Research Article
Lead and Cadmium Levels in Edible Internal Organs and Blood of Poultry Chicken
Not Available
Joy Elaine
Not Available
P. Kaligayahan
Not Available
Maxima E. Flavier
Not Available
The significant amount of toxic elements that are able to gain entry in foods increases the concern of the analysts on the study of these substances. Lead and cadmium are heavy metals that are considered to be the major concerns as environmental contaminants[1]. These toxic elements do not just primarily affect human beings but also animals. Plants and animal are deemed to be the barriers of heavy metal entry to man. According to FDA data, food and feed crops provided approximately 34 to 44% of the dietary intake[2]. Animals like poultry chicken that have stored appreciable amounts of lead and cadmium can transfer these elements to humans when ingested.
Lead and cadmium can primarily enter the environment through natural sources and human activities increase the amount of these metals in the environment[3]. The use of lead containing gasoline and cigarette smoke contributes to the entry of lead and cadmium into the environment. Cadmium and lead are found to be present in processing and manufacturing processes in the industry.
Exposure to these heavy metals even in small amounts causes adverse effects to man. Moreover, contamination of foods and high amounts of lead and cadmium in the environment affect animal’s health. Several diseases arise from high level exposure to these metals like anemia, infertility and changes in the lungs[3].
From the long term exposure to lead and cadmium, these metals are absorbed and are stored mainly in some organ meats like liver and kidneys. Lead and cadmium are absorbed by the blood and are distributed to the organs in the body. Liver is the central organ for metabolism. It plays a major role in the processing of foods and excretion. Kidneys also have metabolic and excretory functions. Due to these roles of liver and kidneys in the body, significant amounts of toxic metals are being absorbed and deposited in these organs of both man and animals[4]. Therefore, it is important to measure the amount of heavy metals in liver and kidney.
Filipinos are very fond of eating internal organs and blood of animals especially poultry chicken. Since heavy metals usually accumulated in internal organs and blood, monitoring the levels of these toxic metal like lead and cadmium are important. With this, the presence of lead and cadmium on the edible internal organs (gizzard, intestine and liver) and blood of poultry chicken was determined. The effect of the sample matrix in the determination of lead and cadmium was also investigated.
Sample preparation: Fresh samples of chicken’s blood, liver and gizzard were obtained from 10 backyard chickens and kept in freezer until the time of analysis. These tissue samples were collected from freshly slaughtered chickens. Edible internal organs weighing half kilogram were washed with tap water and separately chopped into smaller pieces. The internal organs and blood were homogenized three to five times in on Osteorizer. All of these were done on March 2003.
Digestion: The samples were digested using AOAC Standard Method of Digestion (972.23) with modification. About 20 g each of the homogenized fresh samples and blood were weighed into 50 mL crucibles. The samples were dried for 2 h at 135°C and later ashed in a muffle furnace increasing the temperature until 450°C for 216 h. Each crucible was added with 2 mL of concentrated nitric acid (HNO3) and was then evaporated to dryness on a hot plate prior to ashing at 450°C. The ash that was obtained was then dissolved in 10 mL of 1 M HCl by heating and then transferred quantitatively in a 25 mL volumetric flask. The solution was allowed to cool and then made up to volume with 1 M HCl.
Method validation: Standard addition technique was performed on chicken’s intestine to evaluate the presence of matrix interferences. Different known concentrations (0.00-1.60 ug g-1) of lead and cadmium were to the sample’s digest. The standard calibration and standard addition curve were established using linear regression analysis. The matrix effects in the determination of lead and cadmium was verified by determining the slope ratios of standard addition lines and standard calibration line. A slope ratio less than 1.00 indicates matrix suppression while a slope ratio greater than 1.00 indicates matrix enhancement.
Recovery experiments were also done to assess the accuracy of the method of digestion. This was done since there is no Certified Reference Material (CRM) available for lead and cadmium concentrations in the internal organs and blood of chickens. This was carried out by spiking a known concentration of lead and cadmium in chicken gizzard as well as blank samples. The percent recovery was then calculated as follows:
| x | = | Concentration (ug g-1) of the metal determined in the spiked samples |
| y | = | Concentration (ug g-1) of the metal determined in the samples |
| z | = | Concentration (ug g-1) of the spiked metal in the samples |
In addition, Limit of Detection (LOD) and Limit of Quantitation (LOQ) for lead and cadmium was determined using the procedure as cited by Yang et al.[1].
AAS analysis: The content of lead and cadmium in different samples were performed using an GBC Atomic Absorption Spectrophotometer Model 932AA. The range of standards used were 0.1-4.0 mg L-1 for lead and 0.02-2.0 mg L-1 for cadmium.
Statistical analysis: Significant tests of linear regression for standard lines, recovery assays as well as differences were made using Analysis of Variance (ANOVA). The differences among means were analyzed by Duncan’s Multiple Range test (DMR). Comparison of the slope ratios was done using Least Significance Difference test.
Method validation: The slope ratios for chicken intestine for lead was 0.6717 while for cadmium, the slope ratio was 0.6054 (Fig. 1). These results showed that the sample matrix exhibited suppression effects on the determination of lead and cadmium. In addition, the slope of the standard addition lines was significantly different (p = 0.05). From the slope of the standard calibration line for both lead and cadmium that would indicate the adverse effects of the sample matrix on the analysis of lead and cadmium.
The presence of sodium in the solution might contribute to the suppression of the signal due to its high molecular absorption that cannot be overcome efficiently by background correction system[5]. Matrix effect might also be attributed to large amount of reagents used, nature and amount of the sample used and the difference in temperature and viscosity of the samples and standard solutions. The addition of HNO3 minimizes this effect via the formation of sodium nitrate, which volatilizes during ashing. Nitric acid was then found to be effective in removing unwanted matrix prior to the atomization of the analyte[6]. However, results indicated that addition of HNO3 was not enough to minimize the suppression of signal in the determination. Since the addition of nitric acid was not enough to eliminate or even to minimize the matrix interferences, other methods that eliminate interferences should be applied in the determination like the use of chemical releasing agents and matrix modifiers such as EDTA, ascorbic acid, phosphoric acid, thiourea, ammonium nitrate and mineral acids[6].
Difference in viscosity of the standard and sample solutions and the nature and amount of sample used were the factors that were not avoided and eliminated in the signal. The difference in the viscosity of the solution used introduced variation in the signal.
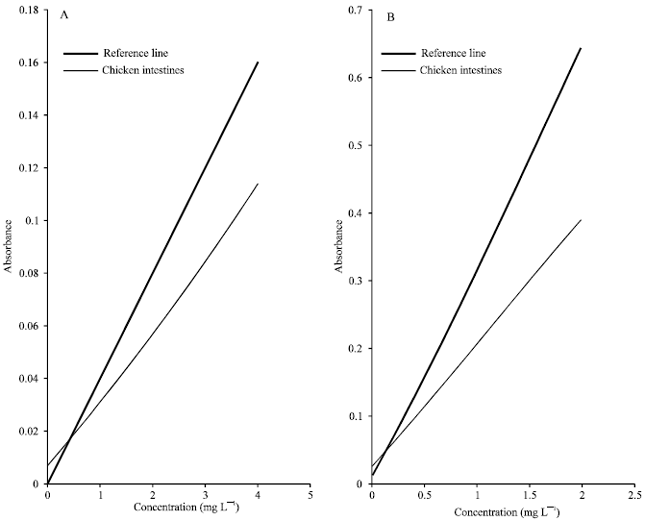 | |
| Fig. 1: | Comparison of standard calibration line and standard addition line for lead (A) and cadmium (B) |
With viscous solutions, the nebulization efficiency of the apparatus is affected. Because viscosity of the solutions was difficult to monitor, suppression contributed by these factors is uncontrollable. Likewise, the digestion method used needed a relatively large amount of sample and considering the nature of the sample and its matrix effects, the sample matrix cannot be minimized. For this reason, the use of smaller amount of sample can be done. Other digestion procedures like microwave digestion can also be used which uses only around 0.5 g of sample.
The standard addition technique can minimize the effect of matrix but the use of this technique has disadvantages. It is too impractical and costly for analysis of a large number of samples. Accordingly, standard addition technique was not applied in recovery assays and sample analysis.
Recovery experiments were performed using chicken’s gizzard. After dilution, the final concentration for low-level concentration of lead and cadmium were 0.08 and 005 mg L-1, respectively. The final concentrations for high level concentrations of lead and cadmium were 0.4 and 0.2 mg L-1, respectively. Percent recoveries of the metals in both levels were calculated and compared using ANOVA. The percent recovery for lead ranged from 65.00 to 115.27% while the percent recovery for cadmium ranged from 68.19 to 102.46%. The percent recoveries for high-level concentration of lead showed significant difference while for low level of lead, no significant difference was observed. For cadmium, both concentration levels showed no significant difference. The percent recoveries for cadmium and lead, considering both the concentration levels were not significantly different.
The differences in the recovery obtained for both lead and cadmium determination in the spiked samples and blank could be attributed to the effect of sample and the digestion used. Since the values of the percent recovery of the samples were not consistent, the nature and type of sample affect the efficiency of the method use. Thus, the sample affects the accuracy of the method. However results of recovery tests in the spiked blanks showed losses that can only be attributed to the method of digestion used.
The LOD obtained for lead and cadmium was 0.080 and 0.0089 ug g-1, respectively. Values obtained below the MOD were considered to be not detected. For the LOQ, the value obtained for lead and cadmium was 0.266 and 0.030 ug g-1, respectively.
Sample analysis: The samples were digested using AOAC Standard Method of Digestion (972.23) with modification. Ashing was difficult since frothing and spattering happened due to the high fat content of the samples. To solve this problem, the samples were carbonized in hot plate to remove the fats and oils prior to ashing.
The amount of lead in the internal organs and blood ranged from 0.082 to 0.164 ug g-1 while for cadmium levels, the concentration ranged from 0.019 to 0.069 ug g-1 with the intestines having the highest concentration of both metals (Table 1). Considering the result from matrix interferences where there was a significant suppression effects, the amount of lead and cadmium obtained were basically lower than the actual values of lead and cadmium in the samples. Thus, the values obtained only serve as a guide on the amount of lead and cadmium in the samples.
Accumulation of lead and cadmium would occur upon their entry to animals like chicken. The bioaccumulation of lead and cadmium in the internal organs and blood that were studied could be related to their functions in the body. Aside from air pollution, significant amount of these metals might be present in the diet of chickens. Consumption of contaminated plants, feeds and water were the main source for lead and cadmium entry in animals. Commercial feed mixture made up of cereals, by-products of cereals, fish products, products from oil plant seeds, by-products from sugar production, minerals, oils and fats, vitamin and amino acids contained 66 ug kg-1 wet weight of cadmium[7]. On the other hand, crops like corn and green bean have high level of lead with 0.938 and 0.209 ug g-1 fresh weight, respectively[2].
The food and material that entered the digestive system of the body are primarily absorbed in the intestine. The gizzards on the other hand, received the food materials and digested them with little absorption. Due to the functions of intestines and gizzard, heavy metals are usually absorbed and stored.
| Table 1: | Mean of lead and cadmium found in liver, gizzard, intestine and blood of chicken |
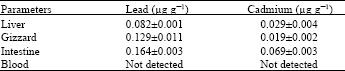 | |
And it is not surprising that the chicken intestines have the highest content of both metals among the edible organs that were analyzed. The chicken gizzard had the next highest lead content while the chicken liver had the next highest cadmium content followed by the chicken gizzard. Liver on the other hand is the main organ where storage, metabolism and biosynthesis activities takes place. It is also the principal site of xenobiotic metabolism. Liver receives all the material that entered the gastrointestinal tract via the portal vein and also exposed to toxic elements. Lastly, although blood absorbs 50% of the lead intake and approximately 0.06% of the total cadmium body burden[8], it was found to contain lead and cadmium levels that were not detected. The result is expected. Although blood is involved in the transport and movement of all the nutrients and toxic materials, these materials in blood are readily exchangeable hence resulting in low value that were not detected by the method used.
Based from the acceptable limit for lead and cadmium[9], which is 0.3 and 0.2 ug g-1, respectively, all samples did not exceed the limit.