ABSTRACT
Background and Objective: As one of branch chain amino acids (BCCA), L-leucine may have potential as a regulator of feed intake in broiler chickens. This study aimed to evaluate dietary supplementation of low-protein and low-energy diets with L-leucine to improve the growth performance, body composition and carcass characteristics of broiler chickens. Materials and Methods: About 80 days old broiler chicks (Cobb) were randomly distributed in a completely randomized design consisting of 4 treatments, each group had 4 replicates, with 5 birds/replicate pen in a battery brooder. The birds were fed a starter diet until the 21st day of age and from day 21-42, they were fed experimental diets that had restricted protein and calorie contents (18% and 3,000 kcal kg–1, respectively). Chickens were fed diets supplemented with different levels of L-leucine (0, 0.42, 0.5 and 0.75% kg–1). Results: Supplementation with different doses of L-leucine strongly and dose-dependently increased feed intake and body weight gain and decreased the feed conversion ratio. Dietary L-leucine supplementation also increased live weight, carcass percentage and breast meat as well as the loss of body fat. Conclusion: Supplementing protein and energy-restricted diets with L-leucine can be used to improve growth performance, body composition and carcass characteristics in broiler chickens.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ijps.2018.28.33
URL: https://scialert.net/abstract/?doi=ijps.2018.28.33
INTRODUCTION
The branched chain amino acid (BCAA), L-leucine (Leu) plays multiple roles in body metabolism and is an essential substrate for the synthesis of new proteins1, including being the energy substrate2 precursor for the synthesis of alanine and glutamine3,4 and a modulator of muscle protein synthesis. Furthermore, these roles include being a key regulator of the translation initiation of protein synthesis in skeletal muscle5 as well as enhancing muscle growth6,7, cell growth and metabolism8-11.
Leu is also a potent activator of protein synthesis in white adipose tissue12, which suggests that Leu supplementation may therefore affect the mass of both muscle and fat. Supporting this hypothesis, Leu supplementation has been reported to result in more fat loss during caloric restriction13. Recently, studies have explored the use of high-protein diets or dietary supplementation with BCAAs or Leu to control body weight14,15 and Leu has been found to improve body weight control when coupled with exercise or energy restriction in mice16. Additionally, Leu is known to improve body weight by maintaining muscle mass due to the stimulation of muscle protein synthesis1. Previous studies have revealed orexigenic effects of oral Leu supplementation in mice17 as well as the suppression of muscle protein degradation18,19.
Because few reports have addressed the need for Leu in growing and finishing broiler chickens, as a practical diet for measuring the responses to Leu while promoting good growth has not been elucidated. Furthermore, Leu supplementation of a low-energy diet increases the carcass weight of grower-finisher chickens20, so it would seem reasonable to expect that Leu supplementation of both low-dietary-energy and low-protein diets may support growth.
Currently, no information is available on the effects of Leu supplementation of both low-dietary-protein and low-energy diets on the growth and carcass characteristics of grower-finisher broiler chickens. Therefore, this study was conducted to evaluate dietary supplementation of low-protein and low-energy diets with L-leucine to improve the growth performance, body composition and carcass characteristics of broiler chickens.
MATERIALS AND METHODS
Animals, housing and diets: The study was undertaken following the guidelines of the Research Policy on Animal Ethics of Universiti Putra Malaysia. About 81 days old broiler chicks (Cobb) were randomly assigned to four treatment groups and each group was replicated four times with 5 birds per replicate pen in a battery brooder.
| Table 1: | Percentage ingredient inclusion and calculated composition of the test diets in the experiment |
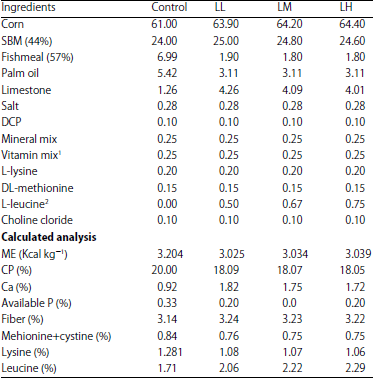 | |
1Premix provided the following per kg or diet: Vitamin A 50.000 MIU, Vitamin D3 10.000 MIU, Vitamin E 75.000, Vitamin K 20.000 g, Vitamin B1 10.000 g, Vitamin B2 30.000 g, Vitamin B6 20.000 g, Vitamin B12 0.100 g, Calcium D-panthothenate 60.000 g, Nicotinic acid 200.000 g, Folic acid 5.000 g, Biotin 235.000 mg | |
All pens were equipped with feeders and water and the floor of each pen measured 0.75 m×0.75 m×0.5 m in height. The birds were fed a starter diet until the 21st day of age followed by experimental diets from days 21-42. This experiment was conducted in accordance with the ethics approval for research projects involving animals and the protocol was approved by the Institutional Animal Care and Use Committee (IACUC), Number 101, Universiti Putra Malaysia. The birds were euthanized by an intravenous injection of sodium and all efforts were made to minimize suffering. Table 1 shows the ingredients and chemical compositions of the four experimental diets, which were formulated to provide 3,200 kcal kg–1 metabolizable energy (ME) as follows: 20% CP without Leu supplementation as the control, which was based on NRC (1994) and 3,000 kcal kg–1 ME, 18% CP supplemented by 0.5% Leu (Diet LL) and 3,000 kcal kg–1 ME, 18% CP supplemented by 0.67% Leu (diet LM) and 3,000 kcal kg–1 ME, 18% CP supplemented by 0.75% Leu (diet LH). Crude protein was determined using the Kjedahl procedure. The experimental diets and fresh water were provided ad libitum.
Growth performance: Body weight (BW) gain and feed consumption were recorded weekly throughout the experiment. Feed consumption was corrected for body weight and mortality, if any, was taken into account. Feed consumption was calculated as the difference between the amount of feed supplied to the birds and the amount of feed that remained at the end of each feeding period and BW gain was calculated as the difference between the final and initial weight of the birds during each of the weighing periods. Feed intake and weight gain was recorded at days 21-42 and the feed conversion ratio (FCR) was calculated as the ratio of feed consumption to weight gain for each period. On the 42nd day of the experiment, six birds from each treatment were randomly selected, slaughtered and defeathered and their viscera were removed to determine the responses of the carcasses to the dietary treatments. The internal organs were also removed and weighed.
Carcass characteristics: The carcass weight, abdominal fat and organ weight, i.e., heart, liver and gizzard, were recorded after slaughter. The carcass weight measurements were performed after defeathering and the removal of the feet, head and viscera. The abdominal fat included the leaf fat surrounding the cloaca and the abdominal muscles, excluding the fat surrounding the gizzard.
Carcass composition: To evaluate the carcasses, ten birds (1 bird/replicate) were randomly chosen from each treatment group and slaughtered at the end of the experiment. Cold carcass weights were calculated after keeping the carcasses at -41°C for 18 h and cold carcass yield was calculated as the cold carcass weight divided by the body weight at slaughter. The physical carcass composition was determined after separation into the lean meat, fat and bones and the weights of the pectoralis major and minor muscles, which were dissected for sensory analysis, were also recorded and included as lean in the analysis of carcass composition. Finally, the carcass without the pectoralis major and minor muscle was dissected to detach the lean, fat and bone.
Statistical analysis: One-way analysis of variance (ANOVA) based on a completely randomized design was applied to statistically analyze the food intake during the experiments. Statistical significance was set at p<0.05 And values were presented as the Mean±SEM. Regression analysis was also performed to determine the correlations between the Leu doses and feed intake, body weight gain and FCR. Correlation equations were similarly obtained for carcass characteristics. The data were analyzed using the statistical analysis system (SAS)21. All data from each group were first subjected to a Thompson rejection test to eliminate outliers (p<0.01) and the remaining data were used for the comparisons among groups.
RESULTS
Food intake and growth: The effect of supplementation with several doses of Leu on the growth performance of chickens from 21-28 days of age is presented in Table 2. A significant effect on food intake was observed among treatments from 21-28 days of age (F [3, 15] = 10.32, p<0.001) and it was significantly positively correlated with the administered doses (p<0.002, R2 = 0.668). Supplementation with Leu significantly (F [3, 15] = 15.44, p<0.0001) increased BW gain, which was also significantly positively correlated with the administered doses (p<0.005, R2 = 0.617). Moreover, Leu supplementation significantly (F [3, 16] = 11.36 p<0.0003) decreased FCR and a significant negative correlation with the administered doses was observed (p<0.0001, R2 = 0.736).
Table 3 shows the effects of supplementation with different doses of Leu on growth performance from 28-35 days of age.
A significant increase in food intake was detected among the treatments from 28-35 days of age (F [3, 14]=4.19. p<0.05). Supplementation with Leu significantly (F [3, 12] = 20.59, p<0.0001) increased BW gain, which was significantly positively correlated with the administered doses (p<0.05, R2 = 0.531). Moreover, Leu significantly decreased (F [3, 12] = 15.33, p<0.0002) FCR and a significant negative correlation was observed between that parameter and the administered doses (p<0.001, R2 = 0.756).
| Table 2: | Effects of supplementation of several doses of Leu on growth performance of chickens during 21-28 days of chickens |
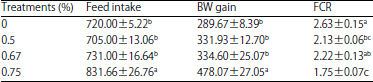 | |
Means with different superscripts within the same column were significantly different at p<0.05, Values are Mean±SEM | |
| Table 3: | Effects of supplementation of different doses of Leu growth performance during 28-35 days-old in chickens |
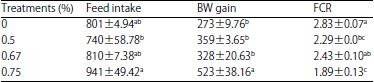 | |
Means with different superscripts within the same column were significantly different at p<0.05, Values are Mean±SEM | |
The impact of the Leu gradations on growth performance of growing-finishing broilers is shown in Table 4. A significant increase in food intake was detected among treatments from 35-42 days of age (F [3, 13] = 3.71, p<0.05) and Leu supplementation with Leu significantly (F [3, 12] = 11.25, p<0.0008) increased BW gain, which was significantly positively correlated with the administered doses (p<0.0001, R2 = 0.797). Moreover, Leu significantly decreased (F [3, 11] = 15.76, p<0.0003) FCR and a significant negative correlation was observed between this parameter and the administered doses (p<0.0001, R2 = 0.863).
Carcass characteristics: The carcass characteristics are listed in Table 5. Increasing doses of Leu significantly (F [3, 53] = 55.17, p<0.0001) increased live weight gain and significant (F [3, 54] = 24.03, p<0.0001) increases in dressing percentages were also observed. Significant (F [3, 53] = 6.51, p<0.005) decreases in the amount of abdominal fat were observed for the Leu groups compared with the control group and a significant increase was also observed in breast meat yield (F [3, 51] = 75.11, p<0.0001) and gizzard weight (F [3, 54] = 15.86, p<0.0001). No significant differences in heart weight were observed among groups.
| Table 4: | Effects of supplementation of several doses of Leu growth performance during 35-42 days-old in chickens |
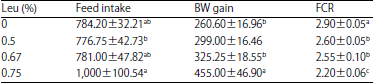 | |
Means with different superscripts within the same column were significantly different at p<0.05. Values are Mean±SEM | |
Carcass composition: The chemical compositions of the carcasses are listed in Table 6. Leu supplementation significantly (F [3, 54] = 68.53, p<0.0001) increased lean and bone (F [3, 53] = 11.58, p<0.0001) and significant (F [3, 54] = 9.59, p<0.0001) decrease in the amount of abdominal fat was observed in the Leu groups compared with the control group. No significant difference in the amount of skin was observed among groups.
DISCUSSION
One of the major results of the present experiment was the interaction between the level of Leu supplementation and growth performance; a high level of Leu supplementation strongly induced growth performance, improved carcass characteristics and decreased the fat content in the muscles. Chicks receiving the Leu-supplemented diet increased their body weight from 21-42 days of age. Previous studies in humans concluded that Leu and BCAA improved body weight when coupled with energy restriction19. Other supporting data include the stimulation of food intake by the intracerebroventicular (i.c.v) administration of Leu at 30 min22 and 2 h post injection in neonatal chicks23. The current study did not provide information as to how Leu modulates feed intake by chickens, but some hypotheses may be proposed.
| Table 5: | Effects of supplementation of several doses of Leu in low energy and protein diets (42 days of age) on carcass composition during 21-42 days old in chickens |
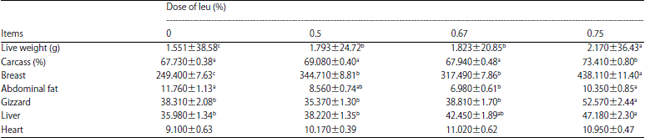 | |
Means with different superscripts within the same row were significantly different at p<0.05, Values are Mean±SEM | |
| Table 6: | Effects of supplementation of several doses of Leu in low energy and protein diets (42 days of age) on body composition in chickens |
 | |
Means with different superscripts within the same row were significantly different at p<0.05. Values are Mean±SEM | |
First, a Leu metabolite may increase feed intake. In the metabolic pathway, Leu is transaminated to produce glutamate and α-keto isocaproic acid, which is converted to acetoacetyl-CoA in the brain24. Secondly, glutamate resulting from Leu supplementation may stimulate feeding behavior. Recently, Reddy et al.25 revealed that BCAAs, particularly Leu, are important as external sources of nitrogen for the synthesis of glutamate and glutamine. Additionally, numerous studies involving pharmacological treatments have investigated the role of glutamate in the regulation of appetitive behavior in mammals and i.c.v. administration or local administration of glutamate26,27 or the glutamate agonist, NMDA, has been shown to induce hyperphagia. Hence, it is possible that the orexigenic effect of Leu was due to an acceleration of glutamate synthesis. The results of this study suggest that Leu induces hyperphagia and thus may play an important role in the feeding behavior of chickens. Third, Leu may also increase feed intake by increasing gastric emptying or by competing with tryptophan, a precursor of serotonin, to cross the blood–brain barrier. The information mentioned above and the results of the present study suggest that central or oral supplementation with Leu has similar effects on food intake in neonatal and growing-finishing chickens. However, further experiments will be required to determine the mechanism underlying how Leu affects the ingestive behavior of broiler chickens.
Moreover, the current study showed that increasing dietary Leu from 0.5-0.75% increased the lean component compared to the control, but it also decreased abdominal fat. One hypothesis is that Leu supplementation might have contributed to an increase in the mass of adipose tissue. First, chickens in the Leu group showed increased feed intake in the first 3 weeks, which was concomitant to their weight gain. Another hypothesis that might explain the smaller amount of body fat in the supplemented group is related to the effect of Leu on the increase in protein synthesis and/or turnover8,12,28. A higher rate of protein synthesis might increase energy expenditure and this additional energy demand can mainly be satisfied by the oxidation of body fat.
Recently, breast muscle growth has become an interesting parameter because of the high economic value of this portion of the poultry carcass29,30. Breast muscle is 30% of the total edible meat and 60% of the edible protein in the carcass30. The data from the experiments in this study indicate that increasing dietary Leu supplementation increased breast muscle growth.
There is evidence that Leu supplementation has a beneficial effect on breast meat and abdominal fat. In a previous study, Erwan et al.31 demonstrated that Leu supplementation of a low-energy diet decreased abdominal fat and in the current study it is demonstrated that Leu decreased the abdominal fat of chickens fed low-energy and low-protein diets. These findings agree with those of Donato et al.13, who demonstrated that Leu supplementation at 5.9 g kg–1 reduced body fat by 25% compared to the control in rats. This phenomenon has also been demonstrated in humans.
CONCLUSION
Overall, this study indicates that Leu supplementation of protein and energy-restricted diets may increase the growth performance and improve the carcass composition of broiler chickens, which indicates that Leu can be used as an alternative food supplement for broiler chickens. However, further experiments will be required to determine the mechanism underlying the effect of Leu on the ingestive behavior of chickens.
ACKNOWLEDGMENT
This study was supported by the Research University Grant Scheme (RUGS) of Universiti Putra Malaysia.
REFERENCES
- Layman, D.K. and D.A. Walker, 2006. Potential importance of leucine in treatment of obesity and the metabolic syndrome. J. Nutr., 136: 319S-323S.
Direct Link - Harper, A.E., R.H. Miller and K.P. Block, 1984. Branched-chain amino acid metabolism. Annu. Rev. Nutr., 4: 409-454.
CrossRefPubMedDirect Link - Ahlborg, G., P. Felig, L. Hagenfeldt, R. Hendler and J. Wahren, 1974. Substrate turnover during prolonged exercise in man: Splanchnic and leg metabolism of glucose, free fatty acids and amino acids. J. Clin. Invest., 53: 1080-1090.
CrossRefDirect Link - Ruberman, N.B., 1975. Muscle amino acid metabolism and gluconeogenesis. Ann. Rev. Med., 26: 245-258.
CrossRefDirect Link - Zeanandin, G., M. Balage, S.M. Schneider, J. Dupont, X. Hebuterne, I. Mothe-Satney and D. Dardevet, 2012. Differential effect of long-term leucine supplementation on skeletal muscle and adipose tissue in old rats: An insulin signaling pathway approach. Age, 34: 371-387.
CrossRefDirect Link - Yoshizawa, F., T. Kido and T. Nagasawa, 1999. Stimulative effect of dietary protein on the phosphorylation of p70 S6 kinase in the skeletal muscle and liver of food-deprived rats. Biosci. Biotechnol. Biochem., 63: 1803-1805.
CrossRefDirect Link - Smith, K., N. Reynolds, S. Downie, A. Patel and M.J. Rennie, 1998. Effects of flooding amino acids on incorporation of labeled amino acids into human muscle protein. Am. J. Physiol., 275: 73-78.
Direct Link - Anthony, J.C., T.G. Anthony, S.R. Kimball and L.S. Jefferson, 2001. Signaling pathways involved in translational control of protein synthesis in skeletal muscle by leucine. J. Nutr., 131: 856S-860S.
Direct Link - Gingras, A.C., B. Raught and N. Sonenberg, 2001. Regulation of translation initiation by FRAP/mTOR. Genes Dev., 15: 807-826.
CrossRefDirect Link - Kimball, S.R. and L.S. Jefferson, 2006. Signaling pathways and molecular mechanisms through which branched-chain amino acids mediate translational control of protein synthesis. J. Nutr., 136: 227S-231S.
Direct Link - Inoki, K., M.N. Corradetti and K.L. Guan, 2005. Dysregulation of the TSC-mTOR pathway in human disease. Nat. Genet., 37: 19-24.
CrossRefDirect Link - Lynch, C.J., S.M. Hutson, B.J. Patson, A. Vaval and T.C. Vary, 2002. Tissue-specific effects of chronic dietary leucine and norleucine supplementation on protein synthesis in rats. Am. J. Physiol.-Endocrinol. Metab., 283: E824-E835.
CrossRefDirect Link - Donato, Jr. J., G.P. Rogerio, F.C. Vinicius, S.O.P. Ivanir and T. Julio, 2006. Effects of leucine supplementation on the body composition and protein status of rats submitted to food restriction. J. Nutr., 22: 520-527.
CrossRefDirect Link - Lynch, C.J., B. Gern, C. Lloyd, S.M. Hutson, R. Eicher and T.C. Vary, 2006. Leucine in food mediates some of the postprandial rise in plasma leptin concentrations. Am. J. Physiol. Endocr. Metab., 291: 621-630.
CrossRefDirect Link - Halton, T.L. and F.B. Hu, 2004. The effects of high protein diets on thermogenesis, satiety and weight loss: A critical review. J. Am. Coll. Nutr., 23: 373-385.
PubMedDirect Link - Nairizi, A., P. She, T.C. Vary and C.J. Lynch, 2009. Leucine supplementation of drinking water does not alter susceptibility to diet-induced obesity in mice. J. Nutr., 139: 715-719.
Direct Link - Zhang, Y., K. Guo, R.E. LeBlanc, D. Loh, G.J. Schwartz and Y.H. Yu, 2007. Increasing dietary leucine intake reduces diet-induced obesity and improves glucose and cholesterol metabolism in mice via multimechanisms. Diabetes, 56: 1647-1654.
CrossRefDirect Link - Buse, M.G. and S.S. Reid, 1975. Leucine. A possible regulator of protein turnover in muscle. J. Clin. Invest., 56: 1250-1261.
CrossRefDirect Link - Mourier, A., A.X. Bigard, E. De Kerviler, B. Roger H. Legrand and C.Y. Guezennec, 1997. Combined effects of caloric restriction and branched-chain amino acid supplementation on body composition and exercise performance in elite wrestlers. Int. J. Sports Med., 18: 47-55.
CrossRefPubMedDirect Link - Erwan, E., A.R. Alimon, A.Q. Sazili and H. Yaakub, 2008. Effect of varying levels of leucine and energy on performance and carcass characteristics of broiler chickens. Int. J. Poult. Sci., 7: 696-699.
CrossRefDirect Link - Izumi, T., K. Kawamura, H. Ueda and T. Bungo, 2004. Central administration of leucine, but not isoleucine and valine, stimulates feeding behavior in neonatal chicks. Neurosci. Lett., 354: 166-168.
CrossRefDirect Link - Wang, S., P. Khondowe, S. Chen, J. Yu and G. Shu et al., 2012. Effects of “Bioactive” amino acids leucine, glutamate, arginine and tryptophan on feed intake and mRNA expression of relative neuropeptides in broiler chicks. J. Anim. Sci. Biotechnol., Vol. 3.
Direct Link - Bixel, M.G., S.M. Hutson and B. Hamprecht, 1997. Cellular distribution of branched-chain amino acid aminotransferase isoenzymes among rat brain glial cells in culture. J. Histochem. Cytochem., 45: 685-694.
CrossRefDirect Link - Reddy, V.M., S.S. Meharg and S. Ritter, 1986. Dose-related stimulation of feeding by systemic injections of monosodium glutamate. Physiol. Behav., 38: 465-469.
CrossRefDirect Link - Wandji, S.A., J.R. Seone, A.G. Roberge, L. Bedard and L. Thibault, 1989. Effects of intrahypothalamic injections of GABA, muscimol, pentobarbital and L-glutamic acid on feed intake of satiated sheep. Can. J. Physiol. Pharmacol., 67: 5-9.
PubMedDirect Link - Ritter, S. and S.L. Stone, 1987. Area postrema lesions block feeding induced by systemic injections of monosodium glutamate. Physiol. Behav., 41: 21-24.
CrossRefDirect Link - Patti, M.E., E. Brambilla, L. Luzi, E.J. Landaker and C.R. Kahn, 1998. Bidirectional modulation of insulin action by amino acids. J. Clin. Inves., 101: 1519-1529.
CrossRefDirect Link - Hickling, D., W. Geuenter and M.E. Jackson, 1990. The effects of dietary methionine and lysine on broiler chicken performance and breast meat yield. Can. J. Anim. Sci., 70: 673-678.
CrossRefDirect Link - Summers, J.D. and S. Leeson, 1985. Broiler carcass composition as affected by amino acid supplementation. Can. J. Anim. Sci., 65: 717-723.
CrossRefDirect Link - Erwan, E., A.R. Alimon, A.Q. Sazili, H. Yaakub and R. Karim, 2011. Effects of levels of L-leucine supplementation with sub optimal protein in the diet of grower-finisher broiler chickens on carcass compositionand sensory characteristics. Asian-Aust. J. Anim. Sci., 24: 650-654.
Direct Link








