Research Article
Temporal and Spatial Expression Pattern of Four Laminin Alpha Chains in Xenopus laevis
Department of Biology, Faculty of Science, University Brunei Darussalam, Jalan Tunku Link, BE1410, Brunei Darussalam
Laminin forms a component of the basement membrane glycoprotein and is composed of three different polypeptide chains, i.e., the α, β and γ chains. Five laminin α, four β and three γ genes have been identified in mammals and these subunits have joined together to form 15 recognized heterotrimers (Miner and Yurchenco, 2004). Laminins have distinct tissue distributions and developmentally regulated expressions and are involved in various biological functions.
Laminin-1 (α1β1γ1), the most commonly studied laminin, has been shown to have a role in the development of the pancreas (Jiang et al., 1999; Crisera et al., 2000; Li et al., 2004). In mouse, the laminin α1-chain distribution was found in the epithelial basement membrane of the urinary tract, the nervous system, the male and female reproductive organs and the eyes (Falk et al., 1999). α1-chain expression was also found along the liver sinusoids formed from cells continuous with the endothelium of the central vein (Ekblom et al., 2003). The role of laminin α2-chains in maintenance and development of muscle was demonstrated when the laminin α2 null mice displayed severely disrupted basement membranes in the skeletal muscle and the peripheral nerve and show severe and progressive muscle degeneration, weakness and atrophy (Colognato and Yurchenco, 2000).
The mRNA of laminin α3-chain was found in the epithelial cells of the developing human bronchioli with the protein present in the basement membrane zones of the developing structures (Miosge et al., 2002). The α4 laminin subunit is expressed in the vascular endothelial basement membranes of certain but not all blood vessels (Tunggal et al., 2000) and has been shown to regulate endothelial cell growth and proliferation (DeHahn et al., 2004). Lama4-null mice showed defects in the branching pattern of blood vessels, distortion of blood vessel architecture and subcutaneous hemorrhages in the tissues of the hind limbs, lower back, head and neck regions (Thyboll et al., 2002).
In humans, mRNA for the laminin-5 (α3β3γ2) α3-chain was found in the cytoplasm of the epithelial cells of the developing tubular structures of the kidney and the laminin 5 protein was found in the various basement membranes zones of the developing glomeruli (Miosge et al., 2002). The identity of the cellular receptors that interact with laminins have also been recognized and studied. These cellular receptors include integrins, dystroglycan, heparin, sulphate proteoglycans, sulfatides, HNK-1 (α1-chain) and lutheran (α5-chain) (Sasaki et al., 2004).
In Xenopus laevis, laminin is first detected in stage 12½ embryos in mesodermal cells in the region of the developing notochord and the developing somites, suggesting its later role in the separation of the organ anlage (Fey and Hausen, 1990). The expression of laminin becomes more intense as development takes place and at stage 17, laminin was detected in the notochord sheet around the dorsal somitogenic mesoderm (Fey andHausen, 1990). At stages 23/24, laminin was detected in regions surrounding individual somites (Fey and Hausen, 1990). The expression of laminin around the somites was also reported by Wedlich et al. (1989), where its expression follows after the expression of fibronectin. In Pleurodeles waltlii, the expression of laminin was first detected in the mid-blastula stage and was associated with the extracellular fibres that underlie the inner ectodermal surface of the blastocoel roof (Darribere et al., 1986).
There is increasing evidence in recent years to suggest the involvement of laminin family of heterotrimers in a wide variety of biological functions such as during tissue organization and cell polarization (Li et al., 2003), the induction and maintenance of cell polarity (Miner and Yurchenco, 2004) and the development of neuromuscular system (Patton et al., 1997).
The present study describes the localisation of four laminin α-chain transcripts during the different developmental stages in Xenopus laevis. Using wholemount in situ hybridisation (Harland, 1991) positive transcripts was found in the domains of the developing neural tube, pronephros, eye vesicle and somite. The expression of the four α-chains indicates a possible role of laminin as signals during the development of these domains.
The EST sequences for laminin α1, α3, α4 and α5 were obtained from GenBank. The accession numbers of the laminin α sequences are α1:BG163032, α3:BG892766, α4:BG438955 and α5: BE576660.
Embryo collection and in situ hybridization
Xenopus embryo collection and culture: Female Xenopus laevis was induced to ovulate by a primary injection of 100 U of follicle stimulation hormone 48 h before laying followed by a second injection of 500 U human chorionic gonadotrophin was given 16 h prior to laying eggs. The eggs were laid in 1X Barth’s X (88 mM NaCl, 1 mM KCl, 2.4 mM NaHCO3, 10 mM Hepes, 0.41 mM CaCl2, 0.33 mM Ca(NO3)2, 0.82 mM MgSO4, pH 7.6). Xenopus laevis embryos ranging from stage 9 to stage 41 were used. Eggs were fertilised in vitro and staged according to the normal table of Xenopus development (Nieuwkoop and Faber, 1967).
Wholemount in-situ hybridisations: Plasmid DNA was linearised with the appropriate restriction enzyme and purified by using a gel extraction kit (Qiagen). 0.5-1 μg of linearised DNA was transcribed from a relevant promoter, T7 and SP6 using the DIG RNA labelling mix (Roche) according to the manufacturer’s instructions. The spatial expression pattern of laminin α-chains was determined by whole-mount in situ hybridization as described by Harland (1991).
Wax embedding and microtome sectioning: Embryos fixed in MEMFA (0.1 M MOPS pH 7.4, 2 mM EGTA, 1 mM MgSO4, 3.7% formaldehyde) were dehydrated with ethanol and cleared with Histoclear washing twice for 10 min each. Embryos were then transferred to histoclear:paraplast (1:1) at 60°C for 30 min. This was replaced with molten paraplast twice for 30 min each and a third change of wax which was left overnight at 60°C. One embryo was then placed in a paraplast mould in a watch glass and allowed to set. The blocks were then sectioned on a Bright microtome at 10 μm thickness and mounted on Superfrost slides and dried overnight. Sections were then cleared in Histoclear, followed by Xylene washes and mounted with Depex.
RT-PCR: Total RNA was extracted from unfertilized eggs up to embryonic stage 41, followed by cDNA synthesis and RT-PCR. The specific primer sequences were determined based on the available sequences in Genbank. The primer sequences for the four laminin α-chains are as follows:
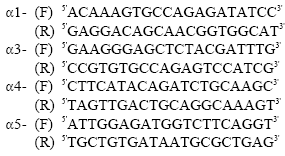 |
The PCR with α1, α3, α4 and α5 chain primers were carried out for 37 cycles at 94°C, 30 sec; 58°C, 30 sec; 72°C, 1 min. Elongation factor 1-α (EF1α) was used as positive control and the PCR reaction was carried out for 27 cycles at 94°C, 30 sec; 55°C, 30 sec; 72°C, 1 min.
Temporal and spatial expression of laminin α-chains: RT-PCR analysis from cDNA obtained from Xenopus laevis eggs and embryos at different stages (stage 6 to 41) shows varying timings of developmental expression of the four different laminin α-chains (Fig. 1). α1-chain mRNA was detected from stage 6 and the levels increase as the embryo develops, up to stage 41. α3-chain mRNA was expressed maternally and zygotic expression does not indicate significant up regulation until about stage 30. α4-chain expression was not observed in the egg.
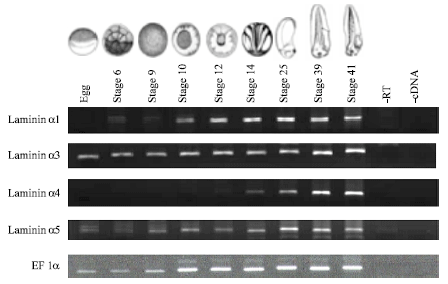 | |
| Fig. 1: | RT-PCR analysis showing the expression pattern of laminin α transcripts in Xenopus laevis unfertilized egg and different embryo stages. Different timings of expression were observed for the different α-chains with α3 and α5 being maternally expressed. EF1α was used as control, -RT and -cDNA negative controls were carried out |
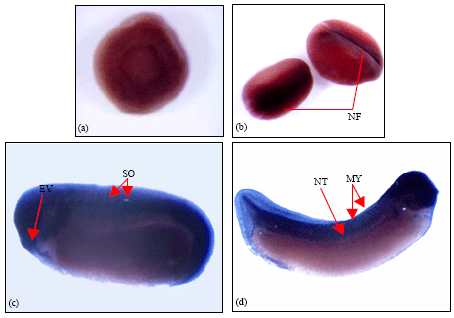 | |
| Fig. 2: | Wholemount in-situ hybridisation of laminin α1-chain. Different embryo stages hybridised with DIG labelled anti-sense probe for laminin α1-chain. (a) A stage 9 blastula, showing faint expression in regions of the marginal zone. (b) Stage 15 embryo where laminin α1-chain transcripts were observed in the neural folds. (c) Stage 25 embryo showing intense expression in regions of the head, developing somites and neural tube. (d) Stage 33 embryo with laminin α1-chain transcripts in the head, mytomes and along the neural tube. NF: Neural Folds; NT: Neural Tube; EV: Eye Vesicle; SO: Somites; MY: Myotomes. The images shown are representative of staining that was observed in approximately 10-15 embryos from three separate experiments performed for each embryo stage |
Low levels of α4-chain mRNA was first detected in the stage 14 embryo and significant increases were observed as the embryo develops up to stage 41. Faint levels of α5-chain mRNA can be detected in the egg with low levels of zygotic expression in stage 6-9.
Spatial expression of laminin α1-chain transcripts in developing Xenopus laevis embryo: Figure 2 shows embryos from the wholemount in-situ hybridization analysis, carried out in order to determine the spatial expression patterns of laminin α1-chain in Xenopus laevis.
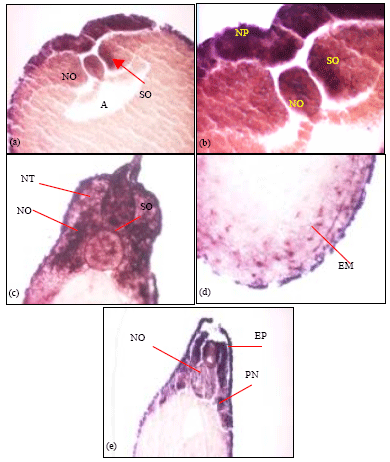 | |
| Fig. 3: | Transverse sections of wholemount in-situ hybridised laminin α1-chain embryos. (a, b) Shows staining in a stage 15 embryo. Regions of transcript expression are within the notochord, the somitogenic mesoderm and the outer epidermis. (c) A stage 25 embryo showing the expression in the neural tube, the somites and within the notochord. (d) α1-chain transcripts were also observed within the endodermal yolk mass of the stage 25 embryo. (e) Stage 33 embryo showing expression of transcripts within specific axons in the neural tube and adjacent somites. Transcripts were also observed in the notochord and the pronephros. NO: Notochord; SO: Somites; EM: Endodermal cell mass, NT: Neural Tube; A: Archenteron; PN: Pronephros. EP: Epidermis, NP: Neural Plate. The staining patterns observed in the transverse sections from the wholemount in-situ embryos were consistent for the embryos sectioned (n = 3) for each of stage 15, stage 25 and stage 33 |
Faint expression was observed in the stage 9 blastula in the region of the marginal zone (Fig. 2a). By stage 15, expression was seen along the neural folds and in the anterior neural plate region which comprises of the anlage that will later form the prospective brain region (Fig. 2b). By stage 25, expression was observed in the head region, eye vesicles, the cement gland and the somites (Fig. 2c). At stage 33, staining was intense in the head region, eye vesicle, along the myotomes and the neural tube (Fig. 2d).
To observe the expression pattern of laminin α1-chain expression internally, embryos from wholemount in situ’s were sectioned as shown in Fig. 3. At stage 15, staining was observed in the notochord and the somitogenic mesoderm adjacent to the notochord (Fig. 3a). α1-chain transcript was also detected in the neural plate region which appear as a thickened area where the neural folds will later form (Fig. 3b). Transverse sectioning of the stage 25 embryo show transcript expression at the neural tube, the somites, notochord and the outer epidermis (Fig. 3c). Positively stained cells were also observed within the endodermal cell mass (Fig. 3d). At stage 33, expression was seen in outer epidermis, somites, the pronephros and specific areas of cells within the neural tube (Fig. 3e).
Laminin α3-chain expression: RT-PCR data from embryos at different developmental stages showed that α3-chain was maternally expressed and expression was seen at the developmental stages analysed up to stage 41. Wholemount in-situ hybridized embryos from stage 10 to stage 33 are shown in Fig. 4. In the stage 10 early gastrula embryo, laminin α3-chain expression was observed at the marginal zone and also where the dorsal blastopore lip and groove was forming (Fig. 4). Figure 4b, shows a stage 13 embryo with the archenteron, blastocoel cavity and the plate dividing the two cavities which were also positive for α3-chain transripts.
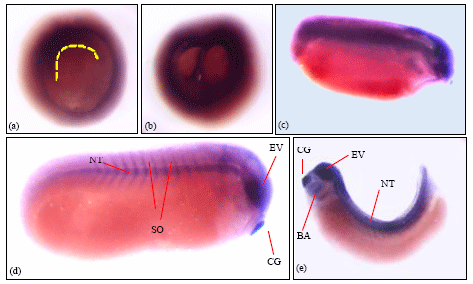 | |
| Fig. 4: | Wholemount in-situ hybridisation with the laminin α3-chain anti-sense probe. (a) Stage 10 early gastrula embryo showing expression of α3-chain transcripts in the forming dorsal blastopore groove (yellow dotted line). (b) Stage 13 embryo showing expression in the plate region between the blastocoel and the archenteron cavity. (c) Stage 20-22 embryo showing expression in the dorsal anterior region along the neural folds. (d) Stage 24-25 embryo with α3-chain expression in neural tube, somites, cement gland and the eye vesicle. (e) Stage 33 embryo showing transcript expression in areas of the neural tube, somites, eye vesicle and in the brachial arches. Approximately 10 embryos were isolated and stained for laminin α3-chain expression. The embryos shown are representative for the staining pattern observed for embryos in stage 10, stage 13, stage 20-22, stage 24-25 and stage 33. Similar results were obtained in one other experiment |
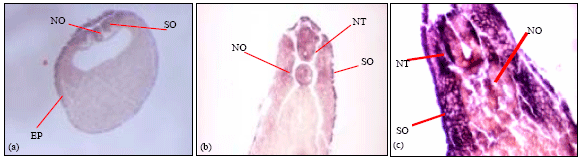 | |
| Fig. 5: | Transverse sections of wholemount in-situ hybridized laminin α3-chain embryos. (a) Staining was observed in the notochord, somite and the outer epidermis of the stage 15 embryo. (b) Stage 25 embryo showing positive transcripts in the neural tube and somites. (c) Stage 33 embryo where the transcripts are expressed in the somites and within a subset of cells in the neural tube. The images show representative staining for three wholemount in-situ embryos sectioned at stages 15, 25 and 33 |
As the embryo develops to stage 20-22 (Fig. 4c), expression was observed in the neural folds and the anterior of the embryo which will give rise to the head region. Figure 4d, shows a cleared stage 28 embryo showing distinct staining in the neural tube, the somites, eye vesicle, cement gland and the brachial arches. The expression in the stage 33 embryo is similar to that seen in the earlier stages where expression was seen in the eye vesicle, brachial arches, neural tube and cement gland (Fig. 4e).
Transverse sectioning of the wholemount in-situ embryos (Fig. 5) with the α3-chain probe shows transcript expression in the notochord and outer epidermis of the stage 15 embryo (Fig. 5a). Staining was also observed in the somitogenic mesoderm adjacent to the notochord. Transverse section of stage 25 embryo (Fig. 5b) shows staining in the neural tube, notochord and the outer epidermis. In the stage 33 embryo, transcript expression was observed in the somites, the outer epidermis and a subset of cells in the neural tube (Fig. 5c).
Laminin α4-chain expression: Laminin α4-chain expression was detected slightly later when compared to the other three α-chains analysed. RT-PCR data (Fig. 1) shows that α4-chain expression was first detected during neurula stage and levels increase as the embryo continue to develop into the tadpole stage. Figure 6 shows whole mount in-situ embryos. Figure 6a, a cleared neurula stage embryo showing staining in the neural folds and the anterior part of the neural plate.
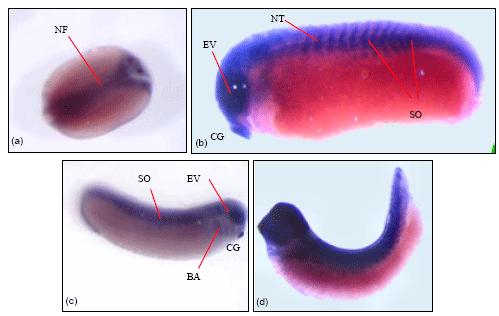 | |
| Fig. 6: | Wholemount in-situ hybridisation with laminin α4-chain anti-sense probe (a) Dorsal view of a stage 16 embryo showing distinct α4-chain expression in the neural folds. (b and c) Stage 28 embryo showing similar expression in the somites, neural tube, eye vesicle, cement gland and in the brachial arches of the stage 28 embryo. (d) α4-chain expression seen throughout the dorsal part of the embryo. 10-15 embryos were isolated and analysed for laminin α4-chain and the images above represent the staining pattern for embryos sampled from three experiments |
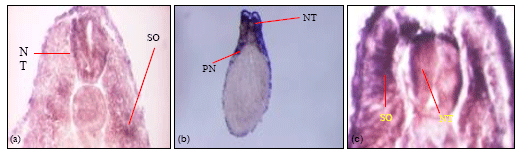 | |
| Fig. 7: | Transverse sections of α4-chain wholemount in-situ embryos. (a) Transverse section of stage 25 embryo showing positive α4 transcripts in the somites and in the neural tube. Similar expression was seen in the stage 33 (b, c) embryo where α4-chain transcripts were seen in the pronephros, somites, outer epidermis and in the neural tube. The staining patterns observed in the transverse sectioning of the wholemount in-situ embryos are representative of 3 embryos sectioned at each stage |
In the stage 24 embryo, staining was observed in the cement gland, the eye vesicle, the developing neural tube and somites (Fig. 6b). In the stage 28 embryo, α4-chain expression was seen in the somites, neural tube, eye vesicle, cement gland and the brachial arches (Fig. 6c). Intense staining was observed for the stage 33 embryo, where staining was seen in the head region and the neural tube and much of the dorsal region of the embryo (Fig. 6d).Transverse sectioning of the laminin α4-chain wholemount in-situ embryos (Fig. 7) was positive in the somites, neural tube and outer epidermis of the stage 25 embryo (Fig. 7a). In the stage 33 embryo staining was observed in specific regions of the neural tube, the somites, the outer epidermis and the pronephros (Fig. 7b, c).
Laminin α5-chain expression: In the RT-PCR data (Fig. 1), α5-chain was maternally expressed and expression continues until the tadpole stage. Wholemount in-situ hybridization on the different embryo stages as shown in Fig. 8, shows expression in the neural folds of neurula stage embryos (Fig. 8a). At stage 25, expression was seen in the somites, the notochord, the eye vesicle and the cement gland (Fig. 8b). In the stage 33 embryo (Fig. 8c), expression of α5-chain was seen in the cement gland, the eye vesicle and the neural tube.
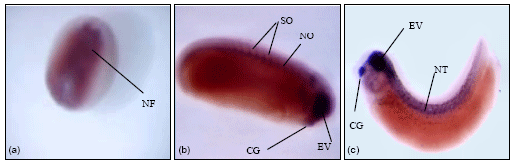 | |
| Fig. 8: | Wholemount in-situ hybridization with laminin α5 probe. (a) Stage 15 embryo, dorsal view showing transcript expression in the neural folds. (b) Expression of transcripts observed in the somites, notochord, cement gland and in the eye vesicle of the stage 25 embryo. (c) Stage 33 embryo with expression observed in the eye vesicle, neural tube and cement gland. 10-15 embryos were isolated from stage 15, stage 25 and stage 33 and the images above show a representative staining for these embryos. Two separate experiments were performed and gave similar results |
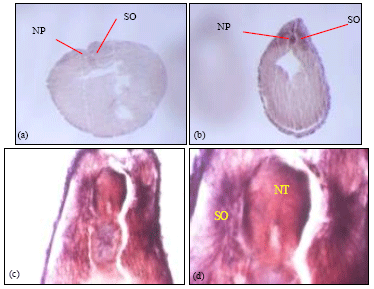 | |
| Fig. 9: | Transverse sectioning of wholemount laminin α5-chain embryos (a) Transverse section of stage 15 neurula showing α5-chain transcript expression in the notochord and at the neural plate. (b) Stage 25 embryo with transcript expressed in the notochord, somites and the neural tube. (c and d), stage 33 embryo with expression in the somites and a subset of cells in the neural tube. The staining patterns observed in the transverse sectioning of the wholemount in-situ embryos are representative of 3 embryos sectioned at the stage sampled |
Figure 9 shows the transverse sectioning of the stage 15 embryo, with faint staining in the neural plate, notochord and in the outer epidermis (Fig. 9a). In the stage 25 embryo, staining was seen in the somite, the neural tube and the outer epidermis (Fig. 9b). Staining was observed in the somites, the neural tube and outer epidermis in the stage 33 embryo (Fig. 9c, d).
The aim of this study was to determine the expression of laminin α-chains during early development of Xenopus laevis. As observed from the wholemount in-situ hybridization patterns, the laminin α-chain transcripts were observed mainly in the developing nervous system, the somites, epidermis, eye vesicle and the pronephros. RT-PCR data shows that the timings of developmental expression of the laminin α-chains analysed relates to the expression of laminin α transcript as seen in the in-situ hybridized embryos. The various laminin α-chains were expressed at different times during the development of the embryo, with some α-chains such as α3 and α5 being maternally expressed. α1-chain expression was observed post fertilization and was also seen in the in-situ blastula embryo with α4-chain expression detected from neurula stage onwards.
Transverse sectioned wholemount embryos at stage 15, showed that for the four laminin α-chains analysed, transcripts were found in the somitogenic mesoderm, notochord and also within the neural plate region (Fig. 3a, b, 5a, 9a). As the embryo develops to stage 25, transcripts was mainly localized in the outer epidermis, the somites, the pronephros and a subset of cells within the neural tube (Fig. 3c, d, 5b, 7a, 8b).
Present findings were similar to earlier studies done using antibodies which were done to determine the expression pattern of laminin in the Xenopus laevis embryo. Using immunohistological analysis on embryo sections, laminin was first detectable in stage 12½ embryos in the region of the mesodermal cells in the region of developing notochord and somites (Fey and Hausen, 1990). The four α-chains analysed, was mainly expressed in the domains of the developing nervous system, the somites, epidermis, eye vesicle and the pronephros suggesting its possible involvement during the development of the various processes.
Laminin α-chain expression during neural tube development: The results obtained shows expression of the four laminin α-chains in the developing neural tube. Laminin α-chain transcripts were detected in the neural plate and expression continued until later stages. The presence of the α-chains during neurulation corresponds to a period of active cell migration. In vertebrates, the notochord is one of the first organs to form which functions as a mechanical support structure and also signaling centre patterning adjacent tissues (Pollard et al., 2006). The appearance of laminin in the developing notochord in Xenopus laevis was also previously reported in studies by Fey and Hausen (1990).
All of the four laminin α-chains analysed in this study was present at the neurula stage suggesting their possible involvement during neural development. The involvement of α-chains during development has been reported in earlier studies where laminins have been shown to be important during morphogenetic events that pattern the nervous system (Lallier and DeSimone, 2000). Mice deficient in α5 were shown to have migratory defects in neural crest cell migration (Coles et al., 2006).
Somite and kidney expression of laminin α transcripts: Expression of laminin α-chains in the somite of the developing embryos suggests its possible role in the mechanism of somite formation. Laminin α transcripts were also detected in regions between somites. This finding was similar to the study of Wedlich et al. (1989) where laminin was found in the intersomitic furrows and also over the entire length of the notochord in Xenopus laevis.
The laminin α-chains were also found in the developing kidney as shown in the sections where the laminin α transcripts were detected in the region of the lateral plate mesoderm (Fig. 3e, 7b, 9c). Sorokin et al. (1997) described the differential expression of the five laminin α-chains in the developing and in the adult mouse kidney. Some basement membranes of the kidney expressed several of the laminin α-chains whereas some basement membranes did not express the α-chains (Sorokin et al., 1997).
Widespread expression of laminin α-chains within developing embryo: These results obtained from the temporal and spatial analysis shows that the expression of the laminin α1, α3, α4 and α5 are widespread in the stage 33 embryo and are present in certain domains.
As previously characterized by Fey and Hausen (1990), laminin was localized to the regions of the mesoderm in the developing notochord and somites. As development of the Xenopus laevis embryo progresses, laminin was found in the notochord sheath around the dorsal somitogenic mesoderm at stage 17. This staining became more prominent as the embryo developed to stage 20 and by stage 23/24, individual somites were surrounded by laminin (Fey and Hausen, 1990). Similar expression of laminin in the somites and the notochord in Xenopus laevis was also reported in another study by Wedlich et al. (1989).
The four α-chains analysed, was mainly expressed in the domains of the developing nervous system, the somites, epidermis, eye vesicle and the pronephros suggesting its possible involvement during the development of the various processes. Functional studies can be carried out in order to determine the exact role of these α-chains as candidate signals during developmental processes.
The temporal and spatial expression patterns of four laminin α chains (α1, α3, α4 and α5) during the different developmental stages in Xenopus laevis was determined in this study. Using in-situ hybridization analysis with DIG-labelled antisense probes, the spatial expression of the laminin α-chains was located mainly in the domains of the developing neural tube, notochord, the somites, epidermis, the eye vesicle and in the developing kidney. Its localisation in these regions suggests its possible role during the development of these organ domains.
The researchers wishes to thank Dr. Robert Old (University of Warwick, United Kingdom).