Research Article
The Effect of Fly Ash on Resistance of Hardened Cement Pastes to Sodium Chloride Attack
Department of Physics, Faculty of Science, King Khalid University, P.O. Box 9004, Abha, Saudi Arabia
The chloride ions are one of substances that cause damage of the passive protection of steel reinforcement inside the concrete (Khater, 2010). These chlorides can be found in the concrete from the moment of mixing, or after the use (sources are, sea water or groundwater). And the presence of chlorides-whatever their sources-in the concrete leads to stimulate the process of corrosion. The free chloride ions attack the reinforcing steel and cause it to rust. Mechanical and chemical reactions in this case are highly complex but we can easily say that the chloride ions are able to disrupt the interactions which occur on the surface of reinforcing steel which provides a passive protection. Because the degree of influence of chlorides on the properties of concrete related to its ability to penetrate into the concrete so it is important and necessary to use mineral additives to reduce permeability and increase the resistance of concrete against chlorides attack, which works to improve the performance and extend the life time of concrete structures. The use of pozzolans which reacts with free lime (calcium hydroxide) which results during the hydration of cement and converting it into calcium silicates and aluminates hydrates which working to fill the gaps and the internal capillary pores inside cement matrix (Thomas and Bamforth, 1999; Bijen 1996). Examples of pozzolana materials are silica fume which are characterized by high surface area (about four times the surface area of cement, 20.000 cm2 g-1). Silica fume is a byproduct in the manufacture of alloys of silicon and ferrosilicon alloys, it interacts with calcium hydroxide comprising calcium silicate hydrate which typically deposits in the pore system and improve microstructure. other materials are fly ash as well as blast furnace slag (Lock, 1978). This research aims to study the effect of sodium chloride salts attack on physical properties (such as bulk density-porosity) of cement paste mixed with fly ash in order to determine the effect of fly ash on cement resistance to chlorides.
MATERIALS AND METHOD
The starting materials used in this study were ordinary Portland cement of Blaine surface area ≈3100 cm2 g-1 and fly ash (FA) of Blaine surface area ≈3000 cm2 g-1. Figure 1 shows the SEM micrograph of class F-Fly ash.
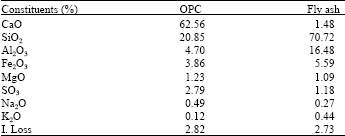
The oxide composition of OPC and fly ash were introduced in Table 1. The blended cement pastes were prepared using ordinary Portland cement that was partially substituted by 35% by weight of cement. The ingredients were homogenized on a roller in a porcelain ball mill with four balls for 1 h to assure complete homogeneity.
| Table 1: | Chemical analysis of the staring materials |
 | |
 | |
| Fig. 1: | SEM micrograph pf fly ash |
The pastes were prepared using the \nwater/cement ratios 0.3. The pastes were molded into 2x2x2 cm cubes. The moulds were vibrated for 1 min to remove any air bubbles. The samples were kept in moulds at 100% relative humidity for 24 h and then it cured under water for 3, 14 and 28 days. The hardened cement pastes were dried at a temperature of 105°C for 24 h in an oven, Then, they were kept in sodium chloride (NaCl) solution of concentration 5000,10000 and 15000 ppm for 3, 14 and 28 days. Each measurement of the physical properties was carried out for three identica cubes of the same mix composition. The bulk density and absorption of prepared samples ware measured according to ASTM C220 (ASTM C 6426-82, 1982).
Determination of density and porosity: The specimens were Immersed in water at room temperature (22°C) for 24 h. The specimens Weighs were recorded while suspended and completely submerged in water. The specimens were Removed from water and water allowed to drain for 1 min by placing specimens on a wire mesh, visible surface water were removed with a damp cloth the saturated weight was determined and recorded as Ws. All specimens were dried in a ventilated oven at 105°C for not less than 24 h and two successive weightings were record at intervals of 2 h until an increment of not greater than 0.2% of the last previously determined weight of the specimen. The weight of dried specimens were Recorded as Wd (oven-dry weight).The absorption and oven-dry density were calculated as:
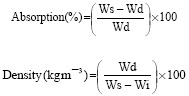 |
where, Wi, Ws, Wd are in kg.
The loss of weight (Lw ) is defined as:
where, Ig is the weight after ignition (g).
The combined water (Wn) is defined as:
where, W' is the weight after ignition for one hour (g) at 1000°C then coaled in a desiccators and weighted again. L ; is the ignition loss of unhydrated specimen defines as:
The physical properties of cement pastes subjected to different concentrations of NaCl solution were studied to clarify its physical performance affected during sodium chloride exposure. The results of bulk density of the hardened cement pastes, which exposed to different concentrations of NaCl in absence and presence of FA are shown in Fig. 2a-b. In general, the bulk density increases gradually in the increasing, the hydration age if the cement pastes. This is due to the decreasing in the total porosity with hydration aga as a result of the filling of pores by the hydration products. The result of Fig. 2a indicate that the bulk density of the hardened cement pastes increases at concentration ≈5000 ppm of NaCl, then decreases gradually with increasing of NaCl. The bulk density of blended cement pastes containing 35% FA increases as the concentration of sodium chloride increases. The increase of the bulk density is mainly due to the activation of blended cement with chloride ions in the samples forming hydrated products which fill some open pores in the samples (Monfore, 1968). Figure 3a,b show the variation in the mass loss versus the concentration of NaCl at different curing ages.
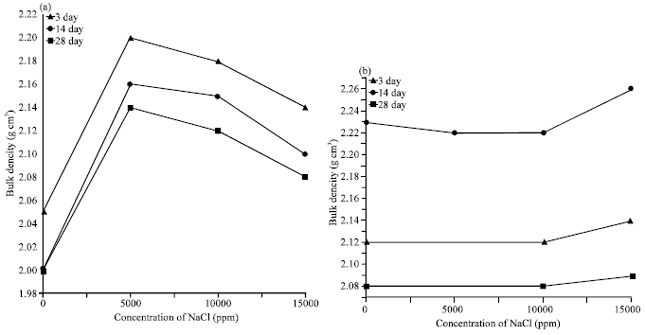 | |
| Fig. 2(a-b): | Variations of bulk density of the hardened cement pastes with the concentration of NaCl at different curing times (a) Plain cement paste and (b) Fly ash-Cement paste |
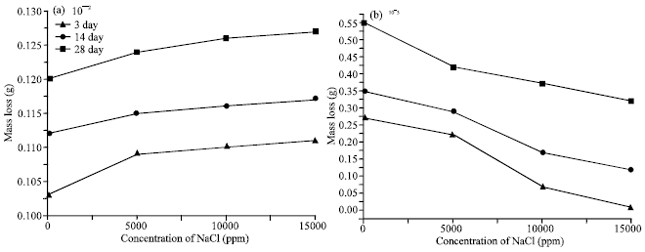 | |
| Fig. 3(a-b): | Variations of mass loss of the hardened cement pastes with the concentration of NaCl at different curing times (a) Plain cement paste and (b) Fly ash-Cement paste |
It is clear that the rate of mass loss decreases with increasing the curing age due to the precipitation of hydrated phases in to the available pores of cement pastes but in Fig. 3a, the rate of the of mass loss increases with the concentration of NaCl in the samples, the larger amount of mass loss is caused by surface scaling.
In Fig. 3b the rate of the mass loss decreases with increasing of NaCl in the samples that is due to the FA reacts with the liberated lime from the hydration of clinker, hence the free lime is nearly consumed. So there is no chance to reaction of lime with chloride group. The total porosity of the blended cement pastes cured for 3,14 and 28 days are graphically represented as a function of the concentration of NaCl in the samples in Fig. 4a and b. It is clear that the total porosity decreases with curing time for all hardened cement pastes. This is due to the filling up of a part of the available pore volume with the hydration products, as the hydration proceeds (El-Didamony et al., 1978). In Fig. 4a the total porosity decreasing up to 5000 ppm, then increasing with the concentration of NaCl in the samples. This variation mainly depending on the rate of the geometrical random arrangement of the material hydrated phases during the hydration mechanism inside the matrix of the sample (Seleem et al., 2010).
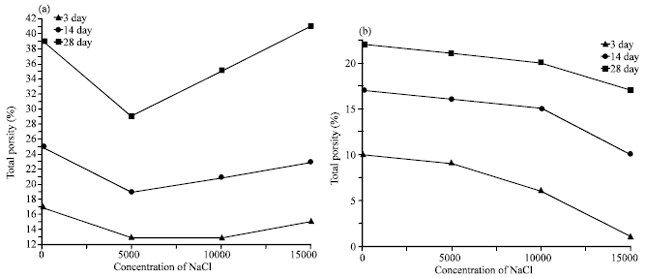 | |
| Fig. 4(a-b): | Variations of total porosity of the hardened cement pastes with the concentration of NaCl at different curing times (a) Plain cement paste and (b) Fly ash-Cement paste |
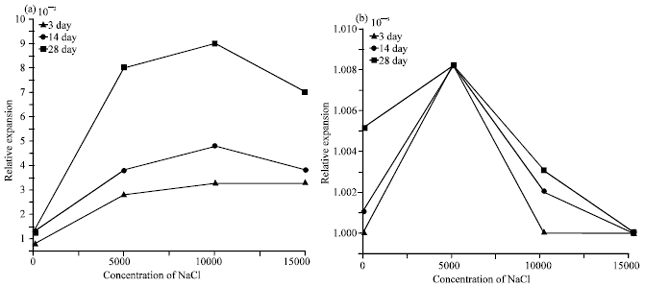 | |
| Fig. 5(a-b): | Variations of relative expansion of the hardened cement pastes with the concentration of NaCl at different curing times (a) Plain cement paste and (b) Fly ash-Cement paste |
The increase in the total porosity values of hardened blended cement pastes after the concentration ≈5000 ppm is related to interaction of chloride with liberated lime from the hydration reaction forming what is known as hydrocalumite (C3A•CaCl2•10H2O) causing softening to the composition with formation of hydration products with a weaker microtexture and a globally more porous cement paste. (Khater, 2010; Bijen, 1996). The variation of the total porosity in Fig. 3b is decreasing with increasing of NaCl in the blended specimens. That is due to the formation of much amounts of (C-S-H) pozzolanic reaction of FA with CH, also the content of FA in the opc matrix to decrease the interaction of the liberated lime (C-H) with the chloride to form hydrocalumite, which increasing the total porosity (Chindaprasirt et al., 2005). The relative expansion of the blended cement pastes as a function in the concentration of the NaCl and at different curing times were represented in Fig. 5a and b. The general behavior of the relative expansion is decreasing with increasing the curing time due to the precipitation of hydrated phases into the pores of the system and that is leading to decreasing in the relative expansion of the samples. In Fig. 5a, the relative expansion increasing with increasing the concentration of NaCl in the sample and that is attributed to the ingress of chloride ions into the cement matrix the through the open pores.
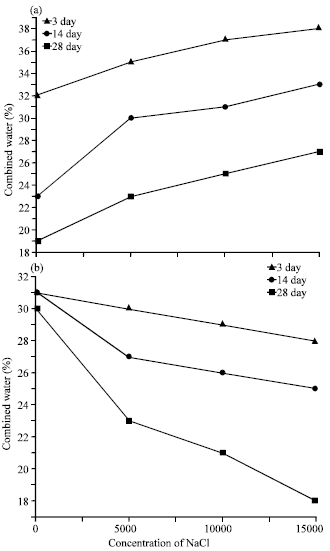 | |
| Fig. 6(a and b): | Variations of relative expansion of the hardened cement pastes with the concentration of NaCl at different curing times (a) Plain cement paste (b) Fly ash-Cement paste |
But in Fig. 5b, the relative expansion decreasing with increasing NaCl in the samples after the concentration ≈5000 ppm and that is due to the random arrangment of the hydrated phases in the cement matrix. The decreasing in the relative expansion with the is mainly due to the increase of the FA hydration, in the cement matrix, chemical combined water for plain and blended cement pastes as a function in the concentration of the NaCl and at different curing times are graphically represented as a function in the concentration of NaCl in Fig. 6a and b. In general in this Fig. 6a and b the chemical combined water increases with the curing time and that is related to the progress of hydration reactions with curing time, that is leading to increase the combined water inside the structure of samples (Monosi and Collepardi, 1989). In Fig. 6a, the combined water increasing with increasing the concentration of NaCl due to the acceleration of hydration reactions caused by chloride ions forming additional amounts of calcium silicate hydrates (C-S-H). But in Fig. 6b, it is clear that the combined water content of the blended cement pastes decreases with the NaCl.
The results are summarized as follows:
| • | The physical properties of cement pastes are affected by the exposure to NaCl solutions |
| • | The rate of the of mass loss increases with the concentration of NaCl in the samples, the larger amount of mass loss is caused by surface scaling |
| • | The use of mineral admixtures (fly ash) increases the ability of cement to withstand aggressive environment and prevent most of the deterioration signs |
| • | The porosity of hardened blended cement pastes increases after the concentration 5000 ppm of NaCl due to interaction of chloride with liberated lime from the hydration reaction forming hydrocalumite (C3A•CaCl2•10H2O) causing softening to the composition with formation of hydration products with a weaker microtexture and a globally more porous cement paste |
| • | The interaction of fly ash with the free lime (CH) during the hydration mechanism leading to form additional (C-S-H) phases and consequently decreasing the formation rate of hydrocalumite which softening the structure of cement pastes |
| • | The chemical combined water increases with the curing time and that is related to the progress of hydration reactions |