ABSTRACT
This study presents alternative methods for the utilizing of Dead Sea resources. These include crystallization through cooling by passing the Dead Sea water across cooled surfaces, or by direct contact between a cooling agent and the water solution. The cooling process of the Dead Sea water leads to the saturation state, after which precipitation of the main salts occurs. However, more work is required to study the effect of freezing on the crystallization process and to obtain the equilibrium data for the Dead Sea solution under cooling conditions. Another alternative method for crystallization of the Dead Sea salts is the adiabatic vacuum crystallization, which depends on the evaporation of water at low temperature as a result of the presence of vacuum. Here the rate of decreasing the Dead Sea level due to evaporation in solar evaporation ponds can be reduced to a minimum by condensing the obtained vapor and recycling it back to the Dead Sea or reuse it for the industrial processes.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jas.2005.1334.1339
URL: https://scialert.net/abstract/?doi=jas.2005.1334.1339
INTRODUCTION
The Dead Sea in Jordan is considered to be the saltiest lake and the biggest natural resources of minerals in the world. The Dead Sea is located 400 m below sea level in the western part of Asia between Jordan and Palestine in the breakdown cavity formed more than 2 million years ago. The area of this lake is 1,000 km2, 75 km length, 17 km width and about 400 m deep. The Dead Sea had its present status for more than 12000 years, a period long enough to have sediments of salts reaching up to 100 m[1,2].
The water of the Dead Sea is a highly concentrated mineral solution 350-420 g L-1. which is about 10 times than that in any water area in the world. Table 1 shows a comparison of minerals concentration between the Dead Sea and other seas and oceans[3,4]. The analysis of these data shows that the salinity of the Dead Sea exceeds that of Atlantic Ocean by about 8 times and that of the Mediterranean by about 7 times, that of the Black Sea by about 14.5 times and that of the Baltic Sea at about 40 times. An attractive point is that despite the water from the river Jordan and the periodical streams and wadis pouring into the Dead Sea not the least effect of such water are noticed to lessen the total mineral concentration in the Dead Sea[4].
| Table 1: | Salts concentration in different water surfaces |
 | |
| Table 2: | Ionic concentration in different water surfaces |
 | |
The concentration of Potassium (K) in the Dead Sea water is 20 times more than that in Atlantic Ocean and that of Magnesium (Mg) is 35 times more and that of Calcium (Ca) is 45 times more, but that of Bromine (Br) is about 90 times more. Yet the concentration of Sodium chloride (NaCl) in the different water areas is about 77% of the total mineral content whereas it is only about 25-30% in the Dead Sea (Table 2)[4].
Surprisingly, Potassium Chloride (KCl) sedimentation occurs as a result of natural evaporation of different seawater surfaces only in the Dead Sea. This important component can be separated by crystallization as a result of the Dead Sea water natural evaporation. The process of exploitation of this mineral began on both banks of the Dead Sea in1930[3].
The high concentration of minerals in the Dead Sea increases the density of its water. The density depends on the concentration, temperature and on the depth of water in the Dead Sea. The density of the Dead Sea water as measured on the basis of salts concentration reaching up to 265 g L-1 is about 1.3-1.4 g cm-3 whereas the density of the Atlantic Ocean water is 1.023-1.030 g cm-3[3] and this explains the buoyancy forces in the Dead Sea.
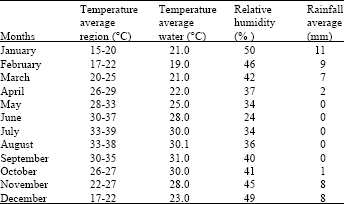
Air temperature in the Dead Sea district reaches 29-39°C in summer and it exceed 40°C in certain years. Therefore, the water is exposed to an intensive evaporation process, which leads to the existence of salty sediments on shores similar in different shapes to coral ones. And as indicated in Table 3, the average temperature over the period from November to April is 22-29°C and over the period from May to September and October is 32-37°C and in June July is 38-39°C. The humidity level is low 27% in summer and 38% in winter. The atmospheric pressure is 800-810 mm Hg; while the oxygen concentration in the atmosphere of the Dead Sea is 15% more than of the Atlantic Ocean (Table 3).
The present situation of existing economic projects on the Dead Sea shores: Nowadays, the Dead Sea is intensively exploited. In addition to the many medical and tourist facilities on its shores there is a group of productive industries to produce minerals, which are used in chemical fertilizers industry.
Such an intensive exploitation of water and minerals of the Dead Sea began to affect the existence of the Dead Sea itself. The river of Jordan after being diverted by Israel, it does not provide enough water to the Dead Sea. The low level of rainfall and the construction of Wadis damps add more to the problem of water evaporation. Numerous studies[5-7] point to the inevitability of the Dead Sea disappearance if such decrease on sea level continues, this decrease is expected to increase within few coming years due to the environmental unbalance around the Dead Sea because of the above mentioned reasons.
The step-by-step disappearance of the Dead Sea will lead to an environmental catastrophe affecting the region around and no one will be able to predict the negative effects of such a catastrophe, which may resemble that which occurred around the Oral Sea, which disappeared because of wrong water policies, which followed the ex-Soviet Union.
The existing industries on the Dead Sea shores are considered as key factors in deteriorating its environment. Such industries have a pioneering position as to the world classification of the chemical fertilizers industries, which depend on potassium.
| Table 3: | Climatic conditions around the Dead Sea |
 | |
Since the early sixties such industries have been working at full capacity. The Israeli treasury in comes in the past few years reached up to eight billions dollars due to the Dead Sea product industries and others depending on these industries.
Dead Sea products are exported to world markets such as the USA, Brazil, European countries, South Africa and some Asian countries.
These factories are trying to increase the productive capacity annually and in recent years other new industries have been inaugurated such as Magnesium producing industry that is an expensive substance as well as the Bromine. These industries utilize the solar evaporation systems to extract salts whereby seawater is pumped to special salt ponds that occupy large areas of land extending to more than 120 km2 only on the Jordanian side[2]. These ponds are working in series, NaCl is precipitated in the first step then the solution is transported to other ponds where the carnallite (KCl MgCl2.6H2O), which is the key product is crystallized and precipitated, after which the remaining solution[2,7] is returned to the sea as it is demonstrated in Fig. 1.
Such process is considered a naïve one in producing salts, which has some defects such as: The process is a seasonal featured one where the solution is pumped to ponds at the beginning of April and the process of natural evaporation and crystallization continue until December.
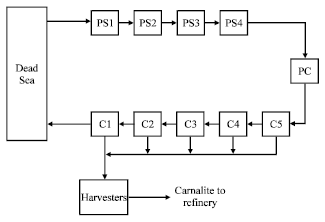 | |
| Fig. 1: | Solar evaporation ponds of Arab Potash Company on the Dead Sea shore |
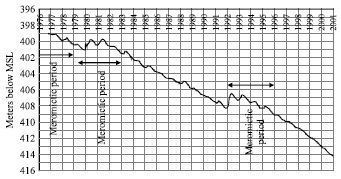 | |
| Fig. 2: | Changes on the Dead Sea level (7) |
Then precipitated salt is harvested by harvesting machines, which float on the pond ’s surface and pump salt from the bottom of ponds through pipes to the shore where salts are separated, dried and stored.
Some seasonal rainfall in summer leads to dilute the solution in the ponds leading a change in water solution-crystal phase equilibrium, as well to slow the crystallization process where the formed crystals may begin to dissolve in the solution again.
The construction of such ponds depends on building a clay or some times salt dams and bars to separate ponds from each other, which requires a lasting maintenance process to stop the leak and on the other hand may cause a considerable quantity of the solution to leak which at times ranges from 10-15% of the whole solution.
The solution may stay for a long period of time in these ponds, which leads to mushroom-type sediments covering the ponds bottom and appearing on the surface, which may require great efforts and high cost to clean and to remove. Such ponds occupy large areas of land pieces, which would possibly be exploited to build tourist areas on the Dead Sea shores.
Evaporation rate process increases as the surface of ponds increases. Some studies[5-7] indicate that the loss rate of the Dead Sea water due to natural evaporation reaches more than one hander million cubic meters annually without any compensation. The plot in Fig. 2 shows that the Dead Sea level has been decreasing about 1 m annually since 1976[7].
Prospective methods for salt extraction by cooling of Dead Sea solution: The expected catastrophe due to the decrease of the Dead Sea level stimulate efforts toward environmentally friendly methods to exploit natural raw materials in the Dead Sea in order to restrict the evaporation rate resulted from the present industrial activity and to bring the balance back to this sea.
One of prospective approaches that can be use in the near future is cooled crystallization processes.
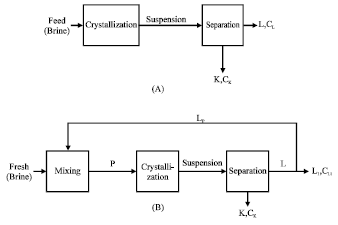 | |
| Fig. 3: | Principle scheme of single mass crystallization process through cooled surface, (a) without recycling part of solution, (b) with recycling part of mother liquor |
In these processes, saturation state of the Dead Sea brine solution can be reached by cooling down to a certain temperature, which is necessary to begin the process of crystal nucleation and growth of salts after which the remaining solution can be returned back to the sea without any loss in water content.
Mass crystallization through cooled surfaces: This process is usually used to separate solutions from impurities on a large scale in chemical industries, petroleum industries and food industries[8,9]. This process depends on the difference of solubility of salts and element in the solution to be separated between the solid and liquid phases[10,11].
The process is usually carried out through two successive stages; crystallization which takes place in the whole volume of the solution and then separation of crystals formed. This process is applied by using batch crystallizer where the solution feed is pumped initially to a crystallizer surrounded by a cooling jacket and when the temperature decreases to a certain degree (crystallization temperature), the crystals nucleus begin to grow directly, the process is carried out for a certain period after which the obtained suspension is pumped to the separation stage where crystals are filtered from the remaining solution. More than one crystallizer can be used by being successively linked to obtain the required degree of separation. The yield of crystal phase and its concentration depend on the type of the equilibrium phase diagram, the concentration of the feed brine solution and temperature of fractionation. A principle scheme of this process is given in Fig. 3.
Mass crystallization through direct contact between cooling agent and solution: This process depends on mixing the solution to be separated directly with a cooling agent[9,12-14].
 | |
| Fig. 4: | Direct contact crystallization processes by using cooling liquid agent |
This process differ from other processes in the following aspects: -The high effectiveness of heat and mass transfer, which due to the increase in the contact surface area between the solution and the cooling agent because of their direct contact resulting in a more effective mixing process.
The simple design of the crystallizer for direct contact between the solution and the cooling agent. This makes maintenance easy and at the same time prevents crystals from being formed on the contact-cooling surface.
Some of the main disadvantages of this process are that it needs an additional stage for the separation of the cooling agent from the solution and the possibility of the crystals being polluted by traces of the cooling agent after separation.
This process can be classified according to the aggregation state of the cooling agent.
Direct contact by using cooling agent in the liquid phase: This process is used to separate many organic materials such as benzene and naphthalene and which is also being used to separate inorganic elements such as salts from their water solutions[9,10].
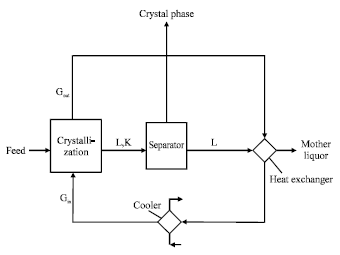
The expenditure of energy consumption for this process can be reduced significantly by mounting two contact columns (Fig. 4). The first one is a crystallizer , where the contact crystallization is taking place and the second one is for heat utilization. In this column the obtained cooled mother Liquor (L) after separation of suspended Crystal phase (K) is crossed with consumed cooling agent coming from crystallizer, as a result of heat exchange between two phases the temperature of recycled cooling agent decreases before it send to a cooler to reach the required temperature for the process.
Direct contact by using cooling agent in the gas phase: This process is used basically to separate minerals from their water solution where the solution reaches a saturation state through cooling and partial evaporation of the solvent.
 | |
| Fig. 5: | Direct contact crystallization by using boiling cooling agent |
The process is operated in a bubble column crystallizer. This process is distinguished for its simple design of crystallization process where there is no need for additional separation stage ,required , between the solution and the cooling agent and here is no pollution of the crystal phase by the traces of the cooling agent[9,10].
One of the most known disadvantages of this process is the great need for huge quantity of gases as a cooling agent because of the low heat capacity of gases compared to liquids.
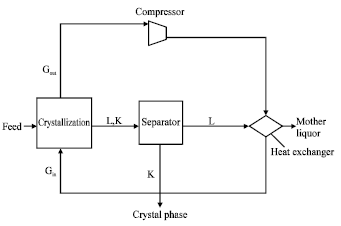
Direct contact by using boiling cooling agent: This process is considered as a variety of the process with gas cooling agent. Here the cooling agent at its boiling point temperature is mixed with a solution. Thus, this process is distinguished by high effectiveness of heat and mass transfer due to the increase of the contact surface area leading to an increase of the efficiency of the mixing process. This process is basically used on large scale in water desalination processes[9,15].
Substances such as methychloride, n-butane and isobutane are used as cooling agents where liquefied gas is mixed with the heated solution (Fig. 5) .The heat is transferred from the solution to the cooling agent, which begins to evaporate at the same time causing the solution to cool down reaching its saturation state required for starting of the crystallization process. Vapor of the cooling agent is compressed by a compressor and directed to the heat exchanger, where the vapor will be condensed as a result of its contact with a cooled crystal phase coming from the separator before it is recycled again to the crystallizer.
 | |
| Fig. 6: | Adiabatic vacuum crystallization |
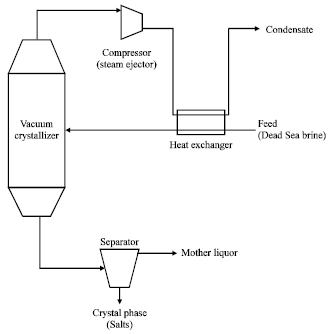
Adiabatic vacuum crystallization: Through this process, the solution is reached to the necessary state of saturation to start the crystallization process by the partial evaporation of the solution under adiabatic conditions and vacuum pressure. The solution temperature decreases as a result of giving up part of its sensible heat and transferring it to a latent heat of vaporization . As a result, the solution begins to boil and cools at the same time until it reaches a saturation state.
This process is used on a large scale in crystallization and separation of minerals in which the salt is not effected by low temperature such as KCl, NaCl and NaNO3. The applied vacuum in this case reaches (25-40 mm Hg)[9,11]. This process can be carried out in vacuum crystallizers of different volume and shapes: single ,multistage, batch and continuous (Fig. 6).
Crystallizers under vacuum pressure are distinguished by their high productivity and can be used in extractive industries; they are also simple in design with voids of special separating surfaces for heat transfer between cooling agent and solution. Such features make possible to manufacture them from any materials provided that these materials can resist corrosion. They are also economical in their operation during which there is no need to introduce or remove any additional quantity of heat because the whole process proceeds under adiabatic conditions.
MATERIALS AND METHODS
In order to compare the visibility of some of the above-mentioned technological methods as a prospective one for separation of minerals from the Dead Sea brine, a series of experiments were conducted to determine some of the thermodynamic properties of the Dead Sea brine such as freezing point, saturation point and the boiling point.
| Table 4: | The boiling point and saturation temperatures of the Dead Sea brine |
 | |
| Table 5: | Boiling point of the Dead Sea brine under different vacuum pressure |
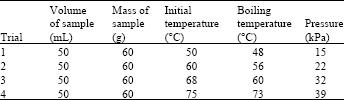 | |
Procedure: A sample of 100 g of Dead Sea water was placed in a 300 mL beaker connected to a condenser and a thermocouple. Heat was introduced to the system by using an electrical hot plate. In order to enhance the heat transfer process a magnetic stirrer was used. The boiling point temperature and the saturation temperature were determined visually. In order to determine the dependence of the boiling point temperature at low pressure, a vacuum pump was introduced and connected to the condenser. The temperature of the initial sample was controlled by placing the beaker in a water bath with controlled heating element. Samples with different initial temperatures were used.
Also a series of experiments were conducted to study the behaviour of the Dead Sea brine under sub cooling conditions. In these experiments a samples of Dead Sea brine were placed in ethanol bath refrigeration. The cooling rate was controlled to be (3°C h-1)
RESULTS AND DISCUSSION
The obtained data show that the difference between the boiling point temperature of the Dead Sea water and that of pure water is estimated to be 7°C (Table 4). This rise is due to a high concentration of salts in the Dead Sea water comparing with other Seas and Oceans (Table 1). The amount of pure water that must be removed from the system to reach the saturation state and then to start the process of crystal formation is determined. The average percent of the water removal is a bout 15% of initial solution (Table 4). The saturation temperature (freezing point) of the Dead Sea brine under sub cooling conditions is also experimentally determined. It is found to be (-32°C), this indicates that a strong effect of salts concentration as a result a big decrease in freezing point temperature of the Dead Sea brine was fixed comparing with that of pure water .
The relationship between the boiling point temperature and the operational pressure is increasing linearly. A noticeable decrease in the boiling point to 48°C obtained at 0.3 atm compared with that of 107°C at atmospheric pressure (Table 5).
This data can be used as a starting point for calculations of the energy balances of different proposed technological methods. This can be used to determine the energy required for each process to be carried out and to study the visibility of such methods.
CONCLUSIONS
This study introduces the current status in utilizing the Dead Sea resources. The solar salt ponds method used to extract salts from the Dead Sea cannot cope with modern technological and environmental requirements.
Due to the absence of any data of the Dead Sea water solutions behaviour under cooling conditions, further research is required to construct a phase diagrams of such equilibrium processes. Using the direct solar system in the ponds method does not make the production cost low. These ponds need continuous maintenance to remove salt precipitation especially the NaCl ponds, Moreover; this method is seasonal and related to weather and climate conditions, which oscillate yearly. It is also known that the technology of developing the solar energy is now progressing to convert it into electrical energy, which could be utilized as a power source for the required depressurizing equipment used for crystallization process under vacuum to reduce the production cost.
It is possible to say that using cooling methods with its different combinations for crystallization may be one of the future substitutes, which have to be used to keep the Dead Sea water from evaporation and to maintain environmental balance in the region.
Cooling method for the solution to reach saturation phase may be considered as one of the substitutes, which can be used if more research performed to identify the behaviour of the Dead Sea solution during cooling.
Crystallization process under vacuum pressure is regarded as one of the important and substitutive methods to produce salts from the Dead Sea. This process needs more studies to identify the ideal operating conditions for the Dead Sea solution. the experimentally obtained thermodynamic properties such as (boiling point, saturation point and freezing point temperatures) can be used as a starting data to performer a calculation of energy required for different above mentioned technological processes of salts separation .
ACKNOWLEDGMENTS
The author would like to acknowledge Dr. Reyad Shawabkeh, Dr. Awni Al-Otoom and Dr. Malek Hami for their revision to this research.








