ABSTRACT
Glass formation was investigated for the La100-xAl0.412x(Cu,Ni)0.588x(x = 30-56.3) alloys. The optimum Glass Forming Ability (GFA) with full glassy rods of 12 mm in diameter was obtained at an off-eutectic composition La62Al15.7(Cu,Ni)22.3, by copper mold casting, while a full glassy rod of only 1.5 mm in diameter was obtained at the La66Al14(Cu, Ni)20 eutectic alloy. A strong dependence of GFA on the composition was observed for these alloys. It has been found that the GFA does not correlate well with the extent of the supercooled liquid region (ΔTx) and is even not related closely to the reduced glass transition temperature Trg.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jas.2006.202.205
URL: https://scialert.net/abstract/?doi=jas.2006.202.205
INTRODUCTION
Glass formation basically is avoiding detectable crystallization[1-10]. Based on theoretical work on crystal nucleation in undercooled liquid metals, Turnbull[1] proposed that the glass-forming tendency should increase with the reduced glass transition temperature, Trg, which was defined by Tg/Tl. Here, Tg is the glass transition temperature and Tl is the liquidus temperature. Later works of Uhlmann[2] and Davies[3] on the crystallization of undercooled liquids further identified this dimension-less parameter as a crucial figure of merit in determining Glass Forming Ability (GFA). As Tg is generally less dependant on the composition, while Tl has the lowest value at eutectic composition, Trg reaches the maximum at the eutectic composition, i.e., the best glass formation should be located around the eutectic composition[1,4-7]. On the other hand, Inoue[8] and his coworkers found that most of their multi-component bulk glass formers have large extent of supercooled liquid region ΔTx (Δtx = Tx- Tg, Tx is the onset temperature of the first crystallization event). Although this quantity has been generally regarded as the thermal stability of the metallic glasses[3], it has been used successfully in finding many bulk metallic glasses[8]. This study, shows our experimental findings that the composition for the optimum glass formation in the multi-component La based La-Al-(Cu,Ni) pseudo ternary eutectic system is actually at an off-eutectic composition. Furthermore, the GFA in this alloy system does not correlate with the ΔTx and even not well with Trg.
MATERIALS AND METHODS
The master alloys of La100-xAl0.412x(Cu,Ni)0.588x (x = 30-56.3) were prepared by arc melting the constituent elements, La (99.9%), Al (99.9%), Ni (99.98%) and Cu (99.999%) under a Ti-gettered argon atmosphere, in which Cu and Ni always have equal amount. The ingots were cast by pouring the molten alloys into a set of copper molds with diameters of 5 to 12 mm with length up to 60 mm. The samples with diameters less than 5 mm were prepared by suction casting in the copper molds. The resulting samples were sectioned transversely, mounted and polished for observation by Scanning Electron Microscopy (SEM). The degrees of amorphicity of all samples were examined by X-ray Diffractometry (XRD) and Differential Scanning Calorimetry (DSC). For measuring Tg and Tx, ribbon sample was also prepared by a single roller melt-spinner in an argon atmosphere. The onset melting temperature Tm (solidus temperature) and the offset melting temperature Tl (liquidus temperature) were measured by DSC with a heating rate of 40 K min-1.
RESULTS AND DISCUSSION
Figure 1 shows Tl and Tm as a function of Al content in the La100-xAl0.412x(Cu,Ni)0.588x alloys and indicates that the eutectic is at La66Al14(Cu,Ni)20 (x = 34) with eutectic temperature at around 675 K (In the Fig. 1, Tm was used instead of Tl for the eutectic alloy). After locating the eutectic composition, we have studied the GFA of these alloys and its correlation with their Trg and ΔTx.
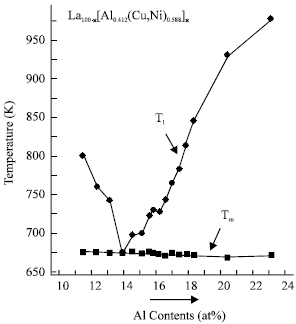 | |
| Fig. 1: | Solidus temperature Tm and liquidus temperature Tl as a function of Al content in the La100-x[(Cu,Ni)0.588Al0.412]x (x = 28-56.3) alloy series |
SEM micrographs of the cross sections of the central parts of the 12 mm diameter cast rods demonstrating a microstructure of a dendritic crystalline phase distributed in an amorphous matrix for the eutectic La66Al14(Cu,Ni)20 alloy (Fig. 2a) and a featureless microstructure for the La62Al15.7(Cu,Ni)22.3 alloy (Fig. 2b) consistent with it being fully amorphous. The insets of Fig. 2 show the corresponding XRD patterns on the sections of 12 mm diameter rods. The inset of Fig. 2a shows a broad diffraction maximum with some crystalline sharp peaks. The dendritic crystalline phase has been identified previously as hcp La[11]. The inset of Fig. 2b only shows a broad diffraction maximum, no crystalline peaks were observed, indicating that the 12 mm diameter rod is full amorphous. Further studies confirm that only 1.5 mm diameter full glassy rods can be formed by copper mold casting for the eutectic alloy La66Al14(Cu,Ni)20 alloy.
DSC curves for the fully amorphous state of the as-spun ribbons showing that the crystallization behavior changed from multiple crystallization events to a single event as the La content decreased and the value of ΔTx increased to a maximum of 76 K (Fig. 3). The values of ΔTx exhibit maximum at about 17 at% Al. The La62Al15.7(Cu,Ni)22.3 alloy with a critical diameter of 12 mm has a ΔTx value of 38 K only (Fig. 3). The critical (limiting) size increased sharply from 1.5 mm at the eutectic composition La66Al14(Cu,Ni)20 to 12 mm for an off-eutectic alloy around La62Al15.7(Cu,Ni)22.3 (x = 38) and then decreased sharply again to 1.5 mm at La56.3Al18(Cu,Ni)25.7 (x = 43.7) (Fig. 3b).
The GFA of La-Al-Cu-Ni-(Co) based alloys has been studied extensively during the past decade[8,12-14].
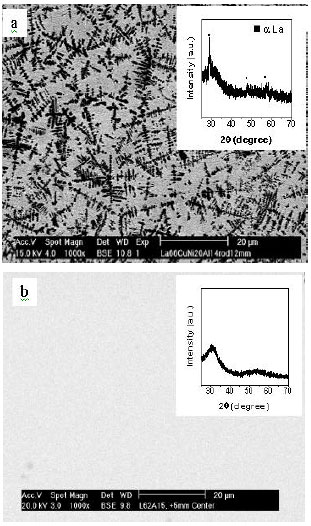 | |
| Fig. 2: | SEM micrographs showing composite of hcp α-La in amorphous matrix (a) and fully amorphous (b) in the center of 12 mm rods of La66(Cu,Ni)20Al14 and La62(Cu,Ni)22.3Al15.7 alloys, respectively. The insets are the XRD patterns on the sections of 12 mm diameter rods |
The best manifestation of GFA for the quaternary La-Al-Cu-Ni alloys was a 7 mm diameter rod of La55Al25Cu10Ni10 alloy obtained by high pressure die-casting and this diameter was further improved to 9 mm in a quinary La55Al25Cu10Ni5Co5[13]. The amorphous La55Al25Cu10Ni10 and La55Al25Cu10Ni5Co5 alloys have values of ΔTx of 88 and 98 K, respectively[13], which are much larger than those for the current alloys. Therefore, within one eutectic system of La-Al-(Cu, Ni) pseudo ternary alloys, large values of ΔTx have no direct correlation with the optimum GFA, similar results were also obtained in the Be containing Zr-based alloys[10].
Furthermore, the correlation between Trg and GFA for the present alloys is also weak. As shown in Fig. 3b, most of Trg are above 0.5.
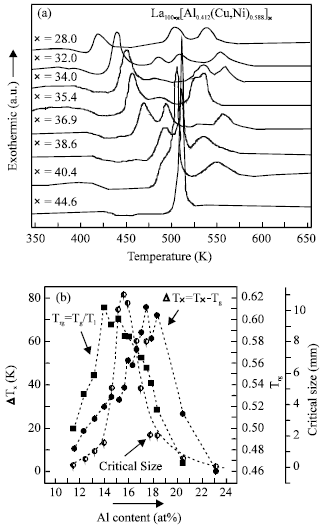 | |
| Fig. 3: | a: DSC curves of La100-x[(Cu,Ni)0.588Al0.412]x (x = 28-56.3) alloys. b: limiting diameter for glass formation as a function of Al content in the La100-x[(Cu,Ni)0.588Al0.412]x (x = 28-56.3) alloy series and Trg and ΔTx as a function of Al content in the alloys |
However, the highest Trg of 0.60 corresponds to the GFA of 1.5 mm for the eutectic alloy, while a slight lower value of Trg 0.58 corresponds to GFA of 12 mm at an off-eutectic composition.
The steady state nucleation rate I can be expressed as[1,5]:
| (1) |
The η can be expressed by a Vogel-Fulcher form[1]:
| (2) |
Substitute Eq. 2 in 1, we can have:
| (3) |
Here, kn is a constant (typically about 1023 N.m), αβ1/3 for metals, b = 16π/3 for spherical nucleus. Tr = T/Tl, ΔTr = 1 -Tr, Tl and T are the equilibrium crystallization (liquidus) and actual absolute temperature, respectively. η0 and A are materials constants. According to Turnbull1, η0 = 10-3.3 Poise (1 Poise = 0.1 Ns m-1) and A = 3.34. From Eq. 3 Turnbull concluded that the higher the Trg is, the higher the glass forming ability is. In particular, liquids with Trg = 2/3 would practically crystallize only within a narrow temperature range, thus they easily could be undercooled to the glass state. However, it is obvious that η0 and A also have strong effect on nucleation rate I and on GFA. The melt viscosities at Tl are very roughly uniform (a few centiPoise) for metals[15]. But it becomes increasingly higher for the glass forming alloys, probably because the alloys become more complex and the melt viscosity at Tl becomes more variable especially at eutectics with extreme depressions of melting temperature[16,17], in much the same way as that of inorganic glass-forming liquids[15,17]. The slopes of the liquidus lines obtained from Fig. 1 are about 30 K/at%, indicating a very sharp depressing melting temperature in our present system. Thus it is possible for an off-eutectic alloy, with a higher melt viscosity to have a better GFA, particularly when their Trg is close to each other like in the present alloy system (0.6 and 0.58). Furthermore, glass formation is also believed to be a competitive process between the formation of glass and the growth of crystals[18] and the eutectic will be different with the thermodynamic eutectic under the higher cooling rate than the equilibrium cooling. Under this consideration, it is also possible that the optimum GFA is located at an off-eutectic composition[19].
CONCLUSIONS
In conclusion, the best GFA in eutectic system of La-Al-(Cu,Ni) pseudo-ternary alloys is at an off-eutectic composition, La62Al15.7(Cu,Ni)22.3 and full glassy rods of 12 mm in diameter were obtained by copper mold casting. The alloy has a small extent of supercooled liquid region. A strong dependence of GFA on the base element in these alloys was observed.
ACKNOWLEDGEMENT
Y. Zhang would like to acknowledge the helpful discussions with Prof Y. Li, Prof. GL Chen and also would like to thank the technical assistance from H. Tan.
REFERENCES
- Ulhmann, D.R., 1972. A kinetic treatment of glass formation. J. Non-Cryst. Solids, 7: 337-348.
CrossRef - Chen, H.S., 1974. Thermodynamic considerations on the formation and stability of metallic glasses. Acta Metall., 22: 1505-1511.
Direct Link - Eckert, J., U. Kuhn, N. Mattern, G. He and A. Gebert, 2002. Structural bulk metallic glasses with different length-scale of constituent phases. Intermetallics, 10: 1183-1190.
CrossRef - Lu, Z.P., H. Tan, Y. Li and S.C. Ng, 2000. The correlation between reduced glass transition temperature and glass forming ability of bulk metallic glasses. Scripta Materialia, 42: 667-673.
Direct Link - Inoue, A., 2000. Stabilization of metallic supercooled liquid and bulk amorphous alloys. Acta Mater., 48: 279-306.
Direct Link - Wang, W.H., Q. Wei and S. Friedrich, 1998. Microstructure and decomposition and crystallization in metallic glass ZrTiCuNiBe alloy. Phys. Rev. Series B, 57: 8211-8217.
Direct Link - Waniuk, T.A., J. Schroers and W.L. Johnson, 2001. Critical cooling rate and thermal stability of Zr-Ti-Cu-Ni-Be alloys. Applied Phys. Lett., 78: 1213-1213.
CrossRef - Tan, H., Y. Zhang, Y.P. Feng and Y. Li, 2002. Synthesis of La based in situ bulk metallic glass matrix composite. Intermetallics, 10: 1203-1205.
CrossRef - Inoue, A., T. Nakamura, T. Sugita, T. Zhang and T. Masumoto, 1993. Bulky La-Al-TM (TM=transition metal) amorphous alloys with high tensile strength produced by a high pressure die casting method. Mater. Trans. JIM, 34: 351-358.
Direct Link - Lu, Z.P., T.T. Goh, Y. Li and S.C. Ng, 1999. Glass formation in La-based La-Al-Ni-Cu-(Co) alloys by Bridgman solidification and their glass forming ability. Acta Mater., 47: 2215-2224.
Direct Link - Ota, R., T. Wakasugi, W. Kawamura, B. Tuchiya and J. Fukunaga, 1995. Glass formation and crystallization in Li2O-Na2O-K2O-SiO2. J. Non-Crystalline Solids, 188: 136-146.
Direct Link - Tan, H., Y. Zhang, D. Ma, Y.P. Feng and Y. Li, 2003. Optimum glass formation at off-eutectic composition and its relation to skewed eutectic coupled zone in the La based La-Al-(Cu, Ni) pseudo ternary system. Acta Mater., 51: 4551-4561.
Direct Link - Akash, B.A., R. Mamlook and M.S. Mohsen, 1999. Multi criteria selection of electric power plants using analytical hierarchy process. Electric Power Syst. Res. J., 52: 29-35.
CrossRef - Khalil, R.A.H., B.A. Jubran and N.M. Faqir, 1997. Optimization of solar pond for electrical power generation system. Energy Convers. Manage., 38: 787-798.
Direct Link - Kolb, G.J., 1998. Economic evaluation of solar only hybrid power towers using molten salt technology. Solar Energy, 62: 51-61.
Direct Link - Kribus, A., R. Zaibel, D. Carey, A. Segal and J. Karni, 1998. A solar driven combined cycle power plant. Solar Energy, 62: 121-129.
Direct Link - Mamlook, R., C. Tao and W.E. Thompson, 1999. An advanced fuzzy controller. Int. J. Fuzzy Sets Syst., 103: 541-545.
CrossRefDirect Link - Mamlook, R., B.A. Akash and M.S. Mohsen, 2001. A neuro fuzzy program approach for evaluating electric power generation systems. Energy, 26: 619-632.
Direct Link - Mamlook, R., B. Akash and S. Nijmeh, 2001. Fuzzy sets programming to perform evaluation of solar systems in Jordan. Energy Convers. Manage., 42: 1717-1726.
CrossRefDirect Link - Mamlook, R. and O.R.A. Jayyousi, 2003. Fuzzy sets analysis for leak detection in infrastructure systems a proposed methodology. Clean Technol. Environ. Policy J., 6: 26-31.
Direct Link - Murakami, M. and A.T. Wolf, 1995. Techno political water and energy development alternatives in the Dead Sea and Aqaba regions. Int. J. Water Res. Dev. 11: 163-184.
CrossRef - Oder, C., H.D. Haasis and O. Rentz, 2007. Analysis of the Lithuanian final energy consumption using fuzzy sets. Int. J. Energy Res., 17: 35-44.
Direct Link - Akash, B.A., O. R. Al-Jayyousi and M.S. Mohsen, 1997. Multi-criteria analysis of non-conventional energy technologies for water desalination in Jordan. Desalination, 114: 1-12.
CrossRefDirect Link - Turnbull, D., 1969. Under what conditions can a glass be formed? Contempt. Phys., 10: 473-488.
CrossRef








