ABSTRACT
In this study Cerebrospinal Fluid (CSF) Hepatocyte Growth Factor (HGF) level was measured, Since it is important in neural cell survival. The study consisted of 35 patients with congenital hydrocephalus and 35 normal children. The author has used western blot and Enzyme-Linked Immunosorbent Assay (ELISA) to study levels of HGF in the CSF. A western blot analysis using anti-HGF antibody confirmed the presence of HGF in the CSF. The level of CSF HGF measured in the hydrocephalic patients was 1005.15±117.84 pg mL-1, which is significantly higher than in normal children (253.45±32.55 pg mL-1). The data from this study indicate that HGF is a constant component of human CSF. It is also suggested that high concentrations of CSF HGF may be partially related to hydrocephalus pathophysiology.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jbs.2007.1249.1253
URL: https://scialert.net/abstract/?doi=jbs.2007.1249.1253
INTRODUCTION
Hydrocephalus is a condition in which there is an abnormal build up of CSF within the ventricles and/or subarachnoid spaces. Human hydrocephalus is a common medical condition that is characterized by abnormalities in the flow or resorption of CSF, resulting in ventricular dilatation. Human hydrocephalus can be classified into two clinical forms, congenital and acquired. It is one of the complex and multifactorial neurological disorders (Jun et al., 2006). Hydrocephalus is far more complicated than a simple disorder of CSF circulation (Czosnyka et al., 2003). It is estimated that about 40% of hydrocephalus cases have a possible genetic etiology. In humans, X-linked hydrocephalus comprises approximately 5-15% of the congenital cases with a genetic cause. Besides genetic factors, many other factors influence the development of congenital hydrocephalus, such as congenital malformations, intracerebral hemorrhage maternal alcohol use, infections and X-ray radiation during pregnancy (Sakata-Haga et al., 2004; Aolad et al., 2003).
The neuropathology of hydrocephalus has been adequately elucidated. Cerebral ventricle dilatation secondary to disturbed CSF has been observed as an inheritable trait in a variety of laboratory animals as well as in humans (Jun et al., 2006).
CSF passes from the lateral ventricles in to the third ventricle via the foramina of Monro and then into the fourth ventricle through the aqueduct of Sylvious. CSF passes out of the brain from the fourth ventricle entering the cisterna magna before passing through the subarachnoid space, circulating around the brain and spinal cord until it exits into the sagittal sinus, facial lymphatics and other unidentified sites through absorptive mechanism including aquaporins and the sodium-potassium ATPase pumps (Miyan et al., 2003).
It is well established that adult CSF has only trace amounts of proteinaceous material. Detectable levels of protein can indicate infection, inflammation, damage, or other pathology in the Central Nervous System (CNS). Coroid plexuses are the main sources of CSF production (Meiniel, 2007) with additional material thought to be derived from the brain parenchyma, specifically the interstitial fluid and major components, in particular large glycoproteins, appearing in the fourth ventricle derived from the subcommisural organ. Our previous study pointed towards a role for CSF in the abnormal cortical development in hydrocephalic texas (H-Tx) rat a widely recognized model for this condition (Mashayekhi et al., 2000, 2001 and 2002).
A number of studies have found important cytokines such as vascular endothelial growth factor (VEGF) (Sampath et al., 2004) transforming growth factor-beta (TGF-β), nerve growth factor (NGF), Brain Derived Neurotrophic Factor (BDNF), neurotrophin-3 (NT-3), insulin-like growth factor (IGF) (Mashayekhi and Salehi, 2007) and HGF (Nayeri et al., 2000) to be present at specific times during development or under specific physiological conditions in CSF. CSF HGF levels increases in the patients with meningitis (Ozden et al., 2004). Changes in the concentration of some of these growth factors can lead to serious consequences of imbalance in CSF production and absorption. Basic-fibroblast growth factor-2 (Johanson et al., 1999) and TGF-β (Moinuddin and Tada, 2000) induce hydrocephalus when elevated in the CSF. Elevated NGF concentrations have been found in the cortices of congenitally hydrocephalic H-Tx rats during progression of hydrocephalus (Hochhaus et al., 2001). It was shown that NGF concentration is elevated in the CSF from congenital communicating hydrocephalus when compared to normal (Mashayekhi and Salehi, 2005).
CSF can be tested for the diagnosis of a variety of neurological disease. In recent years brain specific proteins as markers for structural brain damage have been widely investigated (Beems et al., 2003; Turck et al., 2005). In particular, the growth factors, proteins Neuron Specific Enolase (NSE), Glial Fibillary Acidic Protein (GFAP) and Myelin Basic Protein (MBP) have been widely studied. There seems to be a close relation between the concentrations of these proteins in the CSF and the severity of brain damage. These proteins can also be found at very low levels in serum. Elevated CSF levels of GFAP have been found in normal pressure hydrocephalus (Petzold et al., 2004). Very little is known yet about the value of these brain specific proteins in hydrocephalus.
Hepatocyte Growth Factor (HGF) is a multi functional cytokine that has well-defined mitogenic and morphogenic functions on the epithelial cells (Ozden et al., 2004). HGF is a heterodimeric protein composed of 69 kDa subunit and a 34 kDa subunit linked together by a disulfide bond (Matsumoto and Nakamura, 1991). The various biological effects of HGF are mediated by a tyrosine-kinase receptor, c-Met proto-oncogene (Bottaro et al., 1991). It is an important factor in liver regeneration and plays an important role as a neurotrophic factor in the brain (Honda et al., 1995). Increased level of HGF in the CSF has been seen in Moyamoya disease (Nanba et al., 2004). Intraventricular administration of HGF treats mouse hydrocephalus induced by transforming growth factor beta-1 (Tada et al., 2006). With this background, we have performed this study to determine concentrations of HGF in the CSF from ventricular shunt in the children with congenital hydrocephalus and normal. Since the CSF is in contact with the extracellular space of the brain, biochemical brain modifications could be reflected in the CSF and measurements of intrathecal peptides might identify biomarkers of hydrocephalus.
MATERIALS AND METHODS
This study was conducted in genetics and developmental biology laboratories of the Guilan University, Iran between October 2005 and September 2006. Measurements were performed on 35 CSF aliquots obtained from 35 children with congenital hydrocephalus undergoing surgery and 38 CSF aliquots of normal control children undergoing routine diagnostic lumbar puncture for unrelated reasons. The presence and concentration of HGF in the CSF of normal subjects and congenital hydrocephalic patients was measured by Western blotting and Enzyme-linked immunosorbent assay (ELISA). Samples were aged matched between the two groups analysed and ranged in age between 28-63 months. All affected patients were diagnosed with congenital non-communicating hydrocephalus. None of the patients suffered from known diabetes mellitus, earlier diagnosed tumors of the nervous system or infection. Samples were taken from both male and female patients. For the lumbar puncture the skin were cleaned with 70% alcohol. One milliliter of CSF was collected and used for this study. The samples were centrifuged at 13000 xg for 10 min, the supernatant frozen immediately and stored at -20°C until used. The total number of 35 samples from each group was used for analysis of HGF concentration. Three independent repeats of each analysis were carried on each sample.
HGF analysis
Western blotting: For western blot analysis, aliquots of CSF were mixed with a sample buffer containing 10% SDS, glycerol, 2.8 M β-mercaptoeyhanol and 0.0015% bromophenol blue. Samples were applied to a 10% gradient SDS-PAGE gel (Bio-Rad, Milan, Italy) and the proteins obtained were transferred to nitrocellulose sheets, pore size 0.45 μm (Bio-Rad, Milan, Italy). After incubation for two hours at room temperature in the blocking solution (PBS containing 5% skimmed milk), the nitrocellulose sheets were exposed overnight, at 4°C, to anti-HGF monoclonal antibody (Abcam, Cambridge, UK) and identified with a peroxidase-labeled mouse IgM PK 4010 Vectastain Avidin Biotin complex kit (Vectorlab, Peterborough, UK). The peroxidase activity was revealed with diaminobenzidine (0.5 mg mL-1 in PBS with 0.02% hydrogen peroxide).
ELISA: Immunoreactive HGF was determined by an ELISA, using a commercially available kit (Quantikine HGF immunoassay; R&D Systems, Minneapolis). The CSF samples were centrifuged at 1000 g for 15 min prior to analysis. The method was modified according to the manufacturer’s specifications to determine HGF in CSF. The calibrator consisted of recombinant human HGF with the following concentrations: 0.125, 0.25, 0.5, 1, 2, 4 and 8 ng mL-1. The blank value at 450 nm was subtracted from the standards at the sample values. The lowest detectable amount by this assay was 0.04 ng mL-1.
Statistical analysis: All data presented are expressed as mean±standard error of the mean (SEM). In all experiments, a minimum of 35 measurements were taken in order to calculate a mean±SEM. Statistical analysis was performed using Student’s t-test and only values with p<0.05 were considered as significant.
RESULTS
We analysed CSF from normal subjects and hydrocephalic patients using SDS-PAGE followed by silver staining. A difference on the gel was the presence of a low molecular weight protein in the CSF from hydrocephalic patients, which was very weak in the CSF from the normal samples (Fig. 1A). A western blot analysis using anti-HGF antibody as a probe confirmed the presence of HGF in the CSF (Fig. 1A). An image analyzer was used to determine the intensities of the band in the respective lanes. Relative CSF HGF concentration in normal and hydrocephalic samples was determined.
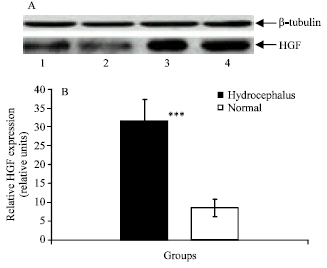 | |
| Fig. 1: | (A), A typical Western blot for HGF protein in CSF from normal control subjects and hydrocephalic patients. Immunoblot analysis Of CSF obtained from normal control subjects (Lanes; 1 and 2) and hydrocephalic patients (Lanes; 3 and 4). Western blots of CSF were probed with antibodies to β-tubulin (loading control) and HGF, as described in material and methods and (B), The relative concentration of HGF in the CSF from normal control subjects and hydrocephalic patients. An image analyzer was used to determine the intensities of the band in the respective lanes. Significance values are shown as stars: 3 stars p<0.001 |
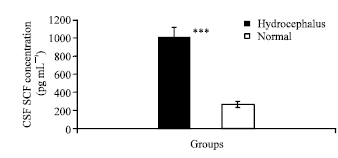 | |
| Fig. 2: | Mean±SEM hepatocyte growth factor concentration (pg mL-1) in normal and hydrocephalic CSF samples. Significant increase in CSF HGF concentration has been seen in hydrocephalic samples when compared with normal. Significance values are shown as stars: 3 stars p<0.001 |
Quantification of western blot gels from repeated experiments showed that the amount of HGF was clearly increased in the hydrocephalic CSF when compared with controls (Fig. 1B). These results show that HGF is present in human CSF. The level of CSF HGF in hydrocephalic patients is more than that in normal CSF (Fig. 2). Using ELISA, it was shown that the level of CSF HGF in the hydrocephalic patients was higher than in normal subjects. The mean±SEM HGF concentration in the CSF of hydrocephalic patients was 1005.15±117.84 pg mL-1, which was significantly higher than the 253.45±32.55 pg mL-1 of the normal subjects (p<0.001) (Fig. 2).
This study has shown that HGF is present in human cerebrospinal fluid. The level of CSF HGF in the patients with congenital hydrocephalus is more than that in normal CSF. Furthermore, since HGF was detected in all samples analyzed in this study, HGF appears to be a constant component of the CSF.
DISCUSSION
HGF and its receptor are expressed in the developing and adult mammalian brain. Recent studies have shown a neurotrophic activity of HGF in the nervous system (Kern et al., 2001). In the present study, we have investigated the concentrations of HGF in the CSF from ventricular shunt in the children with non-communicating hydrocephalus. To our knowledge, data from CSF HGF values in congenital hydrocephalus have not been published before. We investigated HGF as it is one of the most important growth factors and is known to be essential for neural cell survival (Tulasne et al., 2004). It has neurotrophic activities in different types of neurons (Tsuzuki et al., 2000). Changes in CSF HGF are seen in many neurological diseases (Nayeri et al., 2000). We compared ventricular CSF from congenital non-communicating hydrocephalus children at the time of shunt placement with lumbar puncture CSF from normal children, since it is not possible to obtain ventricular CSF from normal. In the present study, CSF HGF levels were found significantly higher in hydrocephalic children than the control group.
HGF promotes the survival and proliferation of several cell types, stimulates migration of different cell types (Gherardi et al., 1993). HGF plays a role in several aspects of neural development (Maina and Klein, 1999). Experiments in chick embryos have suggested that HGF might play a role in neural induction (Streit et al., 1995). Later in embryonic development and in the adult nervous system a variety of neurons and glial cells express HGF and its receptor (Maina et al., 1997). A variety of brain injuries can up-regulate HGF production which plays an important role in neuronal repair. As HGF is known to be involved in the regulation of survival and differentiation of neurons, it may play a role for the recovery of damaged nerve cells in the children with hydrocephalus. HGF is a neuroprotective factor and mediates neurotrophic functions during neurogenesis, that is, promoting survival and/or maturations of CNS neurons in vivo (Maina and Klein, 1999). HGF and its receptor are expressed not only in neurons but also in nonneuronal cells within the nervous system, including microglia and Schwann cells (Di Renzo et al., 1993; Krasnoselsky et al., 1994). In our previous study we have shown that the CSF NGF concentration increases in the children with communicating hydrocephalus when compared to normal (Mashayekhi and Salehi, 2005). The ventricular enlargement in hydrocephalus is a result of primary cortical atrophy, because many neurons in the cerebral cortex are destroyed and replaced with reactive gliosis, the elevation of HGF in CSF is amplified. Production of HGF by glial cells in hydrocephalus represents an active response to neurodegenerative changes. This would explain that why CSF HGF concentration in hydrocephalic children is more than in control group.
Considering the major roles of HGF in the peripheral nervous system, it is possible that a high concentration of HGF in CSF may derive from peripheral circulation. The fact that there is a high penetration rate of injected protein into brain supports this explanation. The comparison between plasma/serum and CSF levels should be included in the future studies to better delineate the site of HGF synthesis.
We conclude that HGF is not only a constant component of human CSF but could also significantly involve in the pathophysiology of hydrocephalus. It is also suggested that the high HGF concentrations that were observed in hydrocephalus might indicate an intrathecal production of this growth factor. This might be useful for hydrocephalic patients.
ACKNOWLEDGMENTS
This study was supported partially by Guilan University, Iran. I am grateful to all postgraduate students in Developmental Biology and Genetics laboratories for their technical assistance. The author thanks Dr. N. Nouri, Shahid Rajaei Hospital, for CSF samples and Dr. M. Kazemi for ELISA.
REFERENCES
- Aolad, H., M. Inouye, W. Darmanto, S. Hayasaka and Y. Murata, 2000. Hydrocephalus in mice following X-irRadiation at early gestational stage: Possibly due to persistent deceleration of cell proliferation. J. Radiat. Res., 41: 213-226.
CrossRef - Beems, T., K.S. Simons, W.J. Van Geel, H.P. De Reus, P.E. Vos and M.M. Verbeek, 2003. Serum- and CSF-concentrations of brain specific proteins in hydrocephalus. Acta Neurochir., 145: 37-43.
Direct Link - Bottaro, D.P., J.S. Rubin, D.L. Faletto, A.M. Chan, T.E. Kmiecik, G.F. Vande Woude and S.A. Aaronson, 1991. Identification of the hepatocyte growth factor receptor as the c-met proto-oncogene product. Science, 15: 802-804.
Direct Link - Czosnyka, M., Z. Czosnyka, E.A. Schmidt and S. Momjian, 2003. Cerebrospinal fluid production. J. Neurosurg., 99: 206-207.
Direct Link - Di Renzo, M.F., A. Bertolotto, M. Olivero, P. Putzolu and T. Crepaldi et al., 1993. Selective expression of the Met/HGF receptor in human central nervous system microglia. Oncogene, 8: 219-222.
Direct Link - Gherardi, E., M. Sharpe, K. Lane, A. Sirulnik and M. Stoker, 1993. Hepatocyte growth factor/scatter factor (HGF/SF), the c-met receptor and the behaviour of epithelial cells. Symp. Soc. Exp. Biol., 47: 163-181.
Direct Link - Hochhaus, F., P. Koehne, C. Schaper, O. Butenandt and U. Felderhoff-Mueser et al., 2001. Elevated nerve growth factor and neurotrophin-3 levels in cerebrospinal fluid of children with hydrocephalus. BMC Pediatr., 1: 2-2.
Direct Link - Honda, S., M. Kagoshima, A. Wanaka, M. Tohyama, K. Matsumoto and T. Nakamura, 1995. Localization and functional coupling of HGF and c-Met/HGF receptor in rat brain: Implication as neurotrophic factor. Mol. Brain Res., 32: 197-210.
Direct Link - Johanson, C.E., J. Szmydynger-Chodobska, A. Chodobski, A. Baird, P. McMillan and E.G. Stopa, 1999. Altered formation and bulk absorption of cerebrospinal fluid in FGF-2-induced hydrocephalus. Am. J. Physiol., 277: R263-R271.
Direct Link - Zhang, J., M.A. Williams and D. Rigamonti, 2006. Genetics of human hydrocephalus. J. Neurol., 253: 1255-1266.
CrossRefDirect Link - Kern, M.A., S. Bamborschke, M. Nekic, D. Schubert, C. Rydin, D. Lindholm and P. Schirmacher, 2001. Concentrations of hepatocyte growth factor in cerebrospinal fluid under normal and different pathological conditions. Cytokine, 7: 170-176.
Direct Link - Krasnoselsky, A., M.J. Massay, M.C. DeFrances, G. Michalopoulos, R. Zarnegar and N. Ratner, 1994. Hepatocyte growth factor is a mitogen for Schwann cells and is present in neurofibromas. J. Neurosci., 14: 7284-7290.
Direct Link - Maina, F., M.C. Hilton, C. Ponzetto, A.M. Davies and R. Klein, 1997. Met receptor signaling is required for sensory nerve development and HGF promotes axonal growth and survival of sensory neurons. Genes Dev., 15: 3341-3350.
Direct Link - Maina, F. and R. Klein, 1999. Hepatocyte growth factor, a versatile signal for developing neurons. Nat. Neurosci., 2: 213-217.
Direct Link - Mashayekhi, F., C.M. Bannister and J.A. Miyan, 2000. Possible role of CSF in a developmental abnormality associated with early-onset hydrocephalus. Eur. J. Pediatr. Surg., 10: 39-40.
Direct Link - Mashayekhi, F., C.M. Bannister and J.A. Miyan, 2001. Failure in cell proliferation in the germinal epithelium of the HTx rats. Eur. J. Pediatr. Surg., 11: 57-59.
Direct Link - Mashayekhi, F., C.E. Draper, C.M. Bannister, M. Pourghasem, P.J. Owen-Lynch and J.A. Miyan, 2002. Deficient cortical development in the hydrocephalic Texas (H-Tx) rat: A role for CSF. Brain, 125: 1859-1874.
Direct Link - Mashayekhi, F. and Z. Salehi, 2005. Expression of nerve growth factor in cerebrospinal fluid of congenital hydrocephalic and normal children. Eur. J. Neurol., 12: 632-637.
Direct Link - Mashayekhi, F. and Z. Salehi, 2007. Infusion of anti-nerve growth factor into the cisternum magnum of chick embryo leads to decrease cell production in the cerebral cortical germinal epithelium. Eur. J. Neurol., 14: 181-186.
Direct Link - Matsumoto, K. and T. Nakamura, 1991. Hepatocyte growth factor: Molecular structure and implications for a central role in liver regeneration. J. Gastroenterol. Hepatol., 6: 509-519.
Direct Link - Meiniel, A., 2007. The secretory ependymal cells of the subcommissural organ: Which role in hydrocephalus? Int. J. Biochem, Cell Biol., 39: 463-468.
Direct Link - Miyan, J., M. Nabiyouni and M. Zendah, 2003. Development of the brain: A vital role for cerebrospinal fluid. Can. J. Physiol. Pharmacol., 81: 317-328.
CrossRef - Miyan, J., M. Nabiyouni and M. Zendah, 2003. Development of the brain: A vital role for cerebrospinal fluid. Can. J. Physiol. Pharmacol., 81: 317-328.
CrossRef - Nanba, R., S. Kuroda, T. Ishikawa, K. Houkin and Y. Iwasaki, 2004. Increased expression of hepatocyte growth factor in cerebrospinal fluid and intracranial artery in moyamoya disease. Stroke, 35: 2837-2842.
Direct Link - Nayeri, F., I. Nilsson, L. Hagberg, L. Brudin, M. Roberg, C. Soderstrom and P. Forsberg, 2000. Hepatocyte growth factor levels in cerebrospinal fluid: A comparison between acute bacterial and nonbacterial meningitis. J. Infect. Dis., 181: 2092-2094.
Direct Link - Ozden, M., A. Kalkan, K. Demirdag, A. Denk and S.S. Kilic, 2004. Hepatocyte Growth Factor (HGF) in patients with hepatitis B and meningitis. J. Infect., 49: 229-235.
Direct Link - Petzold, A., G. Keir, A.J. Green, G. Giovannoni and E.J. Thompson, 2004. An ELISA for glial fibrillary acidic protein. J. Immunol. Methods, 287: 169-177.
Direct Link - Sakata-Haga, H., K. Sawada, T. Ohnishi and Y. Fukui, 2004. Hydrocephalus following prenatal exposure to ethanol. Acta Neuropathol., 108: 393-398.
CrossRefDirect Link - Sampath, P., C.E. Weaver, A. Sungarian, S. Cortez, L. Alderson and E.G. Stopa, 2004. Cerebrospinal fluid (vascular endothelial growth factor) and serologic (recoverin) tumor markers for malignant glioma. Cancer Control, 11: 174-180.
Direct Link - Streit, A., C.D. Stern, C. Thery, G.W. Ireland, S. Aparicio, M.J. Sharpe and E. Gherardi, 1995. A role for HGF/SF in neural induction and its expression in Hensen's node during gastrulation. Development, 121: 813-824.
Direct Link - Tada, T., H. Zhan, Y. Tanaka, K. Hongo, K. Matsumoto and T. Nakamura, 2006. Intraventricular administration of hepatocyte growth factor treats mouse communicating hydrocephalus induced by transforming growth factor beta 1. Neurobiol. Dis., 21: 576-586.
Direct Link - Tsuzuki, N., T. Miyazawa, K. Matsumoto, T. Nakamura, K. Shima and H. Chigasaki, 2000. Hepatocyte growth factor reduces infarct volume after transient focal cerebral ischemia in rats. Acta Neurochir. Suppl., 76: 311-316.
Direct Link - Tulasne, D., J. Deheuninck, F.C. Lourenco, F. Lamballe and Z. Ji et al., 2004. Proapoptotic function of the MET tyrosine kinase receptor through caspase cleavage. Mol. Cell Biol., 24: 10328-10339.
Direct Link - Turck, C.W., G. Maccarrone, E. Sayan-Ayata, A.M. Jacob and C. Ditzen et al., 2005. The quest for brain disorder biomarkers. J. Med. Invest., 52: 231-235.
Direct Link








