Research Article
Synthesis and Characterization of 3,6-(2-pyridyl)-1,2,4,5-tetrazine Complexes with Gold(III)
Department of Chemistry, Mutah University, Mutah, Jordan
Hanan A. Qaseer
Department of Chemistry, Mutah University, Mutah, Jordan
The 3,6-bis(2-pyridyl)-1,2,4,5-tetrazine (dptz) ligand is a best potential polydentate pyridyl ligand (Kaim, 2002). It has been widely used to form mono and dinuclear transition metal complexes (El-Qisairi, 2007; Yeh et al., 2006; Maekawa et al., 2005; Sarkar et al., 2003; Kaim, 2002; Chellamma and Lieberman, 2001; Glöckle and Kaim, 1999; Klein et al., 1998). In mononuclear complexes, the dptz has two uncoordinated imine nitrogen atoms available for hydrogen bonding (Kaim and Kohlmann, 1987). On the other hand, the dinuclear transition metal complexes has found considerable interest for use as electron propagating components in electron transfer agents, model systems for biological studies and light-capturing antennas via charge transfer transitions (Marshall et al., 2000; Ronco et al., 1998; De Munno et al., 1998). However, dptz may exist in two different conformations as shown in Fig. 1. X-ray diffraction analysis show that the free dptz exist in trnas conformation (Klein et al., 1998).
Gold (III) complexes with N-donor ligands are of great importance for their possible biochemical and pharmacological activities (Kolev et al., 2007; Marcon et al., 2003). The discovery of cisplatin in cancer chemotherapy has led other complexes to be investigated as potential anticancer agents (Kaim and Schwederski, 2001; Cowan, 1997). In particularly, various Au(III) complexes which are of sufficient stability in the physiological environment have been prepared and evaluated for in vitro anticancer properties (Marcon et al., 2003). Recently, reactions of several Au(III) complexes with serum albumin have been reported and discussed (Marcon et al., 2003).
All of these considerations encouraged us to investigate the reactions of dptz with Au(III). In the present study, the preparation and characterization of some new mononuclear complexes of gold(III) with dptz ligand is reported.
Materials: All materials were of reagent grade and were used without further purification. The AuCl3.3H2O was purchased from Aldrich Chemical Co. The dptz ligand was prepared following literature procedure (Geldard and Lions, 1965; Butte and Case, 1961).
Instrumentation: Elemental analyses for the complexes were run by M-H-W Laboratories, Phoenix, Arizona, USA. Conductivity measurements were carried out on a Jeneway 4010 digital conductivity meter at 25°C using 1.0x10–3 M solutions in N,N-dimethylformamide (DMF).
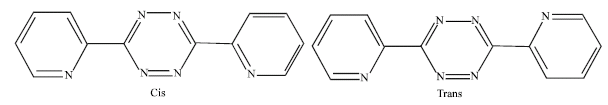 | |
| Fig. 1: | Conformational structures of the free ligand dptz |
IR spectra (KBr pellets) were recorded on a Mattson 5000 FT-IR spectrophotometer. Proton and 13C NMR spectra were determined with a Bruker AC-200 NMR spectrometer in DMSO-d6 using TMS as internal standard. Melting points were obtained using an electrothermal melting point apparatus.
Synthesis of complexes: All of the products were dried under vacuum at 40°C.
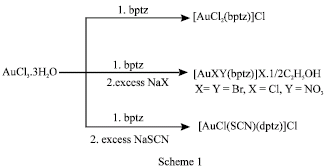
[AuCl2(dptz)]Cl.1/2H2O: To a solution of AuCl3.3H2O (0.08 g, 0.22 mmol) in ethanol (5 mL), a solution of dptz (0.04 g, 0.17 mmol) in ethanol (15 mL) was added. The solution was heated at reflux for 1 h. Then the reaction mixture was allowed stirring at room temperature for overnight. The precipitated formed were collected and washed well with ethanol and diethyl ether.
[AuBr2(dptz)]Br.1/2C2H5OH, [AuCl (NO3) (dptz)] Cl.1/2C2H5OH and [AuCl (SCN) (dptz)] Cl: A solution of dptz (0.04 g, 0.17 mmol) in ethanol (15 mL) was added dropwise to a solution of AuCl3.3H2O (0.08 g, 0.22 mmol) in ethanol (5 mL). Then the solution was treated with an aqueous solution (5 mL) of an appropriate sodium salt (0.44 mmol; NaX, where X = Br¯, NO3¯ and SCN¯). After the solution was heated at reflux for 1 h, the reaction mixture was stirred at room temperature for overnight. During the course of reaction, the product was formed. The product was collected and washed well with water, ethanol and diethyl ether.
Synthesis: The synthetic strategy applied for the synthesis of Au(III) complexes containing the 3,6-bis(2-pyridyl)-1,2,4,5-tetrazine (dptz) ligand is summarized in Scheme 1. The elemental analyses of the new complexes are shown in Table 1. The compounds are colored solid, stable in air and were isolated in fair to good yields (Table 1). They are insoluble in water and common organic solvents while they are soluble in DMF and DMSO.
Conductivity measurements: The ionic or neutral nature of the complexes was checked by measuring their molar conductivity in DMF, in which all compounds proved to be soluble. The molar conductance values for 10–3 M solutions for the complexes (Table 1) are in good agreement with those reported (Angelici, 1977) and confirmed that these complexes behave as 1:1 electrolyte.
IR spectroscopy: The important infrared absorption bands for the dptz ligand and its complexes are presented in Table 2. The bands were assigned by comparison with the reported spectral data for dptz and its complexes (El-Qisairi, 2007; Kaim and Kohlmann, 1987; Jaradat et al., 1986) and other related α-diimine ligands and their complexes (Zaghal et al., 2001, 1999; Nakamoto, 1997; Ghedini et al., 1985). The vibrational spectral data have been used to distinguish the monometallic from the bimetallic complexes (Zaghal et al., 2001, 1999; Nakamoto, 1997; Ghedini et al., 1985).
Infrared spectra of the mono complexes prepared in this study showed the typical features of the coordinated dptz (Table 2).
 |
| Table 1: | Analytical and physical data for the dptz complexes |
 | |
| a: Molar conductance for 10–3 M solutions in DMF at 25°C | |
| Table 2: | Important IR frequencies for dptz and its complexes (KBr pellets; cm–1)a |
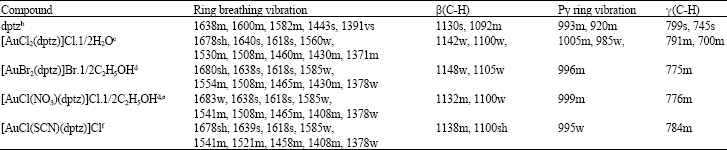 | |
| a: All complexes show v(C-H) for aromatic ring in the regain 3075-3060 cm–1 and band characteristic of coordinated pyridine β(py) in the range 615-625 cm–1, b: El-Qisairi, 2007, c: v(O-H): 3550s, γ(HOH): 1600sh, d: v(O-H) hydrogen bonded ethanol group: 3480sb, e: Coordinated NO3: v(-NO2): 1420m, 1352s; δ(NO): 1000w, f Coordinated SCN: v(SCN): 2105s, s: strong, m: medium, w: weak, sb: strong and broad, sh: shoulder | |
The presence of the band in the range 615-625 cm–1 indicates the coordination of pyridine to the Au(III) (Nakamoto, 1997). Upon complexation, the ring breathing bands are showing appreciable changes (Table 2). They are increased in number as compared to the free dptz ligand. The pyridine-ring vibrations are undergoing increasing in number and/or reduction in intensity. All these changes suggest that upon coordination of Au(III), the ligand dptz is no more symmetric and thus supporting the structure shown in Fig. 2. This result is consistence with what was reported by El-Qisairi (2007). Moreover, based on the fact that most common geometry for Au(III) is four with square planar geometry (Cotton et al., 1999; Greenwood and Earnshaw, 1998) which confirm the proposed structure shown in Fig. 2.
It is to be noted that the bimetallic complexes of related ligands such as 2,2`-bipyrimidine and 3,6-bis(2-pyridyl)pyridazine have been reported to show a decrease in the number of bands in ring breathing with respect to the monometallic complexes (Zaghal et al., 1999; Nakamoto, 1997; Ghedini et al., 1985).
The appearance of strong absorption at 3550 cm–1 (Table 2), together with the other band at 1600 cm–1, supports the presence of lattice water in the complex [AuCl2(dptz)]Cl.1/2H2O (Nakamoto, 1997). The IR spectra of [AuBr2(dptz)]Br.1/2C2H5OH and [AuCl(NO3)(dptz)]Cl.1/2C2H5OH exhibit a strong-broad band at 3480 cm–1 which is characteristic of intermolecular hydrogen bonding of ethanol (Silverstein et al., 2005; Pavia, 2001). In addition, the IR spectrum of [AuCl(NO3)(dptz)]Cl.1/2C2H5OH shows three bands at 1420 m, 1352 s and 1000 w (Table 2) supports the presence of the unidentate NO3¯ rather than the chelating bidentate group (Nakamoto, 1997). The complex [AuCl(SCN)(dptz)]Cl gives a strong band at 2105 cm–1 which confirm the presence of coordinated SCN to Au(III) (Nakamoto, 1997; Zaghal and Ali, 1995).
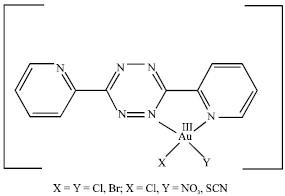 | |
| Fig. 2: | Possible structure of the complexes |
1H- and 13C-NMR: The 1H- and 13C-NMR data are given in Table 3 and 4, respectively. Assignments of signals were made according to previously reported results for dptz and its complexes (El-Qisairi, 2007; Maekawa et al., 2005; Chandra et al., 2002; Kaim and Kohlmann, 1987).
The 1H-NMR spectrum of free dptz ligand displays four different proton resonances for the two equivalent pyridyl groups (El-Qisairi, 2007; Kaim and Kohlmann, 1987). Our complexes give eight different peaks for the two pyridyl groups. This result indicates that these groups are chemically not equivalent upon complexation of dptz to Au(III) and strongly confirms the monometallic rather than bimetallic nature of these complexes. Previous studies (El-Qisairi, 2007; Kaim and Kohlmann, 1987) have reported that the dptz mononuclear complexes showed 8 different proton resonances while the bimetallic ones gave 4 peaks as the free ligand (Kaim and Kohlmann, 1987).
The 13C-NMR spectra for the Au(III) complexes show 12 peaks as expected for the unsymmetrical coordinated ligand while the free symmetrical dptz shows only six peaks. In addition, the 13C-NMR spectrum of [AuCl(SCN)(dptz)]Cl also shows signal at 115.6 ppm, which assigned to carbon atom of the thiocyanato group (Qaseer et al., 2003; Zaghal et al., 1999). The 1H- and 13C-NMR results of the complexes further support the proposed structure shown in Fig. 2.
| Table 3: | 1H NMR chemical shifts for dptz and its complexesa |
 | |
| a: 1H- NMR were obtained in DMSO-d6 with TMS as internal standard, at 200 MHZ. b: El-Qisairi, 2007. d, doublet; dd, doublet of doublet; br, broad; s, singlet; t, triplet; m, multiplet | |
| Table 4: | 13C NMR chemical shifts for dptz and its complexesa |
 | |
| a13:C-NMR were obtained in DMSO-d6 with TMS as internal standard, at 50 MHZ. b: El-Qisairi, 2007 | |
In conclusion, we have successfully synthesized and characterized a new mononuclear gold(III) complexes with dptz of the formula [AuCl2(dptz)] Cl.1/H2O, [AuBr2(dptz)]Br.1/2C2H5OH, [AuCl(NO3) (dptz)] Cl.1/2C2H5OH and [AuCl (SCN) (dptz)] Cl. All data discussed above suggest the proposed structure shown in Fig. 2.
The partial financial support from the Deanship of Research and Graduate Studies at Mu’tah University (Jordan) is greatly appreciated.