Research Article
Isomerization of M-xylene
Department of Bioengineering, Faculty of Engineering, Ege University, Turkey
Gonul Gunduz
Department Of Chemical Engineering, Faculty Of Engineering, Ege University, Turkey
Xylenes are C8 benzene homologues with the molecular formula C8H10 as seen in Fig. 1. The term mixed xylenes describes a mixture of ethyl benzene and the three xylene isomers, namely p-, m-and o-xylene. They are important chemicals and find wide and various applications in industry. O-xylene is a reactant for the production of phythalic anhydride. M-xylene is used in manufacture of plasticizers, azo dyes, wood preservers, etc. P-xylene upon oxidation yields terephthalic acid used in the manufacture of synthetic textile fibers and poly (p-xylene) which is produced by p-xylene has had economic importance since 1960. Mixed xylenes are largely produced from petroleum such as catalytic reformat, pyrolysis of gasoline, toluene disproportionation and coke-oven light oil. However, as a result of important applications to which the individual xylene isomers are subjected, it is often very important to have high concentrations of a particular xylene.
This can be accomplished by converting a non-equilibrium mixture of the xylene isomers, which is low in the desired xylene isomer, to a mixture which approaches equilibrium concentrations. Numerous catalysts have been proposed for use in xylene isomerization processes. More recently, a number of studies have disclosed the use of crystalline aluminosilicate zeolite containing catalysts for isomerization and conversion of C8 alkylaromatics[1].
Most of the studies in the literature for the isomerization of xylenes are related to the isomerization of mixed-xylenes. Only a few are about a pure material such as isomerization of m-xylene and its kinetics. The reaction of m-xylene isomerization is of great interest for organic chemical industry where the demand for the para and ortho isomers exceeds the supply. The good activity of zeolite catalysts was proved in the gaseous phase isomerization of m-xylene but with respect to kinetics relatively few catalysts were investigated. In spite of the considerable industrial interest on the isomerization and disproportionation of xylenes, especially catalysed by zeolites, no satisfactory correlations have been indicated between catalytic activity and the solid state properties as well. Giordono et al.[2] investigated in particular the interaction of m-xylene with various dealuminated mordenites. The conversion of m-xylene over dealuminated mordenites was found to be a function of the acid strength rather than the number of the acidic sites, as the Si/Al ratio was directly proportional to acid strength. Chatterjee and Ganguli[3] have investigated the isomerization and associated reactions of o-xylene over 20 catalysts based on a large variety of multicationic (mono to trivalent) zeolite Y samples. All the catalysts showed very high selectivity for m-xylene, but relative improvement in p-xylene yield was obtained with catalysts containing La3+ and Ni2+. Isuchiya et al.[4], was calculated that AlBr3/Graphite and AlBr3/MSC-showed structure selectivity in the isomerization of xylenes in which 1, 2 methyl shift mechanism has been considered and the isomerization of xylenes was almost first order with respect to the pressures of the reactants. Mavradinova et al.[5] have compared the catalytic action of a series of pentasil type zeolites with almost the same Si/Al ratio synthesized by different methods and they have reported that pentasil type zeolites with close chemical composition exhibited pronounced differences in crystal morphology, acid function (active site number and strength distributions), catalytic activity and selectivity depending on the method of preparation.
 | |
| Fig. 1: | Mixed xylenes |
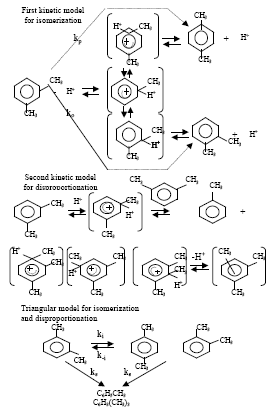 | |
| Fig. 2: | Different kinetic models for isomerization of m-xylene |
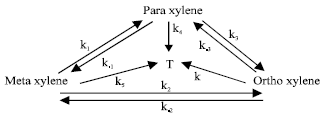 | |
| Fig. 3: | Reaction network |
It was observed that the disproportionation of m-xylene was more sensitive to the acidic function of the catalysts, whereas their activity and selectivity with respect to the yield of xylene isomers were more consistent with the presence of differences in the size and morphology of the zeolite crystallites. In a U.S Patent[6], it is reported that a xylene isomer mixture is treated with hydrogen in the presence of a cracking catalyst comprising a zeolite selected from ZSM-5, ZSM-11, ZSM-12, ZSM-34 and ZSM-48 series at least 20% of its cation site being occupied by a cation of a metal selected from alkali metals and alkaline earth metals such as lithium, sodium and strontium and a refractory inorganic oxide (gamma-alumina) having platinum (in a weight percent of 0.005 to 5% based on the weight of the carrier) supported thereon to suppress side reactions involving a loss of xylenes such as disproportionation and hydrogenation. Dumitriu et al.[7] was found that HZSM-5 had the best selectivity for both the isomerization reaction and p-xylene formation. Acid zeolites catalyse the isomerization of xylenes and also their disproportionation into three methylbenzene and toluene. Two kinetic models, the first taking into account only m-xylene isomerization to p-and o-xylene and the second considering the isomerization and disproportionation reaction of xylene, were adopted. In a triangular model (Fig. 2) involving simultaneous isomerization and disproportionation was also tested and the rate constants were calculated. The calculations of the rate constants indicated that HZSM-5 was a proper catalyst for m-xylene isomerization though its activity was smaller than that of zeolites exchanged with rare earth metals, the selectivity of HZSM-5 was undoubtedly higher. Acid properties of HNaZSM5 [Si/Al=47 and Na/Al=0.85] zeolites of various exchange degrees (0-98%) were studied[8]. Isomerization of o-xylene and alkylation of toluene with methanol were chosen as the test reactions, both of them known as proceeding on the Brönsted acid sites, to investigate the catalytic activity of zeolite. The strength of the acid centres and their concentrations were studied by IR spectroscopy. In the zeolites of exchange degrees higher than 11%, the conversion increased linearly with the number of OH groups of 3609 cm-1 which were strong Brönsted acid sites. Conversion of m-xylene was also studied on H-faujasite and on several acid zeolites with ten-or twelve-membered rings[9,10]. It was reported that in the m-xylene conversion, activity of H-faujasite depended strongly on its degree of dealumination and p/o selectivity was always high for ten-membered ring zeolites with crystals bigger than 1 μm although twelve-membered rings showed much more disproportionation. Leu and Chao studied[11] the properties of crystalline microporous molecular sieve AlPO4 modified by incorporating B, Si, Fe, Ca, Mg or Zn element into its framework. The incorporation of magnesium in the AlPO4 structure gives high preference of p-xylene, whereas the incorporation of silicon and ferric cations results in a value of p-/o-xylene ratio less than unity. The incorporation of cobalt into AlPO4-5 leads to the o-xylene/ p-xylene yield in a state of equilibrium. Sastre et al.[12] investigated the conversion of m-xylene over offretites with different levels of ion exchange. The main conclusion of that study is that the catalytic activity increases monotonically upon exchange of K+ cations. By removal of these cations, the concentration of protons increases as the accessibility to the internal porosity of the zeolite with the result that the selectivity for isomerization is promoted at the expense of disproportionation. Pere’Z Parientence[13], studied the relationship between the pore structure and Si/Al ratio of zeolite β, which is twelve-membered ring zeolite, with its catalytic activity and selectivity in the conversion of m-xylene was elaborated. The isomerization and disproportionation activity of zeolite β depend strongly on the aluminum content, decreasing with increasing framework aluminum content. Maximum activity is found for Si/Al ratios between 14 and 15, while the p/o selectivity remains constant. It was seen that the p/o selectivity in m-xylene isomerization is ca. 1.2, independent of the Si/Al ratio of zeolite β and of the conversion. The studies of Shashikala et al.[14] showed that conversion increased with temperature, but para selectivity decreased and much higher m-xylene conversion, as well as higher stability of the catalyst is achieved by using platinum-exchanged HZSM-35 (0.05wt.% platinum). Lü et al.[15] used a pulse microreactor-chromatography technique to study the xylene isomerization reaction on HZSM5 zeolite catalyst. The kinetic parameters of the reaction network proposed, (Fig. 3), were calculated. The catalytic properties of zeolites ZSM-5 dealuminated by HCl were investigated for isomerization of m-xylene[16]. Besides the increase of the Si/Al ratio, the dealumination leads to an inversion of the Si/Al concentration gradient in crystallites, to a shift of acidity spectrum and consequently to a change in catalytic activity and selectivity. A theoretical model modified by Wugeng et al.[17] was used to explain and predict the high para-selectivities in toluene disproportionation and xylene isomerization. The reaction of m-xylene is a useful tool for characterising zeolite structures especially for all range of pore sizes[18]. The selectivity of these reactions gives information allowing one to estimate pore diameter and architecture for medium through extra-large pore zeolites. As one-dimensional large and extra large pore zeolites give a p/o ratio<1, likely due to a significant occurrence of the bimolecular isomerization mechanism, multi-dimensional large pore zeolites such as USY and beta and the low-silica, uni-dimensional zeolite LTL give a p/o ratio>1.
 | |
| Fig. 4: | Preparation steps of the catalysts |
The straight channel uni-dimensional zeolites SSZ-24, -31, SIT5, UTD-1, ZSM-12, -48 have the lowest p/o ratios over the range of pore size studied. Morin et al.[19] examined influence of coke deposits on the selectivity of m-xylene transformation on the isomerization mechanism over HFAU zeolites and found the stronger the acid sites the faster their deactivation by coke deposits.
As understood from the given literature, much attention has been focused on the shape selective catalysts, especially ZSM-5 type zeolites, for the isomerization of m-xylene. On the other hand, the kinetics of xylene isomerization over some zeolite based catalysts has been studied by several researchers. To understand the kinetic behaviour of xylenes in isomerization reaction on the catalyst, the knowledge of detailed kinetic parameters must be available. It means that more theoretical and experimental study is needed. The aim of this work is to investigate kinetics of m-xylene isomerization over a selective catalyst determined after a selectivity screening-test, to calculate the kinetic parameters of the model and to compare them with the values in the previous studies.
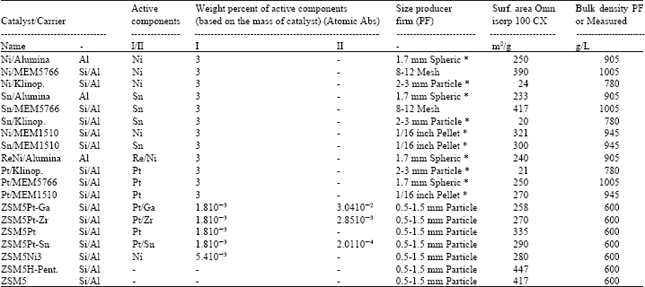
Catalyst preparation: Eighteen different catalysts depending on the information about the catalysts in literature for the isomerization of m-xylene were prepared. Alumina, natural zeolite (rich in clinoptilolite) and several synthetic zeolites such as MEM5766, MEM1510, ZSM5(Na) and ZSM5(H)-Pentasil were employed as carriers and aqueous solutions of salts of metals such as Ni, Sn, Re, Pt, Ga, Zr, Sn were used to add the active components to the carriers. Preparation steps of the catalysts are given in Fig. 4. Origins of materials used in the catalyst preparation and some physical properties of the catalyst were presented in Table 1.
| Table 1: | Origins of materials used in the catalyst preparation |
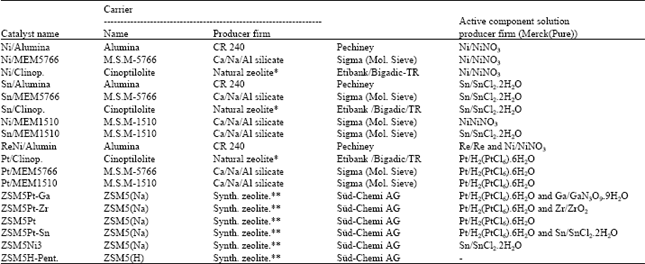 | |
| * Composition: 74.86% SiO2, 0.42% Fe2O3, 0.70% MgO, 0.5%Na2O, 2.71%K2O, 9.074%H2O, 2.42% CaO, 9.35% Al2O3 ** It was on ZSM5(NH4) structure and pretreated with NH4Cl | |
| Table 2: | Percent composition, surface areas and bulk densities of the catalysts used in the experiments |
 | |
| *Divided into 1-2 mm of particles for this study | |
The chemical compositions of the catalysts prepared were determined by gravimetric and volumetric (atomic absorption spectrometer) methods and were given in Table 2. Nitrogen adsorption experiments were performed at 77K using a static volumetric apparatus (Coulter Omnisorp 100 CX) up to P/P0~0.95. The catalyst samples were previously degassed at 400°C for 4 h before nitrogen adsorption measurements. Specific total surface areas and monolayer pore volumes were calculated using Langmuir equation and specific pore volumes were estimated from the nitrogen uptake. Table 3 shows the results of N2 adsorption measurements of ZSM5, ZSM5PtGa and ZSM5Pt catalysts. An attempt was made to apply the Dubinin-Astakhov (D-A), Langmuir and BET adsorption models for the characterization of the catalysts. The Dubinin-Astakhov equation is expressed as:
Log W= Log W0-k (Log P/P0)n |
Where, W is the amount adsorbed at the relative pressure, W0 is the total micropore adsorption capacity, k is a constant, n is the structural parameter. The value of the adsorption capacity, W0, can be obtained from the intercept of the D_A plot (Log W) vs. (Log P/P0)n. Table 3 shows that surface area of ZSM5 decreases with impregnation of Ga and/or Pt.
| Table 3: | The results of N2 adsorption on the catalysts (Degas temperature=400°C) |
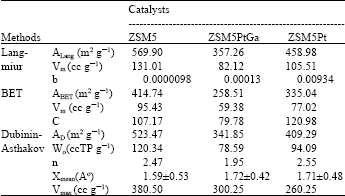 | |
In the Table 3, ALang, ABET and AD-A are surface areas, Vm is monolayer capacity, b and c are constants, Xmean is half width of pores and Vmax is the maximum amount adsorbed. Impregnation with Ga and/or Pt causes a decrease in the pore volume of original ZSM5, resulting in the increase of half width of pores.
Experimental set-up: Set-up is given in Fig. 5. Nitrogen was used as the hydrocarbon carrier. It was cleaned from dust, CO2 and moisture by passing through a series of glass wool, KOH and silica gel, respectively and was metered with a rotameter before fed to the reactor. The hydrocarbon stream from the saturator was fed to the reactor through a pyrex tube that was kept over 140°C in order to prevent xylene condensation. M-xylene was kept in the saturator located in a constant temperature oven and the gas flow leaving the system was also measured with a flow meter. The hydrocarbon heated at constant temperature was carried by the nitrogen flow to the reactor. The amount of hydrocarbon stream carried is dependent on both the velocity of the carrier gas and the temperature of the saturator. During the experiments the temperature of the saturator was kept constant at 70°C, but the gas feed rate was changed between 1 and 2.5 mL sec-1 to carry hydrocarbon in different amounts to the reactor.
The amount of hydrocarbon carried to the reactor at each experiment was sensitively measured by weighing the saturator at the beginning and at the end of the experiment. The tubular reactor employed in the experiments was in 26 mm in diameter, made of pyrex glass. It was located in an electrical oven and heated by radiation. A Fe-constant an thermo element was located in the middle of the catalyst bed to measure the temperature of the catalyst and the temperature was recorded continuously during the experiment and it was kept constant. The product stream from the reactor passed through two cold traps was connected in series. The cold traps were kept in salt-ice mixture and liquid nitrogen, respectively. The products and unreacted m-xylene collected in two traps were analyzed by a gas chromatography (GC) Hewlett Packard 5890/2; on a capillary column of HHP-FFAP using flame ionization detector (FID).
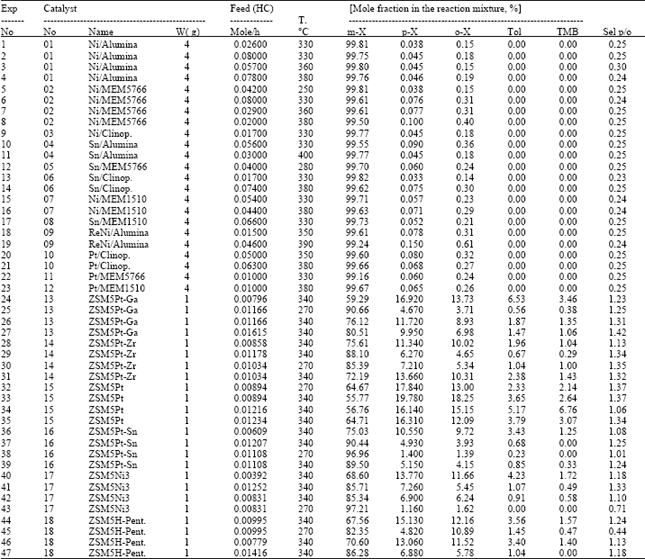
Selectivity studies: These experiments were carried out with a molar flow rate of m-xylene varied 0.00392 and 0.08 mole h-1, at a temperature range of 270-380°C, with a constant fresh catalyst amount (1 or 4 g.) for a reaction time of 4 h. At the end of each experiment, used catalyst was regenerated under nitrogen flow at 380°C for 4 h (Table 4).
Selectivity studies showed that the catalysts numbered from 1 to 12 are almost inactive catalysts in isomerization of m-xylene. Disproportionation products such as toluene (TOL) and threemethlbenzene (TMB) are not present in the reaction mixture over these catalysts even for severer reaction conditions, when compared with catalysts 13-18, higher temperature, higher m-xylene feed and four-fold catalyst amount. Catalysts prepared on ZSM5(Na), numbered from 13 to 18, represent better activity than the above ones. ZSM5Pt (numbered 15) is the most active catalyst under the reaction conditions studied. In the reaction mixture various TMB isomers, toluene are formed as disproportionation products. ZSM5Pt-Ga, ZSM5Pt-Zr and ZSM5Pt exhibit higher selectivity on p-xylene compared to o-xylene, Sp/o, than the others. These catalysts possess the catalytic activity in the order:
ZSM5Pt > ZSM5PtGa > ZSM5PtZr |
Catalysts ZSM5Pt and ZSM5PtGa, which are the ones composed of 1.8*10-3 (in w%) Pt and 3.04*10-3 (in w%) Ga, 1.8*10-3 (in w%) Pt over ZSM5 support, respectively, were chosen for kinetic analysis, due to the their higher activities.
Kinetic study: Kinetic runs were performed under the similar conditions to those in selectivity studies. A kinetic run took 8 h. One gram of catalyst (except one group of experiments, which were carried out to find the effect of catalyst amount on reaction rate) was placed to the reactor. During the runs with different flow rates of m-xylene, the change in the feed rate of m-xylene was obtained by varying the flow rate of nitrogen (between 1 and 2.5 mL sec-1) which is used to carry hydrocarbon to the reactor.
After each run, the reaction mixture collected in the traps was analysed by the gas chromatography. In the reaction mixture p-, o-xylene as isomerization products, various trimethylbenzene (TMB) isomers, toluene were formed as disproportionation products.
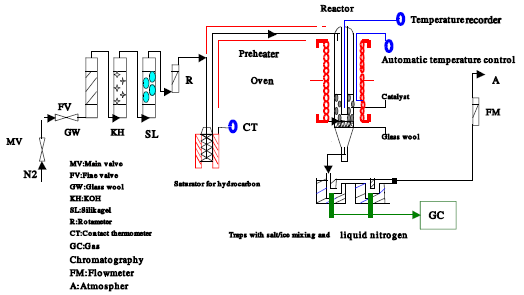 | |
| Fig. 5: | Experimental set-up for the isomerization reaction of m-xylene in gas phase |
| Table 4: | Results of the selectivity experiments |
 | |
| Table 5: | Results of for kinetic experiments |
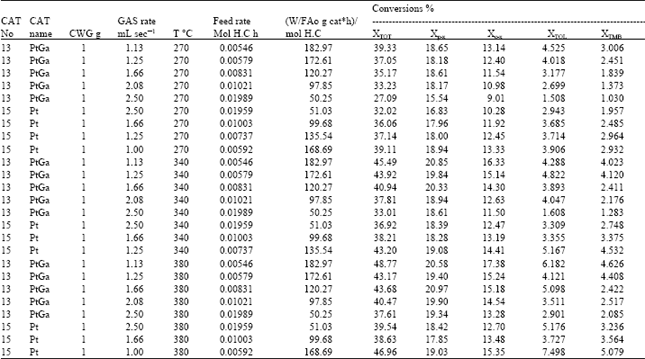 | |
| Table 6: | Correlation of xi versus. W/FAo data (R= Correlation coefficient) |
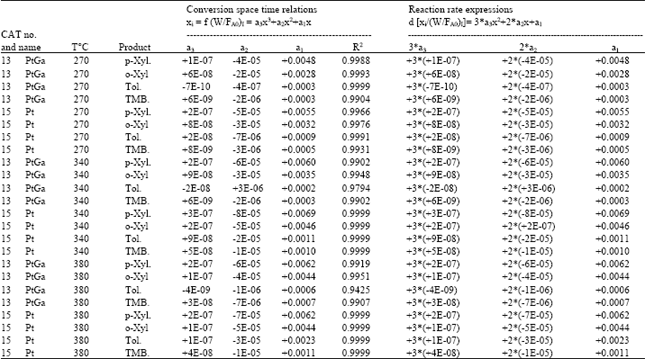 | |
Unreacted m-xylene was measured but no benzene was observed in the reaction mixture. In the Table 5, the total conversion of m-xylene, conversions to p-, o-xylene and conversions to toluene and TMB are represented. The conversions on Table 5 were obtained from mole fractions of related components in the reaction mixture. The reaction network which was used in kinetic modeling, is presented in Fig. 6.
This model takes into account m-xylene isomerization to p-, o-xylene, disproportination and demethylation reactions of m-xylene, The reactions between o-xylene and p-xylene and between methyl groups were neglected, Assuming that the reactions are first order, the rate expressions were given in Fig. 7.
| Table 7: | Reaction rate constants with data in Table 5 |
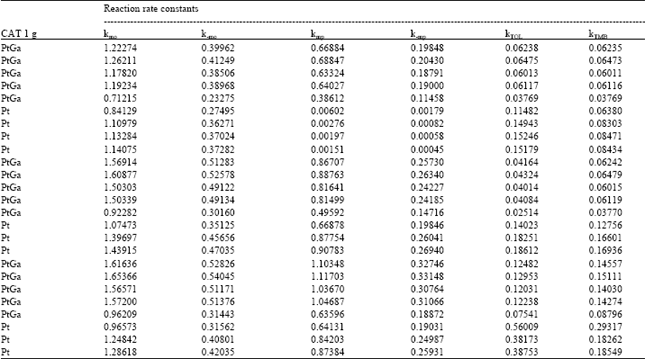 | |
| Table 8: | Activation energies and frequency factors of the rate constants |
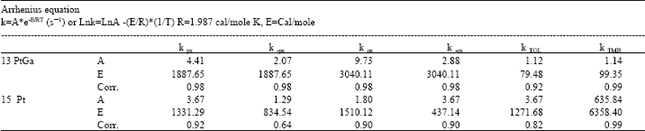 | |
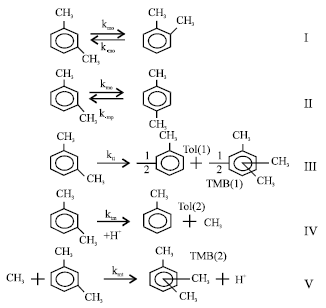 | |
| Fig. 6: | The reaction network used in kinetic modeling |
| Table 9: | Comparison of the kinetic parameters calculated with those of (D) Dumitriu et al.[7] and (L) Li et al.[15] obtained on several catalysts |
 | |
| k5=kTol+TMB | |
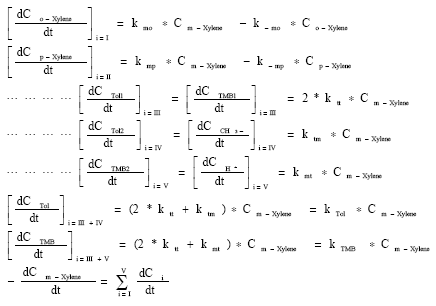 | |
| Fig. 7: | The reaction rates used in kinetic modeling |
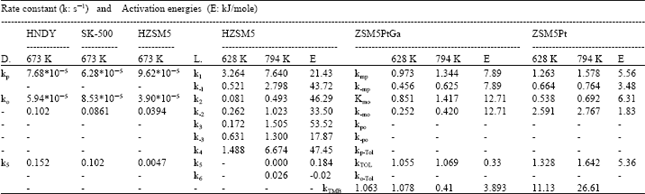
Xylene concentrations in the rate equations were expressed in terms of related conversions which were calculated from the mole fractions in the reaction mixture[20]. Because of the fact that total conversions achieved in all the experiments are greater than ≥10%, the reactor is treated as an integral reactor, By using the continuity equation in a tubular reactor for plug flow behaviour, the ratio of tube diameter/particle diameter should be ≥ 10, which is 19.5 to conclude plug flow behaviour[21] reaction rates were obtained by differentiating the Xi versus W/FA0 curves drawn for all runs for each product where Xi is the conversion to the product, such as o-xylene, p-xylene, toluene and TMB[20]. First of all, the best curve fitting to the Xi versus W/FA0 data, making it pass through the origin is found, For this purpose, several polynomials with different orders are tested for the goodness of fit of the polynomial with experiment, the best fit is determined and the slope of the curve at any point gives the reaction rate at that point. Table 6 presents the correlation of Xi versus W/FA0 data, Table 7 represents reaction rate constants calculated using the experimental data in Table 5. Finally, the activation energies and frequency factors of the rate constants corresponding reactions were estimated by means of Arrhenius equation. Table 8 represents these results with regression coefficients.
Partial and total conversions of m-xylene vs, space time (W/FA0) plots were presented in Fig. 8 for ZSM5PtGa and ZSM5Pt catalysts at different reaction temperatures. It is obvious that there is an appreciable increase in conversion to each product as the temperature increases from 270 to 340°C, Above 340°C, such a significant change couldn’t be observed in the partial and total conversions of m-xylene.
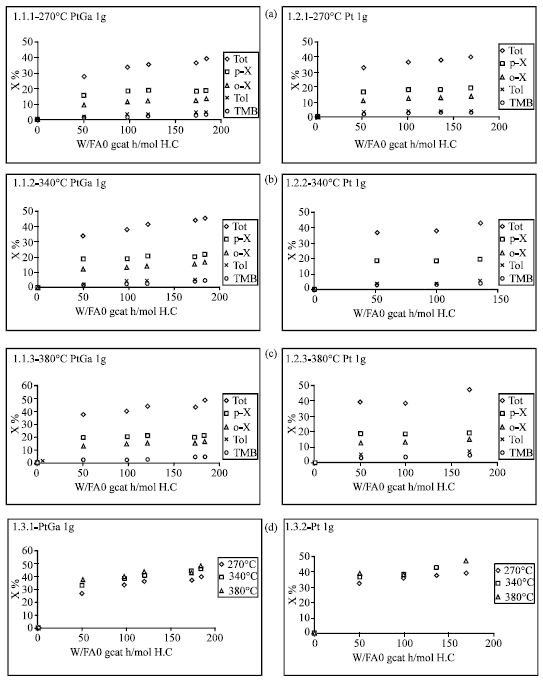 | |
| Fig. 8: | Partial and total conversions of m-xylene vs space time (W/FA0) |
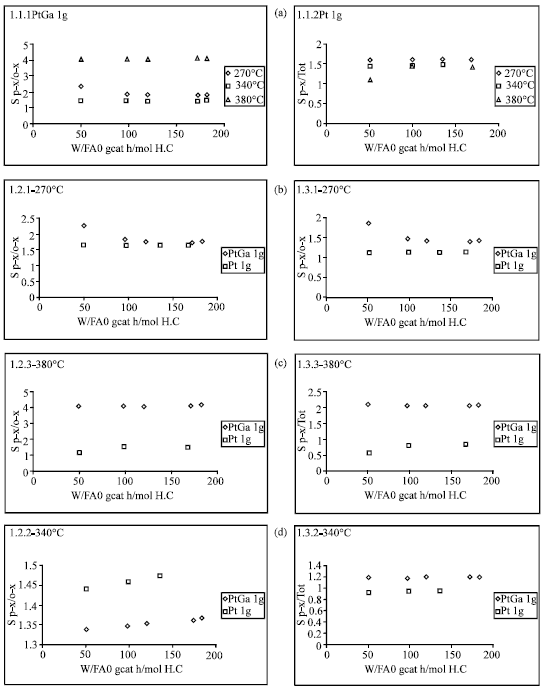
Figure 9 illustrates the effect of temperature on the selectivities to several products calculated from data in Table 5. Important increases (almost four fold) in the selectivities sp-/o-and sp-/TOT were obtained over ZSM5PtGa catalyst at 380°C and addition of Ga to ZSM5Pt catalyst improved the selectivity to p-xylene.
P-/o-selectivities remained almost unchanged as the total conversions increased. The Fig. 9 presents p-/o-selectivities at all the temperature studied over the two catalysts and for all the temperatures studied. It was observed that in general p-/o-selectivity has not been affected significantly with increasing temperature from 270 and 340°C over ZSM5PtGa and ZSM5Pt catalysts. It was observed that there was no a significant difference in selectivities over ZSM5PtGa and ZSM5Pt catalysts in the temperature range of 270 and 340°C. The kinetic model taking into account m-xylene isomerization to p-and o-xylene, disproportionation and demethylation reactions of m-xylene fitted the experimental data fairly well. First order reaction mechanism was used and isomerization was taken reversible, but disproportionation and demethylation irreversible[20].
 | |
| Fig. 9: | Effect of temperature on the selectivities to several products |
By use of standard procedures[22] it was shown that both external and internal diffusion do not influence the reaction rate, External diffusion effects were calculated by use of the generalised JD factor and it was found that (Cb-Cs)/Cb was equal to 3.5*10-4 for ZSM5PtGa and 2.1*10-4 for ZSM5Pt catalyst at the maximum reaction temperature of 380°C, so it was assumed that Cb=~Cs, To calculate the internal diffusion effects, the generalised Thiele modulus based on the reaction rate was determined, For this purpose, Weisz’s criterion, rk2(rHCρk/DeCs)=<1,0, where, rk:diameter of catalyst, ñk:density of catalyst, De: effective diffusion coefficient, Cs:concentration of reactant on the catalyst, surface, rHC: reaction rate, was tested for the experiments, The ratio mentioned above was found to be 0.1396 and 0.1351 for ZSM5PtGa and for ZSM5Pt catalysts, respectively, It means that Weisz’s criterion was satisfied, Hence the effectiveness factor can be taken equal to unity which means that internal diffusion resistance can be neglected.
The kinetic parameters calculated in this study were compared with the ones obtained by Dimitriu et al.[7] and Li et al.[15]. Dimitriu et al.[7] tested the zeolites SK-500, HndY and HZSM5 in the reaction of m-xylene isomerization, It was found that HZSM5 had the best selectivity for p-xylene formation, Three kinetic models including isomerization and disproportionation reactions were employed to explain the kinetic data, It was observed that as the temperature was increased, the rate constants were also increased, as expected, Li et al.[15] used a pulse micro reactor chromatography technique to study the xylene isomerization reaction on HZSM5 zeolite catalyst, A mathematical model including diffusion, adsorption and reaction steps were developed, They also declared that the reaction rate constants increased with increasing temperature, They observed that m-xylene and o-xylene couldn’t produce any toluene, i.e. k5 and k6 in Fig. 3 had to be small, because m-xylene and o-xylene were strongly adsorbed and dealkylation was very difficult under the experimental conditions used by them, As seen from Table 7 clearly, rate constants calculated in this study also increase with increasing temperature, which is the right trend for the constants, Activation energies obtained in this study are very low with respect to the activation energies given by Li et al.[15]. But there is a good fitness between the values of the reverse reaction rate constants of the isomerization reaction calculated at 628 K in this study by using Arrhenius equation obtained from experimental data (Table 9) over ZSM5PtGa catalyst and by Li et al.[15], over HZSM5 catalyst. It is obvious that the numerical values of the kinetic parameters given by Dumitriu et al.[17] are very different from those given by this study and by Li et al.[15]. It should be kept in mind that the catalyst, the experimental conditions, the kinetic models used in the studies mentioned above have significant differences which cause important deviations in the numerical values of kinetic parameters.
An experimental run took 8 h and no significant catalyst deactivation was noted. Just to have an idea about the magnitude of the deactivation, isomerization of m-xylene was carried out over ZSM5Pt and ZSM5PtGa catalysts for durations longer than the reaction time used in kinetic analysis (8 h) at the same temperature (340°C) and at a constant feed rate of m-xylene. As the reaction time increased from 8 to 40 h, a significant decrease in total conversion of m-xylene (over ZSM5PtGa from 40 to 20% and over ZSM5Pt from 42 to 18%) was observed[20], but up to a reaction time of 8 h, an appreciable decrease in total conversion was not measured and even a significant change in color was not noted over two catalysts employed. However, it was clearly seen that addition of Ga to catalyst lengthened its life.
In this study, gas-phase isomerization reaction of m-xylene was studied over various catalysts including Ni, Sn, Pt, Ga, Re or Zr in different compositions over alumina and zeolite catalysts. The catalyst composed of 1,8*10-3% Pt and the one composed of 1.8*10-3% Pt and 3,04*10-3 % Ga, over ZSM5 support were found to be the selective ones to p-xylene, The ZSM5 catalysts including Pt and Zr and the HZSM5 type of zeolite have also given rather good results when compared to the other ones prepared, The kinetic model with first order mechanism taking into account m-xylene isomerization, disproportionation and demethylation fitted the experimental data fairly well.
Funding for this work from Ege University Research Fund through Grand 91Müh029 is gratefully acknowledged.