fish larval feeds. Aquaculture, 155: 117-127.
CrossRefDirect Link








