ABSTRACT
Amorphous silica-alumina catalyst samples of different Si:Al ratio as of 90:10, 70:30 and 50:50 were prepared from sodium metasilicate and aluminum sulphate by co-precipitation and gelation methods without pore regulating agent. Surface acidity of these catalyst samples were determined by Tamele method (amine titration method) using n-hexane and n-heptane as solvent instead of benzene used in Tamele method that having a carcinogenic effect and using n-butylamine solution as adsorbate. The effects of concentration of adsorbate, amount of catalyst sample and types of catalyst sample on the surface acidity were studied. Investigation of reproducibility measurement indicated that by selecting a moderate amount of sample and concentration of n-butylamine solution as minimum as possible surface acidity of the silica-alumina catalyst sample could be estimated with a fair degree of confidence. The total surface acidity of all the catalyst samples was greater in n-hexane than that of in n-heptane used as solvent. This result reveals that n-hexane or n-heptane can be used for the measurement of surface acidity of catalyst sample, as solvent instead of benzene in Tamele method but n-hexane is better.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jas.2005.1275.1278
URL: https://scialert.net/abstract/?doi=jas.2005.1275.1278
INTRODUCTION
Surface acidity of finely divided solid means the action of acid surfaces in catalytic reaction. Acidic strength of a solid is defined in terms of its ability to convert a base that is absorbed on its surface into the corresponding conjugate acid. In catalytic reaction, the number, strength and nature of the active sites control the distribution as well as the nature of adsorbed reactants, intermediate and products, as these surface adsorbates are used as probes for activity and selectivity of a catalyst.
In acid catalyses reaction, amorphous and crystalline zeolite (aluminosilicates) are found to be excellent catalysts. The nature of the acid sites on both types of oxides is same but the distribution is different[1]. Acid sites are commonly regarded as the active centers on certain oxides that catalyze many reactions involving hydrocarbons e.g., catalytic cracking, isomerisation, alkylation, cationic polymerization and reforming. The proposed mechanisms of these processes mostly involve in the formation of carbenium or carbonium ions as reactive intermediate and silica-alumina catalysts are highly active for these processes due to the acidic nature of the mixed oxide, where tetravalent silicon in the silicon lattice has been replaced by trivalent aluminum[2]. The acid sites on oxide surfaces are of two types which described by Lewis and Bronsted[3]. A proton transformation is occurred at the Bronsted site from the solid to the adsorbed molecule as an ion is emerged and an ion dipole interaction with the solid takes place while Lewis acid sites accept electrons from the adsorbed molecule and a coordinate bond with the surface is formed. Cumulative research on cracking catalyst reveals that these highly porous solid possesses acidic surface, stable at high temperatures and capable of cracking hydrocarbons by an ionic mechanism rather than the free radical mechanism characteristics of thermal cracking[4]. The acid-base properties of certain mixed oxides can be varied by choosing different oxide constitutions at different concentrations and by changing the treatment of the sample.
A lot of work has been developed for understanding the effect of the support acid/base on the catalytic properties of the metal. The explanations have involved the formation of metal-proton adducts[5]. But the relation between changes in the electronic properties as well as catalytic properties of the metal particle is poorly understood. In general, an acid catalyzed can be characterized for the interaction with an adsorbed species, if the number adsorption sites, strength of adsorption and the nature of the interaction as well as the chemical nature of the adsorbed species are ascertained.
For cracking catalysts adsorption sites are considered to be the acid center and therefore surface acidity of catalysts is correlated with it acidity. Consequently, surface acidity measurements of solid catalysts is of immense important from technological point of view because it does not help only to characterize a catalyst sample but also provides a method of screening a catalyst sample for improved yield in a process. Many methods are available for determining of surface acidity of solid catalyst, of which Tamele method (amine titration aqueous method), temperature programmed desorption method, gas-chromatographic method, spectrophotometric and calorimetric methods are common. The aqueous method, base-exchange method and method of titration of aqueous slurry with a standard base measured the Bronsted acid sites only. Tamele method is a non-aqueous method which determines the total surface acidity (Bronsted and Lewis sites) and has been very popular. But it involves benzene as a solvent that is known to be carcinogenic.
In this study, Tamele method was modified by using n-hexane and n-heptane as solvents instead of benzene and feasibility of this method was studied. For this purpose, amorphous silica-alumina catalyst samples of different Si:Al ratio were prepared by the co-precipitation and gelation method and their surface acidity was measured at different sample amount and also at different concentration of adsorbates.
MATERIALS AND METHODS
All the materials used were of laboratory reagent grade as listed in Table 1.
Preparation of amorphous silica-alumina catalysts and other reagents: The catalyst samples were prepared in the ratio of Si:Al, 90:10, 70:30 and 50:50 by mixing of two solutions of sodium metasilicate and aluminum sulphate, those were prepared separately by dissolving them with requisite amount of de-ionized water.
| Table 1: | List of the used materials |
 | |
In order to complete reaction for gel formation, it was kept standing for 24 h and filtered. The precipitated gel was washed with de-ionized water several times to completely free from sulphate ions. It was then dried in an oven at 120°C and ground in a mortar by means of a pestle applying moderate pressure. They were sieved through 25-200 mesh and activated at 450°C for about 3 h. After cooling in a desiccator, the sample was again ground and sieved through 100-200 mesh that was used as silica-alumina catalyst sample.
n-butylamine solution (adsorbate) of different concentration (0.05-0.6) was prepared by dissolving n-butylamine in n-hexane and in n-heptane, separately. These solutions were used for titration.
The indicator (p-dimethylaminoazobenzene) used in this titration, was prepared by dissolving 0.5 mg of p-dimethylaminoazobenzene in 250 mL n-hexane and also in n-heptane separately. 0.05 N trichloroacetic acid solution was prepared in n-hexane and in n-heptane.
Total acidity of catalyst samples was measured by modified amine titration method where n-hexane or n-heptane was used as a solvent instead of benzene, using the indicator solution of p-dimethylaminoazobenzene in n-hexane and in n-heptane. The variation of total surface acidity of catalyst sample was studied for three different ratios of Si:Al of sample, different sample amount and also for different concentration of adsorbate. All titrations were conducted at room temperature 29-30°C.
RESULTS AND DISCUSSION
Measurement of total surface acidity by the modified Tamele method: 0.1 g (100-200 mesh) of freshly prepared Si:Al catalyst sample was measured into a 10 mL screw cap vial and 5 mL of indicator solution was added that reacts with the solid catalyst to form red acidic form of indicator. The known strength of n-butylamine (adsorbate) was added drop wise from the micro burette until the disappearance of red color of the solution. This solution was allowed to stand about 2 days for minimizing adsorption of amine on non acidic portions of catalyst surface. In order to confirm that no excess amine was present at the end point of titration, a drop of 0.05 N trichloroacetic acid was added indicated that no appreciable excess of amine had been present while the color was unchanged. The acid content (surface activity) of the solid catalyst was expressed as milliequivalent (meq) of acid per g of solid catalyst. Other experiments for different catalyst sample with different concentration of n-butylamine were conducted in the same way.
| Table 2-4: | Comparison of surface acidity of catalyst samples (Si:Al) using different concentration of n-butylamine in n-hexane and n-heptane |
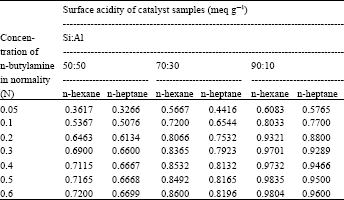
| Table 2: | Surface acidity data of 0.1 g catalyst sample |
 | |
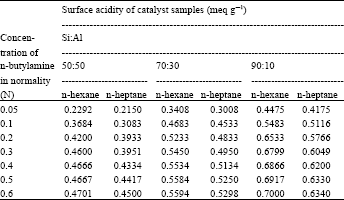
| Table 3: | Surface activity data of 0.2 g catalyst sample |
 | |
| Table 4: | Surface activity data of 0.3 catalyst g sample |
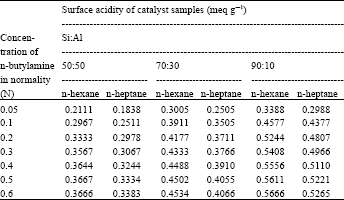 | |
Surface activity data of catalyst samples with different concentration (0.05, 0.1, 0.2, 0.3, 0.4, 0.5, 0.6 N) of n-butylamine in n-hexane and in n-heptane solvent, with different ratios of Si:Al in the catalyst sample and with using the different amount of catalyst sample are listed in Table 2-4 and in Fig. 1- 3. From these Fig. 1-3, it is observed that a concentration of 0.2 N n-butylamine solution is sufficient to saturate all the catalyst samples. Though the catalyst samples show the little increase of surface acidity with the increase of strength of n-butylamine but it resembles BET type-I (Langmuir type) adsorption isotherm.
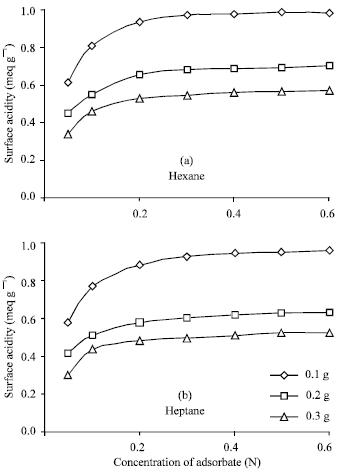 | |
| Fig. 1: | Comparison of surface acidity of Si:Al (90:10) catalyst sample with the amount of sample at different concentrations of adsorbate in (a) hexane and (b) heptane |
From the nature of the isotherm it may be assumed that chemisorption of n-butylamine takes place on catalyst surface giving monolayer coverage, so that relatively dilute solutions of n-butylamine may be used for the present method of surface activity measurement.
Variation of surface acidity with the silica (Si) content in the catalyst sample and also with the amount of sample used: There were used three types of catalyst sample (Si:Al, 90:10, 70:30 and 50:50) and also three different amounts (0.1, 0.2 and 0.3 g) of sample to study the variation of surface acidity with the change of Si:Al ratio in the sample The results were summarized in Table 2-4 reveal that for all the catalyst samples, surface acidity increases noticeably as the sample amount decreases irrespective of the solvents (n-hexane and n-heptane) shown in the Fig. 1 and the effect of Si:Al ratios in the sample on the surface acidity is shown in Fig. 2, using 0.1 g sample. Figure 2 shows that surface acidity of the sample increases with increase in Si content giving the highest value 0.9804 and 0.96 for n-hexane and n-heptane, respectively, for the sample with 90% silica content.
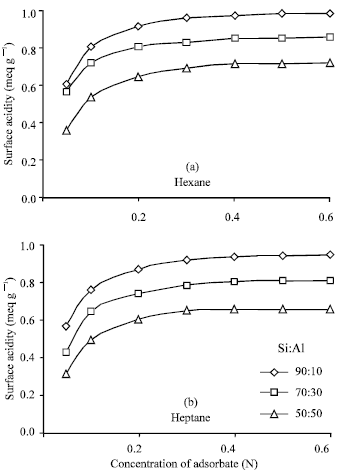 | |
| Fig. 2: | Comparison of surface acidity of Si:Al catalyst sample (0.1 g) with their ratios at different concentrations of adsorbate in (a) hexane and (b) heptane |
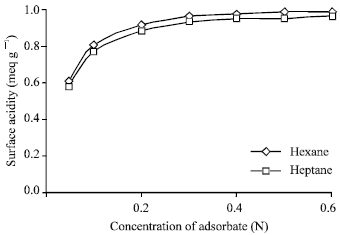 | |
| Fig. 3: | Effect of solvent on the surface acidity of Si:Al catalyst at different concentrations of adsorbate |
This result may be reported that for each base-exchange measurement a typical equilibrium reaction takes place, so as to the proportion of acid released in solution increases with the decreases of sample amount[6].
Comparison of total surface acidity of catalyst sample for using various solvents: n-hexane and n-heptane were used separately as solvent instead of benzene in modified Tamele method to measure the surface acidity of the catalyst sample is shown in Fig. 3, reveals that both solvents n-hexane and n-heptane can be satisfactorily used but n-hexane gives the greater value than that of n-heptane.
CONCLUSIONS
Surface acidity of silica-alumina (catalyst sample) was measured at different silica content in the sample and at different amount of sample by using various concentration of n-butylamine (adsorbate) in n-hexane and in n-heptane that suggests the use of higher content of silica and smaller amount of sample with dilute n-butylamine solution. Considering all the above findings, the total surface acidity of silica-alumina catalyst can be measured by dilute n-butylamine solution using n-hexane or n-heptane as solvent instead of benzene in Tamele method, named modified Tamele method.








