ABSTRACT
The present study deals with the effect of Melatonin (MT) against thioacetamide (TAX) induced hepatic damage in rats and to investigate the aging variation in the sequenced responses of injury, regeneration and capacity of recovery. For this purpose three groups of male Wistar rats were used. The first group aging 21 days, the second group aging 12 months and the third group aging 30 months. Thioacetamide treatment induced a significant loss of body weight and an increase in liver weight in all groups which was reversed with melatonin treatment. The histopathological alterations exhibited age-dependent changes related to liver injury and regeneration. Marked dilatation in sinusoids as well as the central and portal veins and necrosis of most hepatocytes were noticed especially in liver sections of adult rats. The cirrhotic features were reduced markedly in the liver of all animals treated with TAX and MT in comparison with rats treated with TAX alone. However, these improvements did not return to the normal liver structure of control group. From this study it can be concluded that melatonin participates in hepatocyte replication after toxin induced liver injury.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jbs.2007.10.18
URL: https://scialert.net/abstract/?doi=jbs.2007.10.18
INTRODUCTION
Thioacetamide (TAX) , which was originally used as a fungicide, is a hepatotoxin commonly used to induce liver cirrhosis in rats and other species (Dashti et al., 1997). The oral intake of TAX caused nodular liver cirrhosis in rats characterized by extensive fibrosis occupying most of the hepatic parenchyma. TAX caused to cell damage and affected the ultrastructure of hepatocytes leading to a decrease in cytoplasmic area together with increase nuclear and nucleolar size (Torres et al., 1998). Clawson et al. (1997) reported that rats were treated with low dose of the hepatocarcinogen TAX showed microscopic foci of hepatic injury. However, TAX has also been reported to cause chemically induced cell death via both apoptosis and necrosis (Witzmann et al., 1996). Following a single low-dose administration of TAX produces apparently permanent alterations in a suppopulation of hepatocyte nuclei, which encompass increased nuclear size (Brini et al., 1993), DNA content (Clawson et al., 1992) and enzymatic activities (Clawson et al., 1984).
The slowly developing cirrhosis induced by TAX has been proven to be morphologically well defined and uniform. It also appears to reflect the major features of human disease (Zimmermann et al., 1987). Until now, observations in experimental animals were not convincing with regard to the reversibility of fully developed liver cirrhosis. Human experience suggests that reversibility is seen only in very rare cases after the withdrawal of the causative agents (Tamayo, 1983; Zimmermann et al., 1987; Guardigli et al., 2005).
Melatonin (MT), an indole amine product of the pineal gland, was shown to be an endogenous hydroxyl radical (OH) scavenger and effective antioxidant (Tan et al., 1993; Poeggeler et al., 1994) with its scavenging ability (Pieri et al., 1994) as well as somewhat hydrophilic (Shida et al., 1994). Melatonin easily passes all known morphophysiological barriers and enter all subcellular compartments. It protect cells, tissues and organs from oxidative damage induced by a variety of free radical generating agents and processes (Reiter, 1998; Maestroni et al., 2001). MT as an antioxidant is effective in protecting membrane lipids, nuclear DNA and protein from oxidative damage both in vivo and in vitro (Reiter et al., 1998a, b; Lusardi et al., 2000; Atkinson et al., 2003).
Melatonin may exert certain biologic effects such as the inhibition of tumor growth and counteraction of stress-induced immunodepression by augmenting the immune response (Maestroni, 1993; Drijfhout et al., 1996).
Little histological studies deals with the effect of melatonin on cirrhotic liver have been reported. Therefore, the aim of the present study is to determine whether administration of melatonin will have any improvements on rat's liver injury in the different ages induced by thioacetamide.
MATERIALS AND METHODS
Animal groups: Male Wistar rats aged 21 days-newly weaned- (80-100 g), 12 month-adult- (450-500 g) and 30 months-old- were obtained from animal house of king Abdel Aziz University. The animals were allowed to acclimatize for 2 weeks before experimentation. Excess food and water were allowed ad libitum. The animals housed in good aerated chambers with 12 h light and dark cycle. The animals were arranged into three groups (15 rats/each) as follows:
Group 1: Animals aging 21 days
Group 2: Animals aging 12 month
Group 3: Animals aging 30 month
Each group divided into three subgroups (5 rats/each) as follows:
Group 1:Control group.
Group 2:Thioacetamide treated group.
Group 3:Combined treatments of thioacetamide and melatonin.
Drug and applied dose-treatment: Thioacetamide was intraperitoneal (ip) injected in a single high dose (300 mg/kg/body weight) freshly dissolved in 0.9 NaCl. Melatonin, (TLC) was purchased from Sigma Company, USA. It was daily injected orally at the dose of 5 mg/kg/body weight as recommended by Laurido et al. (2002) for 4 weeks.
At the end of treatment, the animals of control and treated groups were sacrificed and the liver was separated. Small pieces were fixed in 10% formol saline, dehydrated in ascending grades of ethyl alcohol, cleared in xylol and mounted in molten paraplast at 56-60°C and cut at 5 μm on rotary microtome. The paraffin sections were stained with haematoxylin and eosin for histological studies and examined under light Leitz microscope.
RESULTS
Body and relative liver weight: Animal treated with thioacetamide showed a significant loss of body weight which was regained with melatonin treatment (Fig. 1). The enlargement of the liver in all treated animals were noticed visually and confirmed by a gain in liver weight of thioacetamide treated animals. These changes were also reversed by melatonin treatment (Fig. 2) .
Histological examination of liver of control rats: The liver of control rats of all experimental groups examined by light microscope revealed the same normal hepatic structure.
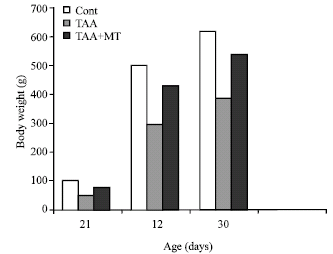 | |
| Fig. 1: | Effect of aging on body weight in melatonin and thioacetamide treated rats. A significant decrease at p<0.01 was noted in TAX treatment compare to control rats |
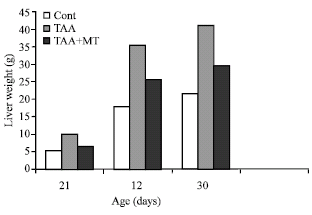 | |
| Fig. 2: | Effect of aging on liver weight in melatonin and thioacetamide treated rats. A significant increase at p<0.01 was noted in TAX treatment compare to control rats |
The structural unit of the liver is the hepatic lobule, which is made up of radiating plates, cords or strands of cells forming a network around a central vein (Fig. 3). The liver strands are alternating with narrow sinusoids. These sinusoids have irregular boundaries composed of only a single layer of fenestrated endothelial cells and large irregularly phagocytic cells which are know as Kupffer cells. The hepatocytes are polyhedral in shape with relatively large size and granular cytoplasm. Each cell has a centrally located nucleus and sometimes, binucleate hepatocytes were observed. Outside the hepatic lobules, at certain angles, lay the portals areas of normal distribution of the collagen fibers and contained branches of portal vein, hepatic artery and bile duct.
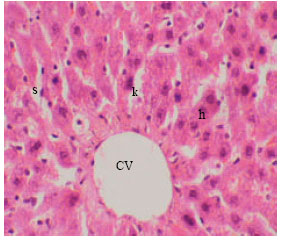 | |
| Fig. 3: | Section in the liver of control rat showing hepatic cells (h), sinusoidal spaces (s) with Kupffer cells (k) and central vein (CV) (X125) |
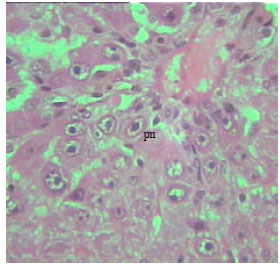 | |
| Fig. 4: | Section in the liver of 12 months old rat treated with TAX showing loss of the normal architecture of the liver with marked cytoplasmic vacuolation. Most of nuclei were pyknotic (pr) (X250) |
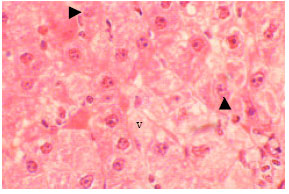 | |
| Fig. 5: | Section in the liver of 21 days old TAX treated rat, showing loss of the normal architecture of the liver. Hepatocytes showed marked cytoplasmic vacuolation (v), karyolysis in some nuclei (arrow). (X250) |
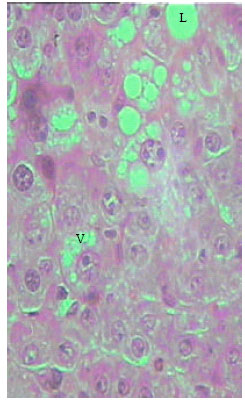 | |
| Fig. 6: | Section in the liver of rat 12 months old treated with TAX. Disorganization of the hepatic strands is clearly visible. Severe necrosis appears in most cells, cytoplasmic vacuolation (v), Lipids droplets (L) (X250) |
 | |
| Fig. 7: | Section in the liver of rat 30 months old treated with TAX, Disorganization of the hepatic strands is clearly detected. Marked cytoplasmic vacuolations (v), dilated sinusoids (s). Necrosis of some hepatocytes (X250) |
Histological examination of liver of TAX treated rats: Liver sections obtained from 21 days, 12 and 30 months old rats following thioacetamide intoxication showed age-dependent changes related to liver injury. One of the symptoms of hepatic injury is loss of the normal architecture of the liver with marked cytoplasmic vacuolation (Fig. 4). The sinusoids between the hepatic cells, the central and portal veins were markedly dilated. Obvious necrosis of most hepatocytes could be noticed especially in liver sections of adult rats. The severity of the necrosis was lower in the old rats compare (Fig. 5-7).
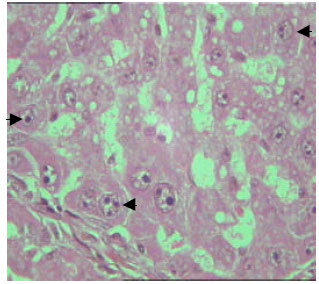 | |
| Fig. 8: | Section in the liver of rat 12 months old treated with TAX. Some hepatocytes contained enlarged nuclei which encompass increased nuclear size and showed intense nuclear staining. The nuclear envelope appeared irregular in some hepatocytes (arrow) (X125) |
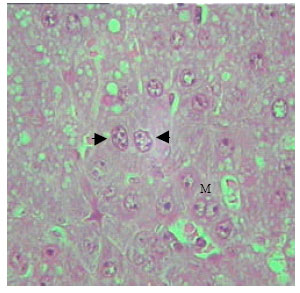 | |
| Fig. 9: | Section in the liver of rat 21 days old treated with TAX. The enlarged hepatocytes nuclei showed intranuclear inclusions, irregularity of nuclear envelope (arrow). Mitotic figures (M) (X250) |
The dispersion of nuclear and nuclear chromatin was observed in most nuclei. In some hepatic cells, the nuclear envelope appeared irregular (Fig. 8 and 9). Vast majority of enlarged hepatocyte nuclei showed intranuclear inclusions almost in all observed hepatocytes of TAX treated groups (Fig. 10). Only few mitotic cells could be observed in liver sections of all control animal groups. However, cells in mitosis were detected in all sections of liver obtained from newly weaned and adult rats (Fig. 9, 10) and was slightly detectable in liver sections of old rats.
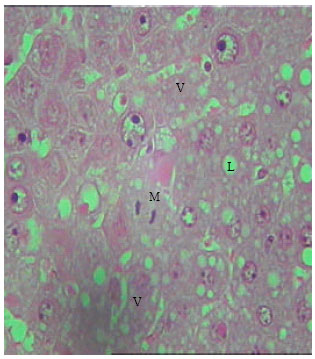 | |
| Fig. 10: | Section in the liver of rat 12 months old treated with TAX, showing cytoplasmic vacuolations (v), Lipids droplets (L). Mitotic figures (M) (X250) |
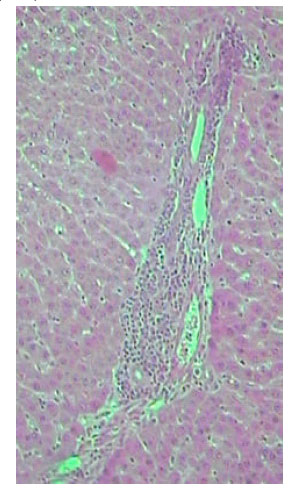 | |
| Fig. 11: | Section in the liver of rat 12 month old treated with TAX, showing central region with small hepatocytes admixed with macrophages (X250) |
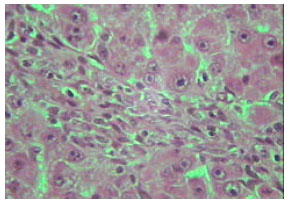 | |
| Fig. 12: | Section in the liver of rat 21 days old treated with TAA showing marked proliferation of collagen fibers (X250) |
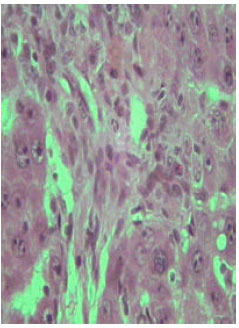
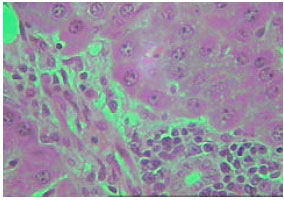 | |
| Fig. 13: | Section in the liver of rat 12 months old treated with TAA showing marked proliferation of collagen fibers (X250) |
 | |
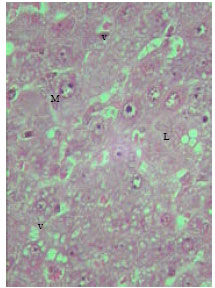
| Fig. 14: | Section in the liver of rat 30 months old treated with TAA showing increase in connective tissue of the cirrhotic liver with accumulation of microphages (X250) |
 | |
| Fig. 15: | Section in the liver of rat 12 months old treated with TAA, intercellular spaces with lipid accumulation (L), cytoplasmic vacuolations (v). Mitotic figures (M) (X250) |
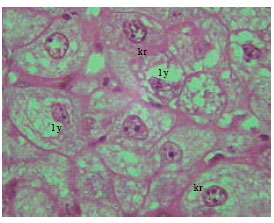 | |
| Fig. 16: | Section in the liver of 12 months old rat treated with TAA. Note different stages of necrosis, karyorehxes (kr), lysis (ly) (X500) |
 | |
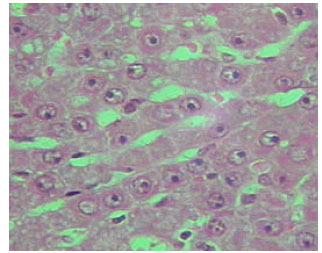
| Fig. 17: | Section in the liver of rat 12 months old treated with TAA, showing acute inflammation in blood vessels (X250) |
 | |
| Fig. 18: | Section in the liver of 21 days old treated rat with TAX+MT, showed less necrosis and less inflammation. (X250) |
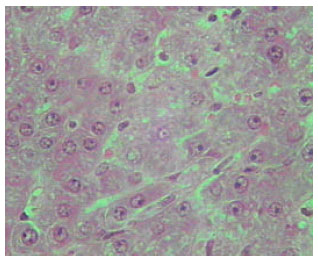 | |
| Fig. 19: | Section in the liver of 12 month old treated rat with TAX+MT, showed less necrosis and less inflammation, reduction in collagen content and fat accumulation (X250) |
Microscopic foci of hepatic injury were observed consisting of central region with small hepatocytes admixed with macrophages. This central region was surrounded by peripheral rim of histologically normal hepatocytes. These peripheral hepatocytes generally contained enlarged nuclei and showed intense nuclear staining (Fig. 11). An increase in connective tissue of the cirrhotic liver was apparent in newly weaned and adult rats as well as old rats liver treated with TAX showing marked proliferation of collagen fibers (Fig. 12-14). Enlarged intercellular spaces with lipid accumulation and few microphages could be obviously observed in liver sections of 12 month aged rats (Fig. 15), the nuclei of most cells revealed clear signs of karyolysis (Fig. 16). Acute inflammation in blood vessels could be detected in sections of adult rats (Fig. 17).
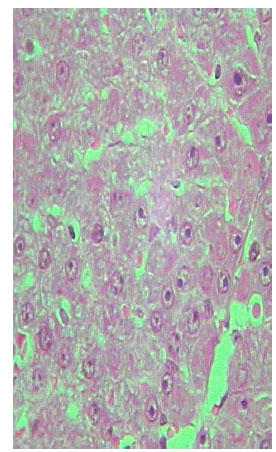 | |
| Fig. 20: | Section in the liver of 30 month old treated rat with TAX+MT, showed some normal hepatocytes, reduction in fat accumulation. Less necrosis (X250) |
Histological examination of liver of TAX and MT treated rats: The histological examination of liver of MT treated groups showed less necrosis and less inflammation. It exhibited some degree of histological regeneration, but the reversed cirrhotic rats were considerably lower in the older animals (Fig. 18-20) .
Sections of liver from MT treated rats of TAX intoxicated groups showed reduction in collagen content and fat accumulation. Also, the level of connective tissue was reduced in these sections. It is worth to mention that these improvements are not completely similar to the normal liver structure of control group (Fig. 18-20). Inhibition of mitotic activity was noticed in liver sections of MT treated animals.
DISCUSSION
Melatonin is a very potent and efficient endogenous free radical scavenger. It reacts with the highly toxic radical and provides one side protection against oxidative damage. Melatonin acts as a primary nonenzymatic antioxidative defense against the devastating action of the extremely reactive free radicals (Abou-El-naga et al., 2002; Abdel Wahab, 1997). It has been proposed that thioacetamide induced hepatic damage which is designated by significant increase in the liver function tests (AST, ALT) and the liver triglyceride concentration (LTG). This damage is due to peroxidation of membrane lipids. It is cleaved to produce reactive free radicals that attack membrane component and/or vital proteins culminating in cell death (Abdel Wahab, 1997; Dashti et al., 1997; Toyokuni et al., 1994). In the present study, i.p. injection of TAX revealed decrease in body weight and increase in liver weights. In agreement, many investigators reported that TAX caused a significant decrease in body weight and increase in liver weight (Al-Bader et al., 1998; Masumi et al., 1999). This decrease in body weight may be through TAX effect on calcium metabolism or by direct action on central nervous system (Hilgier et al., 1999) .The increase in liver weight reflect the morphological enlargement in liver of rats after TAX treatment. Age-dependent changes related to liver injury and regeneration were detected in rats in the present results following intoxication with TAX, which exerted striking nuclear changes assayed on hepatocyte necrosis, enlarged nuclei with intense nuclear staining. Clawson et al. (1997) reported in his study on rats treated with low doses of TAX that such results are indicative of nuclear oxidative damage. Also, similar changes as enlarged hepatocyte nuclei with intranuclear inclusions and decrease in rate of urea production and glutamate dehydrogenase activity in cirrhotic livers were observed on adult rats when compared to old ones were by Huang et al. (1999) and Masumi et al. (1999) after acute treatment of rats with TAX. Sanz et al. (1999) on their study on age-related changes on parameters of experimentally induced liver injury and regeneration in rats following a sub lethal dose of TAX reported that rats aged 30 months recorded the lowest serum tumoral necrosis factor TNF alpha level and necrosis compared to rats aging 2 and 12 months old and that was regard to the delay in the metabolism of thioacetamide in the oldest rats.
The present observations showed that the severity of injury was higher in the adult rats than that in newly weaned and old rats. Similar results were reported by Hilgier et al. (1999) and Sanz et al. (1999) when they study age related changes on liver rats induced injury with TAX. Sanz et al. (1998a) reported that these differences indicate that the lower necrogenic response against the same dose of thioacetamide in newly rats may be due to the lower rate of thioacetamide biotransformation and to the earlier onset of cell division. According, the growing liver from newly weaned rats presents advantages against the necrogenic aggression of thioacetamide, first, because the diminished activity of its specific microsomal detoxification system and second because the earlier increase in the proliferative response prevents the progression of injury permitting an earlier restoration of liver function.
The expansion of metabolic capacity in adult rats results in higher generation of oxidants, developing an increased severity and demanding a higher response against oxidative stress (Rikans et al., 1993). Thioacetamide hepatotoxicity is bioactivation dependent through the microsomal FAD monoxygenase system. Age-dependent changes in the basal FAD-monoxygenase activity, as well as in the induction of this system following thioacetamide, partially explain the decrease liver injury in the youngest and oldest groups because the necrogenic response in all age groups was parallel to previous rise in FAD monoxygenase activity, which was significantly higher in the adult group (Sanz et al., 1999).
Our examination of liver rats treated with TAX showed nodular cirrhosis with loss of the normal architecture of liver, proliferation of collagen and enlarged nuclei with intranuclear inclusions. Similar observations were reported by Fernandes et al., 1996; AL-Bader et al., 1998; Huang et al., 1999). Also, the present observations revealed that, mitotic cells were very few in the old rat group compared to the other two groups, which may be due to that the old rats diminished ability of hepatocytes to regenerate. (Sanz et al., 1998b) summarized the lower process of liver injury and regeneration in old rats due to a lower increase in antioxidant enzyme system.
Data in the present study demonstrate that melatonin has beneficial effect on liver cirrhosis produced by thioacetamide administration. Similarly, melatonin has been faunal to cause marked decrease in parameters of hepatic damage but did not return them to the normal level (Abdel Wahab, 1997). This protective effect of melatonin may be due to: (1) Its ability to scavenger the free radical induced by thioacetamide. (2) It also functions as an indirect antioxidant by stimulating m (RNA) levels and the activities of superoxide dismutase (Kotler et al., 1998) , glutathione peroxidase and glutathione reductase (Pablose et al., 1998). These enzymes function to reduce OH generation by metabolizing as precursors to non-toxic products. Limson et al. (1998) reported that melatonin and its precursors have a high metal binding affinity.
In addition, melatonin influence the growth of spontaneous and induced tumor in animals and inhibits the proliferation of cultured epithelial breast cancer cell and malignant melanoma cell in dose dependent manner (Molis et al., 1994 ) and reduces hepatic DNA damage of rats exposed to the carcinogen safral (Tan et al., 1994). The mechanism may be the result of down regulation of gene expression (Molis et al., 1994) .
From this study it can be concluded that melatonin participate in hepatocyte replication after toxicity with thioacetamide-induced liver injury .
REFERENCES
- Al-Bader, A.A., T.C. Mathew, D. Al-Mosawi, D. Panigrah and H. Dashti, 1998. Bacterial translocation in thioacetamide induced liver cirrhosis in rats. J. R. Coll. Surg. Edmbrugh., 43: 278-282.
Direct Link - Atkinson, G., B. Drust, T. Relliy and J. Waterhouse, 2003. The relevance of melatonin to sports medicine and science. Sports Med., 33: 809-831.
Direct Link - Brini, A., G. Lee and J. Kinet, 1993. Involvement of Alu sequences in the cell- specific regulation of transcription of the gamma chair of Fe and T cell receptors. J. Biol. Chem., 268: 1355-1361.
Direct Link - Clawson, G.A., C.M. Benedict, M.R. Kelley and J. Weisz, 1977. Focal nuclear hepatocyte response to oxidative damage following law dose thioacetamide intoxication. Carcinogenesis, 18: 1663-1668.
Direct Link - Dashti, H.M., T.C. Mathew, M.M. Jadaon and E. Ashkanani, 1997. Zinc and liver cirrhosis: Biochemical and histopathologic assessment. Nutrition, 13: 206-212.
CrossRefPubMedDirect Link - Drijfhout, W.J., A.G. Van-Der-Linde, J.B. De-Vries, C.J. Grol and B.H. Westerink, 1996. Microdialysis reveals dynamics of coupling between noradrenaline release and melatonin secretion in conscious rats. Neurosci-Lett., 202: 185-188.
CrossRef - Fernandes, I., I. Torres, E. Moreira, L. Fontana, A. Gil and A. Rios, 1996. Influence of administration of long-chain polyunsaturated fatty acids on process of histological recovery in liver cirrhosis produced by oral intake of thioacetamide. Digestive Dis. Sci., 41: 197-207.
Direct Link - Guardigli, M., M. Marangi, S. Casanova, W.F. Grigioni, E. Roda and A. Roda, 2005. Arole for asymmetric dimethylarginine in the pathophysiology of portal billary cirrhosis. Hepatology, 42: 1382-1390.
Direct Link - Hilgier, W., M. Zielinska, H.D. Borkowska, R. Gadamski, M. Walski, S.S. Oja, P. Saransaari and J. Albrechi, 1999. Changes in the extracellular profiles of neuroactive amino acids in the rat striatum at the asymptomatic stage of hepatic failure. J. Neurosci. Res., 56: 76-84.
Direct Link - Huang, Z., J.M. Mato, G. Kanel and S.C. Lui, 1999. Differential effect of thioacetamide on hepatic methionine adenosyltransferase expression in the rat. Hepatology, 29: 1471-1478.
Direct Link - Kotler, M., C. Rodriguez, R.M. Sainz, I. Antolin and A. Menendez-Pelaez, 1998. Melatonin increases gene expression for antioxidant enzymes in rat brain cortex. J. Pineal Res., 24: 83-89.
Direct Link - Laurido, C., T. Pelissie, R. Sotomoyano, L. Valladares, F. Flores and A. Hernandez, 2002. Effect of melatonin on rat spinal cord nociceptive transmission. Neuroreport, 13: 89-91.
Direct Link - Limson, J., T. Nyokong and S. Daya, 1998. The interaction of melatonin and its precursors with aluminium, cadmium, copper, iron, lead and zinc: An adsorptive voltammetric study. J. Pineal Res., 24: 15-21.
Direct Link - Lusardi, P., E. Piazza and R. Fogari, 2000. Cardiovascular effects of melatonin in hypertension controlled by infedipine: A 24 h study. Br. J. Clin. Pharmacol., 49: 423-427.
Direct Link - Masumi, S., M. Moriyama, Kannan, M. Ohta, O. Koshitani, O. Sawamoto and I. Sagano, 1999. Changes in hepatic nitrogen metabolism in isolated perfused liver during the development of thioacetamide-induced cirrhosis in rats. Toxicology., 135: 21-31.
Direct Link - Molis, T.M., L.L. Spriggs and S.M. Hill, 1994. Modulation of estrogen receptor mRNA expression by melatonin in MCF-7 human breast cancer cells. Mol. Endocrinol., 8: 1681-1690.
PubMedDirect Link - Pablose, M.I., R.J. Reiter, G.G. Ortiz, J.M. Guerreto and M.T. Agapito et al., 1998. Rhythms of glutathione peroxidase and glutathione reductase in brain of chick and their inhibition by light. Neurochem. Int., 32: 69-75.
Direct Link - Pieri, C., M. Marra, F. Moroni, R. Recchioni and F. Marcheselli, 1944. Melatonin: A peroxyl radical scavenger more effective than vitamin E. Life Sci., 55: PL271-PL276.
Direct Link - Poeggeler, B., S. Saarela, R.J. Reiter, D.X. Tan, L.D. Chen, L.C. Manchester and L.R. Barlow-Walden, 1994. Melatonin a highly potent endogenous radical scavenger and electron donor: New aspects of the oxidative chemistry of the indole accessed in vitro. Ann. NY Acad. Sci., 738: 419-420.
Direct Link - Reiter, R.J., 1998. Oxidative damage in the central nervous system: Protection by melatonin. Prog. Neurobiol., 56: 359-384.
CrossRefDirect Link - Reiter, R.J., D.X. Tan, B. Poeggeler, A. Menendez-Pelaez, L.D. Chen and S. Saarela, 1994. Melatonin as free radical scavenger implications for aging and age-related decreases. Ann. NY Acad. Sci., 719: 1-12.
Direct Link - Rikans, L.E., Y. Cai, S.D. Kosante and P.S. Venkatarman, 1993. Redoxcyclin and hepatotxicity in aging male Fischer 344 rats. Drug. Metab. Dispo., 21: 605-610.
Direct Link - Sanz, N., C. Diez-Fernandez and M. Cascales, 1998. Aging delays the post-necrotic restoration of liver function. Biofactors, 8: 103-109.
Direct Link - Sanz, N., C. Diez-Fernandez, S. Lourdes-Fernandez, A. Alvarez and M. Cascales, 1998. Necrogenic and regenerative responses of liver of newly weaned rats against a sublethal dose of thioacetamide. Biochem. Biophys. Acta, 1384: 66-78.
CrossRef - Sanz, N., C. Diez-Fernandez, M. Alberto, S. Lourdes- Fernandez and M. Cascales, 1999. Age-related changes on parameters of experimentally-induced liver injury and regeneration. Toxicol. Applied Pharmacol., 154: 40-49.
CrossRef - Shida, C.S., A.M. Castrucci and M.T. Lamy-Freund, 1994. High melatonin solubility in aqueous medium. J. Pineal Res., 16: 198-201.
Direct Link - Tan, D.X., R.J. Reiter, L.D. Chen, B. Poeggeler, L.C. Manchester and L.R. Barlow-Walden, 1994. Both physiological and pharmalogical levels of melatonin reduse DNA adduct formation induced by the carcinogen safrole. Carcinogenesis, 15: 215-218.
Direct Link - Torres, M.I., M.I. Fernandez, I. Gil and A. Rios, 1988. Dietary nucleotides have cytoprotective properties in rat liver damaged by thioacetamide. Life Sci., 62: 13-22.
Direct Link - Toyokuni, S., K. Uchida, K. Okamoto, H. Hiai and E.R. Stadtman, 1994. Formation of 4- hydroxyl-2nonenal-modified proteins in the renal proximal tubules of rat treated with a renal carcinogen ferric nitrilotriacetate. Proc. Natl. Acad. Sci. USA., 91: 2616-2620.
Direct Link - Zimmermann, I., A. Muller, G. Machinik, H. Franke, H. Schubert and R. Dargel, 1987. Biochemical and morphological studies on production and regression of experimental liver cirrhosis induced by thioacetamide in rats. Z. Versuchstierkd, 30: 165-180.
PubMed








