ABSTRACT
Different ratio of substrates combination between sawdust and cattails were used for Pleurotus ostreatus cultivation and 3-6 flushes were obtained from these substrates. A substrate combination between cattails+sawdust or cattails alone was not accelerated the mushroom growing processes. The mycelial completed colonization, primordium initiation and fruiting body formation were delay when compare to sawdust alone (control). Less yields were revealed from all cattails cultivation substrates (112.10-289.63 g) and these yields were significantly different to those found from control (536.85 g) at a confidence level of 95%. Moreover, the percent biological efficiency (%BE) values obtained from all cattails cultivation substrates were nearly two times less than those found in control (95.02%). Even the cattails are undesirable weeds and provides an economically acceptable substrates but less yields, low %BE and more time consuming were obtained in mushroom growing processes therefore the substrate combination of sawdust+cattails or cattails alone have shown low potential for use as a raw material for P. ostreatus cultivation.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jbs.2007.218.221
URL: https://scialert.net/abstract/?doi=jbs.2007.218.221
INTRODUCTION
Cattails (Typha latifolia) are colonial, rhizomatous, perennial plants and can be invasive in the wet pure sand, peat, clay and loamy soils. T. latifolia is in division Magnoliophyta, class Liliopsida, subclass Commelinidae, order Typhales, family Typhaceae. Cattails are edible plant and their roots have medicinal value, high starch and protein content. The young shoots can be cooked like vegetables and pollens are used in baked and roasted products. The composition of cattail consists of 40% fibre, 8.9% moisture content, 63% cellulose, 8.7% hemicellulose, 9.6% lignin, 1.4% wax and 2% ash (http://www.rspg.thaigov.net). The cattail core can be ground into flour and this flour contains approximately 80% carbohydrate and 6-8% protein (Harrington, 1972). Even cattails have been desired as a potential source of foods but in Thailand this plant is an undesirable weeds. Cattails have high rapid growth rate therefore they become monotypic cultures and eliminate other native plant species when hydrology, salinity, or fertility change.
Pleurotus ostreatus (oyster mushroom; OM) is white rot fungi and edible basidiomycete. OM has high protein, rich in mineral contents and medicinal properties (Bano and Rajarathnam, 1988). P. ostreatus cultivation is attractive to the Thai farmers since it has short life cycle, cultivation easily, can decompose and grow well in many agricultural wastes and less demand on resources and technology (Wang et al., 2001; Yildiz et al., 2002; Kalmis and Sargin, 2003). Contamination in OM cultivation substrates is a major problem for the Thai farmers and about 40-50% of P. ostreatus substrates were contaminated by black and green fungi. From previous study, the rate of contamination and wastage of OM cultivation substrates mixed with effective micro-organisms (EM; pH 3.49) were less than those found in not mixed with EM cultivation substrates by a factor of about four (Vetayasuporn, 2004). In this study, EM was used to reduce the contamination in cultivation substrates since one of the main micro-organism found in EM is the group of lactic acid bacteria (Higa, 2001). Therefore, lactic acid produces by EM may help to destroy the impure micro-organisms in the OM substrate.
In the northeast of Thailand, the major agricultural by-product utilised for OM cultivation is sawdust, but this raw material is relatively high cost and not available in this areas. The aim of the study described here was to search for the possible substrate which is readily available for OM cultivation instead of sawdust. Since cattails are undesirable weeds which have no-value and widely found in many wetlands in the northeast of Thailand. Moreover, cattail’s composition contains carbohydrate, cellulose, hemicellulose. Not only digestion of cellulose produces glucose and cellobiose, while digestion of hemicellulose produces mostly xylose, but also hydrolysis of carbohydrate will release many sugars (Albersheim, 1976; Clarke, 1997; Keller, 1993). These released sugars can convert into carbon sources for growth of P. ostreatus. With the characteristic of lignocellulosic materials, cattail was selected for use as a substrate for OM cultivation. In this experiment, the contamination, growth and yields of P. ostreatus were to determine when mixed with 15% EM of cattail substrate has been used alone or supplemented with sawdust and compare their growth to the control substrate (sawdust alone).
MATERIALS AND METHODS
All the mushroom growing processes were carried out between April to August 2006 in the farmer’s mushroom house at Mahasarakham province in the northeast of Thailand. The temperature, relative humidity and ventilation were not controlled.
Preparation of substrate of OM: The composition of the substrate for OM cultivation was as following: lignocellulosic materials (1000 kg); soft rice bran (80 kg); pumice (10 kg); lime (10 kg); gypsum (2 kg) and soaked with EM dissolved in water at concentrations of 15% until suitable of moisture content is gained. The substrate types of lignocellulosic materials were prepared as follows:
| a) | 25% cattails (2 cm long) + 75% sawdust + 15% EM |
| b) | 50% cattails (2 cm long) + 50% sawdust + 15% EM |
| c) | 75% cattails (2 cm long) + 25% sawdust + 15% EM |
| d) | 100% cattails (2 cm long) + 15% EM |
| e) | 100% sawdust + 15% EM (control substrate) |
Each substrate was mixed thoroughly together and left to stand for 7 days in the shade to allow the EM to breakdown the culture medium. After 7 days, each substrate was put in cylindrical plastic bag without adjusting the moisture content. Cotton wool was used to block the entrance to the OM blocks and then they were tightly sealed with paper before the bags were sterilised. One thousand bags of each substrate were used in this study.
Method of OM cultivation: The sterilized OM culture blocks were spawned with pure OM culture using a sterile method. The room was acclimatized at room temperature until the mycelia were widespread. They were then moved to the farmer mushroom house, the block entrance was opened up; the sorghum seeds were pulled out and left for the large mycelia to develop into O.Ms. In laying out the cultivation blocks in the mushroom house, the bags were arranged in a Randomized Complete Block Design (RCBD).
Method of irrigation: Each culture medium block was irrigated using tap water with irrigation being done every morning and evening.
Method of data concerning and harvesting OM
Spawn running data and the percentage contamination of the OM substrates were recorded. Moreover, primordium initiation, fruiting body formation and initial moisture content of each substrate before put in cylindrical plastic bag were determined. Bunches/clusters of OM flowers were harvested by pulling them off from the block and weighted. Harvests were started 1-2 weeks after the first primordial emerged. Harvesting was done until full OM culture medium consumption. At the end of the harvesting period, yield and % BE (Biological efficiency) were calculated. BE is the ratio of kg of fresh mushroom weight per kg dry substrate and counted as a percentage.
Method of Analysis: Analyses was performed to find the percentage contamination of the OM substrates; compare the rate of growth of the mycelia; pin head and fruiting body formation; compare the mean weights and %BE and then data groups were analyzed using SASS for windows 10.0. Treatment means were compared using Ducan’s multiple range test.
RESULTS
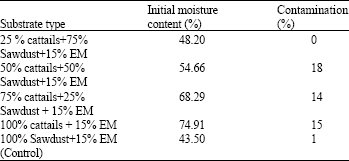
Initial moisture content and contamination of the OM substrates: The substrates initial moisture content obtained in this study were ranged between 43-74% and 100% cattail substrates showed the highest percent of moisture content (74.91%; Table 1). Even a substrate combination of 25 % cattails+75% Sawdust showed no contamination but the remaining cattails substrates showed higher percentage of contamination (14-18%) compared to the control (sawdust; 1%; Table 1). However, growth of spawn running, primordium initiation and fruiting body formation were found in the OM substrates which contaminated with black fungi. In this experiment, the mushrooms yields derived from black fungi contaminated substrates were excluded from the analysis data.
| Table 1: | Initial moisture content and percentage of P. ostreatus contamination |
 | |
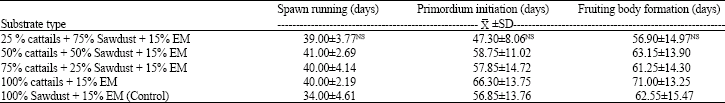
| Table 2: | Time periods of spawn running, primordium initiation and fruiting body formation |
 | |
| NS = Not Significantly different | |
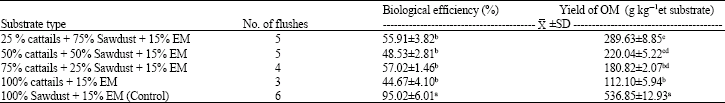
| Table 3: | Comparison of the number of flushes, % Biological efficiency and yield of P. ostreatus |
 | |
| Means±SD in each column with different superscripts indicate statisticant differences (p<0.05) | |
Growth of spawn running, primordium initiation and fruiting body formation: The results found that the growth of the P. ostreatus mycelia in substrate combination of cattails and sawdust were ranged between 39 and 41 days and these period were slower than those found from sawdust (34 days; Table 2). The primordium initiation was formed within 2-3 weeks after spawn running. In all cultivation substrates, the primordium initiation were ranged between 47 and 66 days and 100% cattails showed long length period of primordium initiation (66 days). Similar results were found in fruiting body formation which the lengthen days (71 days) was found in 100% cattails cultivation substrate. However, all mushroom growing periods obtained from all cattail substrates were insignificantly different to those found in sawdust alone at a confidence level of 95% (Table 2).
Comparison of the yield of OM (g kg-1 of wet substrate), number of flushes and%BE: It was found that the cultivation substrate sawdust gave the highest mushroom yield (536.85 g) and it was significantly different to those found from all cattails cultivation substrates (112.10- 289.63 g) at a confidence level of 95% (as shown in Table 3). Three to six mushroom flushes were obtained from the cultivation substrates and higher mushroom flush was found in sawdust (6 flushes) than all cattails substrates (3-5 flushes). In all cultivation substrates, the percentages of biological efficiency were ranged between 44-95% (Table 3). The percentages of biological efficiency obtained from cattails alone (44.67%) was significant differences to those found in sawdust (95.02%) at a confidence level of 95%. The highest percentage of biological efficiency obtained from sawdust was significant differences at a confidence level of 95% to those found in all cattails cultivation substrates.
DISCUSSION
Poor growth, slow colonization of mycelia and long fruiting period may decrease the mushroom yield and number of flushes during the fructification stage. In this study, the cattail substrates provided low mushroom yield than sawdust. A long period for complete mycelia colonization, primordium initiation and fruiting body formation was found in all cattail substrates. These results can be explained by the poor ability of Pleurotus sp. to grow on cattails substrate since thin mycelium was found on cattail substrate. Also, the ligninolytic and other adaptive enzymes of Pleurotus sp. life cycles may not performed effectively (Jennings and Lysek, 1999). Moreover, the low nitrogen content in cattails substrate may affect in the mushroom yield. The results from this experiment are similar to those of Yildiz et al. (2002), who found the yield of P. ostreatus decreased with increasing the ration of leaf of hazelnut into sawdust substrate.
Controlling the initial moisture of P. ostreatus substrate is known to present difficulties, since humidity varies between lots and even within a single lot (Lopez et al., 1996; Contreras et al., 2004). High risk of contamination and differences in fruiting body formation will be obtained in different substrates. Optimal control of initial moisture content of substrate can limit contamination of pure mushroom culture from foreign microorganisms. 100% cattail substrate showed the lowest BE (44.67%) and maximum contamination (15%) might be a result from its structure which contains high initial moisture content (74.91%).
CONCLUSION
In conclusion, it is clearly indicated that even the cattails are undesirable weeds and provides an economically acceptable substrates but less yields, low %BE and more time consuming were obtained in mushroom growing processes therefore the substrate combination of sawdust+cattails or cattails alone have shown low potential for use as a raw material in P. ostreatus cultivation.
REFERENCES
- Bano, Z. and S. Rajarathnam, 1988. Pleurotus mushrooms. Part 2. Chemical composition, nutritional value, post-harvest physiology, preservation and role as human food. discipline of fruits, vegetables and plantation crops, central food technological research institute, Mysore, India. Crit. Rev. Food Sci. Nutr., 27: 87-158.
- Contreras, E.P., M. Sokolov, A. Mejia and J.E. Sanchez, 2004. Soaking of substrate in alkaline water as a pretreatment for the cultivation of Pleurotus ostreatus. Hortic. Sci. Biotechnol., 79: 234-240.
Direct Link - Kalmis, E. and S. Sargin, 2004. Cultivation of two Pleurotus species on wheat straw substrates containing olive mill waste water. Int. Biodeterior. Biodegrad., 53: 43-47.
CrossRefDirect Link - Lopez, A., G. Huerta-Palacios and J.E. Sanchez, 1996. Contamination encountered during various phases of cultivation of pleurotus ostreatus in tropical Mexico. Proceedings of the 2nd International Conference on Mushroom Biology and Mushroom Products, June 9-12, 1996, Pennsylvania State University, USA., pp: 495-502.
- Vetayasuporn, S., 2004. Effective microorganisms for enhancing Pleurotus ostreatus (Fr.) Kummer production. J. Biol. Sci., 4: 694-699.
CrossRefDirect Link - Wang, D., A. Sakoda and M. Suzuki, 2001. Biological efficiency and nutritional value of Pleurotus ostreatus cultivated on spent beer grain. Bioresour. Technol., 78: 293-300.
CrossRefDirect Link - Yildiz, S., U.K. Yildiz, E.D. Gezer and A. Temiz, 2002. Some lignocellulosic wastes used as raw material in cultivation of the Pleurotus ostreatus culture mushroom. Process Biochem., 38: 301-306.
CrossRefDirect Link








