Research Article
Incidence of Fig Leaf Mottle-associated Virus and Fig Mosaic Virus in Eastern Province of Saudi Arabia
Department of Agriculture Botany, Plant Pathology Research Institute, Agricultural Research Center, Saudi Arabia
Fig plant, Ficus carica L., Brown Turkey, is grown mainly as individual trees in farm, rarely in specialized orchards in Hofuf, Saudi Arabia. A wide range of discolourations, i.e, various patterns of chlorotic mottling and blotching, vein banding, vein clearing and malformations of the leaves are commonly observed on fig tree throughout Saudi Arabia. Fig mosaic virus is a graft-transmitted disease. It is also transmitted by the eriophyid mite Aceria ficus (Condit and Horne, 1933; Flock and Wallace, 1955). Recent studies have reported that tissues of infected fig leaves have shown particles of this virus those are likely to be intracellular enveloped structures (Bradfute et al., 1970; Castellano et al., 2007; Elbeaino et al., 2007a, b). Other viruses associated with fig mosaic diseases were found such as fig leaf mottle associated virus 1 (FLMaV-1) and fig leaf mottle associated virus 2 (FLMaV-2) that are members of Closteroviridae (Elbeaino et al., 2006; Elbeaino et al., 2007a, b). This study is aimed at identification of such mosaic-causing viruses i.e. FLMaV-1, FLMaV-2 and FMV in Saudi Arabia.
Collection of samples: The main symptoms as described by Castellano et al. (2007) were chlorotic mottling, blotching and various types of leaf deformation. A survey was carried out in autumn seasons of 2008-2009.
 | |
| Fig. 1: | Map of the area where survey has been conducted in Hofuf, Saudi Arabia |
Samples were collected, with consideration of the economic importance and distribution of the cultivars, from different areas of Hofuf Saudi Arabia (Fig. 1). Each sample was consisted of 10-15/leaves. Samples were labeled and stored in plastic bags at 4°C then transferred to the laboratory.
Extraction of total nucleic acids (TNAs): TNAs were extracted from leaf veins and/or cortical scrapings of fig samples, 100 mg of tissue were macerated in 1 mL grinding buffer (6 M guanidine isothiocyanate, 0.2 M sodium acetate, 1 M potassium acetate, 0.025 mM EDTA, 25% PVP-40). TNAs were recovered with a silica-capture procedure (Foissac et al., 2001); stored at -20°C, and then used as described by Elbeaino et al. (2009 a,b).
Molecular detection of Fig mosaic disease in tissue extract: Fig samples were assayed by RT-PCR using the following specific primers (Elbeaino et al., 2006, 2009):
| • | LMaV- N17s: 5’- CGTGGCTGATGCAAAGTTTA-3’ |
| • | N17-a: 5’-GTTAACGCATGCTTCCATGA-3’ |
| • | FLMaV-2 F3-s: 5'- GAACAGTGCCTATCAGTTTGA TTTG-3' |
| • | F3-a: 5'-TCCCACCTCCTGCGAAGCTAG AGAA-3' |
| • | FMV E5-s: 5’-CGGTAGCAAATGGAATGAAA-3’ E5-a |
| • | 5’-AACACTGTTTTTGCGATTGG-3’ |
From 8-10 μL of TNA extract were mixed with 1 μL random hexamer primer, (Boehringer Mannheim, GbmH) (0.5 μg μL-1), denatured at 95°C for 5 min and quickly chilled in ice. Reverse transcription reaction was done for 1 h at 39°C by adding 4 μL M-MLV buffer 5x, 2 μL of 10 mM DTT, 0.5 μL f 10 mM dNTPs, and 200 units Moloney Murine Leukaemia virus (M-MLV) reverse transcriptase (Bethesda Research Laboratories USA) in a final volume of 20 μL. For RT-PCR with the specific primers detection, 2.5 μL cDNA mixture were submitted to PCR amplification by adding 2.5 μL of 10xTaq polymerase buffer (Promega Corporation, USA), 1.5 mM as final concentration of MgCl2, 0.5 μL of 10 mM dNTPs, 0.5 μL of 10 μM forward and reveres of each specific primers, and 0.2 μL of Taq polymerase (5 unit μL-1) in final volume of 25 μL. PCR products were electrophoresed in agarose gels, and visualized by UV transillumination.
Collection of samples: As a survey conducted from different areas of Hofuf, Saudi Arabia; fig orchards and nursery were infected about 4%. Most of nursery fig trees were brought from Lebanon, Jordan and Syria. Symptoms were similar to those described by Castellano et al. (2007), chlorotic mottling, blotching, vein banding, vein clearing and malformations (Fig. 2, A-C). Symptoms were shown very clear in fig trees; however, sometimes in summer season same symptoms were not shown. This indicates that high temperature plays role in the development of these viruses.
Molecular detection of Fig mosaic disease in tissue extract: Fig mosaic disease virus was amplified from leaf samples showing typical symptoms (Castellano et al., 2007). Only infected fig trees were PCR-positive where no PCR products were obtained from healthy fig tree (Martelli et al., 1993; Elbeaino et al., 2007a, b, 2009a, b). RT- PCR assay of leaf extracts of infected fig accession using FLMaV-1 and FMV specific primers had gave positive results and non with FLMaV-2. The three sets of primers amplified DNA fragments of 350 and 302 bp for FLMaV-1 and FMV, respectively (Fig. 3a, b). However, mixed infections were obtained of FLMaV-1 and FMV from Fig mosaic disease (Elbeaino et al., 2007a).
 | |
| Fig. 2(a-c): | (a) Mottle and mosaic symptoms on naturally-infected leaf. (b) Ring spots in fig fruit of naturally infected plant and (c) Mosaic viral symptoms on fig leaf during the heat of summer in Hofuf |
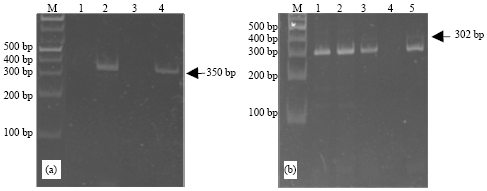 | |
| Fig. 3(a-b): | Gel electrophoresis of RT-PCR assay showing a product of 350 and 302 bp for FLMaV-1 and FMV, respectively. (a) lane, 2 infected samples by FLMaV-1; lanes 1 and 3 healthy samples; lane, 4 positive control. (b) lanes 1, 2, 3 infected samples by FMV; lane 4 negative sample; lane, 5 positive control. M, 1 kb marker |
FLMaV-1 and FMV have been recorded in Italy (Elbeaino et al., 2006, 2009a, b), Albania, Algeria, Syria, Chile, Greece, Mexico and South Africa (Castellano et al., 2007), Lebanese (Elbeaino et al., 2007b) and Tunisa (Nahdi et al., 2006). To our knowledge this is the first record of identification of FLMaV-1 and FMV in Saudi Arabia. Further studies are needed to investigate the fig mosaic disease throughout the country.
This research was supported by Deanship of Scientific Research, King Faisal University, Saudi Arabia. The author thanks Dr. Elbeaino, Istituto Agronomico Mediterraneo, Bari, Italy for his assistance and support.