Research Article
Physiochemical Performance of Leachate Treatment,A Case Study for Separation Technique
Laboratory of Environmental Technology, Department of Environmental Engineering,Sepuluh Nopember Institute of Technology, Surabaya, Indonesia
INTRODUCTION
Leachate from a biodegradable solid waste landfill is a potentially polluting liquid. Leachate contains significant concentration of substances such as ammoniacal-nitrogen, which is toxic to many organisms. Run-off arising from a landfill containing only soil and rubble may contain suspended solids, be turbid and threaten fish and other aquatic organisms (SEPA, 2003). Unless leachate returned to the environment in a carefully controlled manner, it may cause harmful effects on the groundwater and surface water surrounding a landfill site and therefore leachate treatment has to be provided. Various treatment systems have been investigated in order to remove organic matter and ammoniacal-nitrogen such as constructed wetlands (Nivala et al., 2007), electrochemical oxidation for ammonium (Cabera et al., 2007), periodic biofilter with granular biomass followed by a chemical oxidation step using ozone or Fenton (Iaconi et al., 2006) and combine process consisting of partial nitritation reactor, an anaerobic ammonium oxidation and two underground soil infiltration systems (Liang and Liu, 2007). However, due to the complex nature of landfill leachates, a significant degree of chemicals-containing leachate variation was encountered and influenced the performance of leachate treatment plants (Fan et al., 2006). The variation of leachate quality was irregularly monitored at the largest municipal solid waste disposal site in Surabaya, Indonesia, the so called TPA Benowo which was operated since 2001. An amount of 60-70% of total waste was in the form of degradable materials (Mangkoedihardjo et al., 2007), indicating the leachate produced was characterized by high organic matter content. In addition to landfill, a leachate treatment system was provided, aiming to reduce substantial amount of organic matter and other hazardous substances. The existing leachate treatment process was primarily separation technique, composing in a sequential unit operation of leachate collection pond, flocculation followed by sedimentation, filtration and effluent collection pond. Addition of aluminium sulphate, polyelectrolyte and calcium hydroxide was carried out for flocculation and pH adjustment prior to sedimentation.
This research investigates the performance of separation technique in response to meet the requirement of East Java Government Regulation on Effluent Standard Class III (EJGR45, 2002). Regular monitoring was carried out since May 2005, aiming to evaluate seasonal variability of leachate quality and unit operation efficiency. These would be primary data sources and valuable for the next developing plan of leachate treatment system at the site in addition to control the increasing amount of daily solid waste disposal. The results could indicate which leachate treatment system is suitable in controlling seasonal variability of leachate quality.
Leachate treatment system consists of four units as show in Fig. 1. Leachate collection pond receives liquid which is generated in the solid waste mass as a result of waste decomposition in addition to rain precipitation.
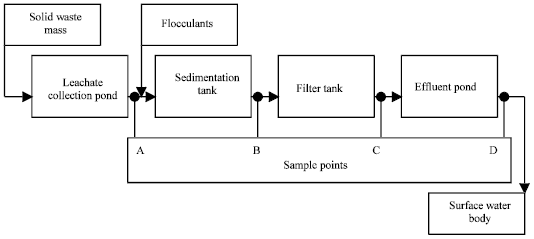 | |
| Fig. 1: | Leachate treatment diagram at TPA Benowo Surabaya |
The pond effluent flows to sedimentation tank following addition of flocculants in order to separate solids. The solid suspension of sedimentation tank is further separated in a sand filter tank and the filter effluent is collected in an effluent pond before discharging to surface water body.
For performance evaluation, the sample points were selected as leachate collection pond (A), sedimentation outlet (B), filter outlet (C) and effluent pond outlet (D). Liquid samples were collected monthly, commenced in May 2005 and completed at the end of April 2006. Dry and wet seasons were commenced in April and September, respectively. Measurements of physiochemical parameters were referred to EJGR45 (2002) on Effluent Standard class III. The standard consists of three physical parameters, i.e., temperature, Total Dissolved Solids (TDS) and Suspended Solids (SS) and twenty-seven chemical parameters, i.e., pH, Ba, Fe, Mn, Cu, Zn, Cr(VI), Cr, Cd, Hg, Pb, As, Sn, Se, Ni, Co, CN, S, F, Cl2, NH4-N, NO3-N, NO2-N, BOD, COD, Anionic Detergent (LAS), Phenol. Their laboratory analyses were carried out in accordance with Standards Methods for the Examination of Water and Wastewater (APHA, 1995).
RESULTS AND DISCUSSION
Six parameters of effluent pond discharging to surface water body (sample point D) exceeded the effluent standard during May 2005 to April 2006. These were Total Dissolved Solids (TDS), Suspended Solids (SS), Sulphide (S), ammonia (NH4-N), BOD and COD. The standard value of each is 4000 mg TDS L-1, 200 mg SS L-1, 0.1 mg S L-1, 5 mg NH4-N L-1, 150 and 300 mg L-1, respectively (EJGR45, 2002). Results of physiochemical parameters for the effluent discharge to water body were reported in Fig. 2, representing the monthly average values. It was found that TDS, SS, S, BOD and COD exceeded the effluent standard, ranging from 2.6 to 4.5 times of the standard. These chemical parameters were reached monthly maximum level in the range of 4.5 to 7.0 times of the standard. The highest concentration of chemical-containing effluent was found for ammonia (NH4-N), which accounted fifty fold on average and two hundred fold on maximum.
The effectiveness of existing treatment system was evaluated based on the six effluent parameters and the six parameters of leachate in collection pond (Fig. 3). It was clearly shown that all parameters at collection pond were higher in various levels than effluent levels. Table 1 show that TDS and ammonia (NH4-N) were reduced to the level of less than 50 %. The facts revealed insufficiency of the existing treatment system to treat the two chemicals.
Further investigation on system performance was addressed to each treatment units. Leachate treatment unit performance was evaluated based on removal efficiency of physiochemical parameters in sedimentation tank, filter and effluent pond. Removal efficiency was defined as the difference of chemical concentration between inlet and outlet over the inlet concentration. Results of the investigation were as follows: (1) TDS was not reduced sufficiently (less than 50%) in each treatment unit; (2) TDS in addition to SS, BOD and COD were characterized low performance of treatment units in wet season while low reduction of sulphide was shown in dry season. These variability might be due to the presence of high level of ammonia, exceeding hundreds fold of effluent standard.
On seasonal performance, leachate collected in pond showed that TDS was frequently higher level in rainy season than in dry season. This might be due to the infiltration pressure of rainfall to solid waste mass, resulting in resuspension of solids in the mass and delivering them to collection pond.
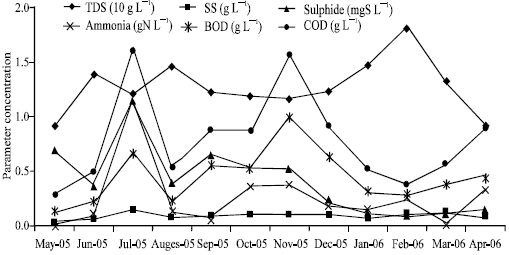 | |
| Fig. 2: | Physiochemical performance of effluent |
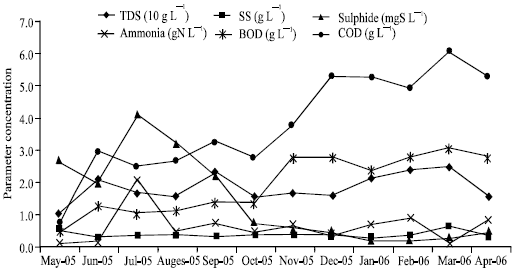 | |
| Fig. 3: | Physiochemical performance of leachate |
The soluble solids contained mostly organic matter as indicated by the higher level of BOD and COD in rainy season than in wet season. The high level of organic matter, deficient of oxygen conditions due to water saturated solid waste mass in wet season and the presence of sulphate (WHO, 2002) might be the reasons of high sulphide accumulation in dry season.
Ammonia fluctuation was shown to be similar with sulphide fluctuation (Fig. 3), i.e., high level was found in dry season. Table 1 shows monthly average ammonia level in leachate was three times of monthly average level in treated leachate. Moreover the treated leachate was fifty fold and two hundred fold exceeded the effluent standard on average and maximum, respectively. These suggest that ammonia was not volatilized in solid waste mass (Erskine, 2000; Sevee et al., 2000), delivering it to leachate collection pond.
| Table 1: | Monthly average physiochemical levels of leachate and treated leachate |
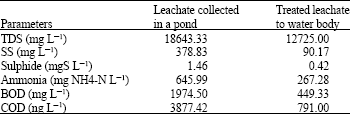 | |
Subsequently, the ammonia of hundreds fold of the effluent standard was affected removal efficiency of treatment system. The presence of ammonia in leachate during a year was similar to other solid waste disposal in many countries which was observed by Nivala et al. (2007), Cabera et al. (2007), Kim et al. (2007) and Isaka et al. (2007). All of these revealed that an improved or a proposed leachate treatment system should be able to treat ammonia variability.
Removal efficiency of SS was significantly higher than TDS in all treatment units. BOD and COD were reduced sufficiently following sedimentation and filtration, suggesting organic matter was mostly in soluble form. Special emphasize was addressed to biodegradability level of leachate which was measured as BOD/COD ratio of more than 0.5. This suggest that the soluble organic matter was biodegradable (Borglin et al., 2004; Mangkoedihardjo, 2006) and hence, microbial treatment could be applied for combination process to remove ammonia (Liang and Liu, 2007).
The results revealed that solids and organic matter content were ranging from three to seven fold of the effluent standard during a year, as well as sulphide in wet season. Physiochemical performance of leachate was characterized by solids and organic matter for wet season and sulphide and ammonia for dry season. Ammonia was hundreds fold of the effluent standard during a year and it might be a parameter that determines leachate treatment performance.
ACKNOWLEDGMENT
The author wishes to thank Dinas Kebersihan Kota Surabaya for funding this research project.