ABSTRACT
Background: The eventual switch to light-emitting diode (LED) light fixtures as the standard in the poultry industry has resulted in the need to reevaluate the standard management practices relating to lighting. Current management practices utilized by the industry are based on research that used lighting fixtures no longer used and therefore may no longer be relevant. One such practice is to raise broiler chickens under dim lighting to maximize growth and minimize stress and fear. Objective: To determine if broilers could be reared under brighter light using LEDs. Methodology: To investigate whether birds could be raised under brighter conditions using LED lighting compared to the traditional incandescent light without negatively affecting growth, feed conversion, stress and fear broilers (n = 120 per treatment) were raised under incandescent (INCAN) or LED lighting at either dim (5 lux) or bright (20 lux) intensity. Growth, feed conversion, fear and stress susceptibility were all quantified. Results: The LED-bright broilers weighed more after 45 days than all other treatments (p<0.05). The INCAN treatments overall had lower 45 days weights and higher 45 days feed conversion than the LED treatments (p<0.05). Intensity did not affect weights or feed conversion. The INCAN treatments exhibited more fear than the LED birds though the INCAN-dim birds behaved similarly to the LED treatments in tonic immobility. The INCAN treatments showed more stress susceptibility than the LED treatments (p<0.05). Conclusion: These results indicate that it is possible to raise broilers under brighter light when using LEDs without lowering growth or efficiency and still attaining the positive benefits of LED lighting on fear and stress.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ijps.2016.425.431
URL: https://scialert.net/abstract/?doi=ijps.2016.425.431
INTRODUCTION
The poultry industry is currently undergoing a switch from traditional lighting sources like incandescent and fluorescent to Light Emitting Diode (LED). Benefits of this switch include decreased energy usage, increased bird performance and improved bird welfare via reduced fear and stress1-4. With this switch to a new and very different light source than historically used in the rearing of poultry it is important to review management recommendations. Current management recommendations are based on research utilizing lighting such as incandescent and fluorescent. The practice of raising broilers under dim lighting is one example of a management practices based on outdated research.
Lighting levels in the U.S.A are often kept below 10 lux in commercial broiler houses while the Commission of the European Communities5 restricts the use of low intensities (<20 lux). Low light intensity is used to increase feed efficiency and keep birds calm6,7. High intensity light has been observed to lower body weight and poorer feed conversion8-10. However, numerous other studies have shown that intensity of light has little effect on food intake, final body weight and feed conversion8,11-14. The problem with all this study is that it utilized lighting types other than LEDs making it possibly irrelevant as the industry moves to LEDs.
Recent study has shown that growth and feed conversion can be affected by lighting source type. Rozenboim et al.15 and Zimmermann16 observed that there were differences between even types of fluorescent bulbs and incandescent bulbs in body weights of broilers. Mendes et al.17 found that birds raised under LEDs performed better overall than birds reared under CFLs. Using halogen lighting has resulted in greater live weight than incandescent light18. The LED lighting has also been shown to improve weight gain and feed conversion over CFL lighting and incandescent lighting3,4. This makes it necessary to reevaluate current management of lighting if LEDs are being used.
Furthermore, different lighting types and intensities have been shown to affect fear and stress in poultry3,4. Fear has also been shown to be affected by different spectra of light and given that spectral output can vary drastically from light source to light source19,20. Low light intensities also result in broilers having more interrupted sleep which could lead to increased stress21, however, it has been observed that light intensity did not affect heterophil/lymphocyte ratios or corticosterone concentrations bothwhich are measures of stress22,23. Bright lighting (20 lux) has been shown to increase fear in broilers but this study used incandescent lighting24.
Since current management practices of light intensity are based on research that used older lighting technology there is a need to reevaluate. The objective of this study was to evaluate how light intensity may have differential effects when using incandescent or LED bulbs. Specifically it compared stress, fear and production to best determine how light intensity when using incandescent or LED may differently affect bird behavior, performance and efficiency. It is hypothesized that the use of LEDs at higher intensities will result in similar or lower growth and stress susceptibility when compared to dim incandescent lighting.
MATERIALS AND METHODS
Animals and husbandry: This experiment involved 4 treatments: Overdrive (LsA19DIM 5000K) LEDs at high and low intensity and General Electric Incandescent (60 W) at high and low intensity. A comparison of spectra between these bulbs can be seen in Fig. 1. Each treatment consisted of 6 pens containing 20 cobb broiler chicks each in a light tight room outfitted with one of the light sources. Each of the 4 rooms utilized was set up in an identical pattern, with the only difference being the light bulbs in the fixtures or the intensity of light. The room measured 8.1×5.8 m, constructed of thick concrete walls and sealed to prevent any outside light from entering. Ventilation was provided by a single fan on the North end of the room exhausting air, which created negative pressure in the room and drew air in through cooling pads on the South wall. Each of the pens measured 1 m wide, 2 m long and 0.6 m high (stocking density 1.47 kg m–2). The pens were constructed of solid black plastic on all but the front side, which was made of mesh wire. The birds were managed according to the guidelines set forth in the Guide for the Care and Use of Agricultural Animals in Research and Teaching25 and methods were approved by the Texas A and M institutional animal care and use committee. The pens were lined with several inches of pine shavings. One feeder and a single row of 6 nipple drinkers were provided per pen and adjusted for height as the birds grew. All feed was weighed and recorded (Ohaus Champ CD-11, Pine Brook, NJ) and the residual feed at the end of the study was subtracted from the total. There were 6 light fixtures in each room with overhead lighting and 4 of them were directly over the pens 3 m above the floor. All lights were connected to a single dimmer and timer per room. For the 10 days all birds were given 23 L:1 D at 20 lux of light as measured at bird head height using a light meter (Extech 401027, Extech Instruments, Nashua, NH).
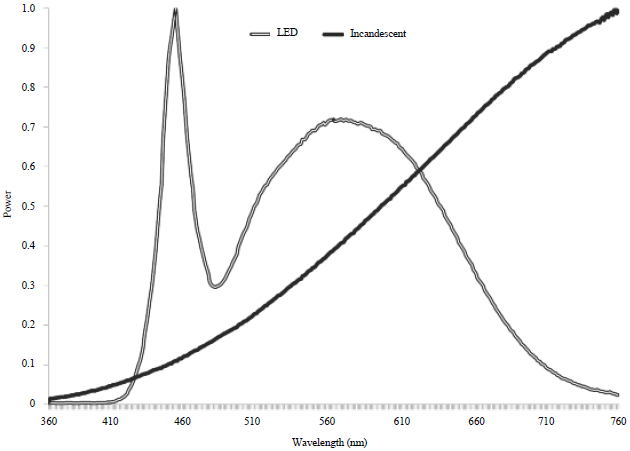 | |
| Fig. 1: | Spectral output of the LED and incandescent light fixtures used in this study |
For the rest of the trial the lights were dimmed down to 5 lux and 20 L:4 D which are commonly used by commercial poultry producers in the United States in one LED (LED-DIM) and one incandescent room (INCAN-DIM) while the other two rooms remained at 20 lux (LED-Bright and INCAN-Bright). For the first 3 weeks, heat was provided by a single ceramic heat lamp hung in each pen which produces no visible light. Upon conclusion of the study, all birds were euthanized with a mixture of air and CO2.
Growth and feed conversion: The birds in each pen were weighed at day 0 and day 45 and body weight gain was calculated by subtracting day 0 weight from day 45 weights. All pens had the same initial starting weight. Feed was weighed before it was added to the feeder in each pen and residual feed was weighed back on bird weigh days so that feed intake could be calculated. Feed Conversion Ratio (FCR) was calculated by dividing the total feed intake per pen by the total body weight gain per pen and was corrected for mortality.
Fear tests: When fear testing began at 10 days of age, 10 birds were selected from each pen and marked with a different colored livestock paint on each wing so individual birds could be identified. The color sequences were: (left wing/right wing) pink/pink, green/green, yellow/yellow, black/black, orange/orange, pink/green, green/yellow, yellow/black, black/orange and orange/green. These patterns were used in every pen in every treatment to insure that no effect of marking the birds would affect the results.
Isolation: The isolation tests were performed 10 days of age by collecting the 10 marked birds from a pen, bringing them to a separate room and placing them in a holding container. The birds were then individually placed in an unlidded 19 L bucket. A timer was set for 3 min and the number of vocalizations produced by the bird during this time was counted. Afterward, the bird was placed in a separate holding container. After all 10 birds had been tested, they were returned to their pen and the 10 marked birds from the next pen were collected and tested. Modified from methods outlined by Archer and Mench26, more vocalizations was considered to indicate more fearfulness27.
Tonic immobility: Tonic Immobility (TI) was conducted at 40 days of age by again collecting the 10 marked birds from a pen, bringing them to a separate room and placing them in a holding container. Methods were modified from previous research by Archer and Mench26. About 21 cm wide by 22 cm high by 30 cm long wooden cradle with the sides sloping out at a 108° angle from the base was obtained, covered in a black cloth and placed on a table. Each bird was individually taken and placed on its back in the cradle. The head of the bird was covered with one hand while the breast was held with the other for approximately 15 sec to induce tonic immobility, after which time contact was removed and a timer was started. If the bird righted itself in under 15 sec, the timer was reset and the above procedure was performed again. If again the bird righted in under 15 sec, it was recorded as a time of 0. Otherwise the time of first head movement and time of righting (or attempting to right) was recorded, with a maximum of 10 min. After all 10 birds had been tested they were returned to their pen and the 10 marked birds from the next pen were collected and tested. Longer times to first head movement and righting were considered to indicate more fearfulness28.
Stress measures: At 44 days of age blood samples were collected from 20 birds per treatment. The area around the wing vein was sanitized with 70% alcohol and in preparation, the inside of a 3 mL syringe was lined with a small amount of heparin. Between 1-2 mL of blood were collected from each bird. The blood was injected into a plasma separation gel and lithium heparin vaccutainer (BD 368056, BD, Franklin Lakes, NJ), which was temporarily stored in an ice bath. Once all samples had been taken, the vaccutainers were spun down in a Beckman GS-6R centrifuge (Beckman Coulter, Brea, CA) for 15 min at 4000 rpm to separate the cells from the plasma. The plasma was poured off into 2 mL microcentrifuge tubes and stored at -19°C until further analysis. Plasma corticosterone concentrations were measured using a commercially available ELISA kit (Enzo Life Sciences, ADI-901-097, Farmingdale, NY). The inter and intra-assay CV (%) were both under 5%.
Physical asymmetry of each marked bird was measured at 45 days, immediately after each was euthanized using a CO2/air mixture and before rigor mortis began to set in, following the protocol outlined by Archer and Mench29. Using a calibrated Craftsman IP54 Digital Caliper (Sears Holdings, Hoffman Estates, IL), the middle toe length, metatarsal length and metatarsal width were measured for both the right and left legs. The composite asymmetry score was calculated by taking the sum of the absolute value of left minus right of each trait, then dividing by the total number of traits. Thus the formula for this trial would be (|L-R|MTL+|L-R|ML+|L-R|MW)/ 3 = Composite asymmetry score.
Statistical methods: To investigate treatment effects on composite asymmetry, corticosterone, isolation, inversion, tonic immobility, weight gain and feed conversion using the GLM procedure was used with lighting type, light intensity and lighting type×light intensity as factors. The least significant difference test was used to test all planned comparisons. All of the assumptions were tested (Shapiro-Wilk test for normality, Levene’s test for homogeneity of variance). No transformations were needed to meet assumptions. All analyses were performed using SAS 9.3 for Windows (SAS Institute Inc.). Significant differences were at p<0.05.
RESULTS
Growth and feed conversion: There was an effect of lighting treatment on 45 day weight and 45 day feed conversion (Table 1). The LED-bright broilers (3.07±0.03 kg) weighed more after 45 days than all other treatments (pooled mean = 2.92±0.03 kg, p<0.05). The LED treatments overall weighed more (3.02±0.03 kg, p = 0.004) than incandescent birds (2.90±0.03 kg). There was no difference observed in intensity or the interaction of lighting type and intensity (p>0.05) for 45 day weight. The INCAN treatments overall had higher 45 day feed conversion (1.48±0.02) than the LED treatments (1.42±0.02, p = 0.05). There was no difference in feed conversion observed for intensity or the interaction of the two in 45 day feed conversion (p>0.05).
Fear response: The number of vocalizations in response to isolation at 10 days of age was affected by treatments (Table 1). The LED treatments vocalized less (47.3±3.8 vocalizations/3 min) than the INCAN treatments (65.8±5.3 vocalizations/3 min, p = 0.005).
| Table 1: | Forty five days weight (kg), 45 day feed conversion (FCR), No. of vocalizations during isolation (Number/3 min), latency to right during tonic immobility (sec), composite asymmetry score (mm) and plasma corticosterone concentrations (pg mL–1) of broilers raised under either incandescent or LED lights and either at a light intensity of bright (20 lux) or dim (5 lux), Means±SE |
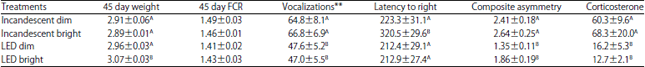 | |
| A,BDifferent superscripts within column significantly different p<0.05, **At time of testing 10 days of age dim and bright were both at 20 lux of light intensity | |
The latency to right during the tonic immobility test was affected by treatments (Table 1). The INCAN-bright broilers had longer latencies to right (320.5±29.6 sec) than all other treatments (pooled mean = 219.2±9.2 sec, p<0.02). Overall INCAN treatments had longer latency to right (271.9±21.8 sec) than LED treatment (212.7±19.9 sec, p = 0.04). There was no difference observed between light intensities overall (p>0.05).
Stress response: The composite asymmetry scores were affected by treatments (Table 1). The INCAN treatments had higher composite asymmetry (2.52±0.15 mm) than the LED treatments (1.61±0.11 mm, p<0.001). Bright treatments had higher composite asymmetry (2.25±0.16 mm) than the dim treatments (1.88±0.12 mm, p = 0.05). There was no interaction of lighting type and intensity (p = 0.47). The plasma corticosterone concentrations were affected by treatment (Table 1). The INCAN treatments had higher plasma corticosterone concentrations (64.7±11.6 pg mL–1) than the LED treatments (14.5±2.8 pg mL–1, p<0.001). There was no effect of intensity or the interaction of lighting type and intensity (p>0.05).
DISCUSSION
This study set out to determine if the current knowledge base on how intensity of light affects broilers was still valid when using LED lights. To do so the study utilized a traditional light source incandescent lighting and compared it to LED lighting using a dim light level commonly used in commercial poultry houses in the United States and a brighter intensity required by the European Union. Overall these results demonstrated that LED lights can be used at higher intensities without negatively impacting production or animal welfare.
Light intensity did not affect final weights within incandescent or LED treatments. This disagrees with the common thinking and research that brighter lighting results in birds that weigh less at the end of production8,9. However, it does agree with other previous research that had observed no difference in body weights when broilers were raised under different light intensities8,11-13. No differences in feed conversion were observed between light intensities in this study. This again disagrees with the idea and research that brighter light results in poorer feed conversion9,10. However, it again Blatchford et al.14 which saw no difference in feed conversion in relation to light intensity. These results indicate that broilers could be raised at 20 lux without losing body weight or feed conversion.
The birds raised under incandescent lighting grew to a lighter final weight at 45 days of age when compared to those reared under LED lighting. This agrees with Archer3 which observed that birds reared under LED lighting ended up heavier at 45 days of age compared to those raised under incandescent lighting. It is also important to note that others have documented that lighting type can affect growth of broilers during production4,17,18. The fact that lighting type can impact growth is often overlooked but as evidenced by this research and others it is clear that it must be considered to optimize weight gain. Feed conversion was not affected by lighting type in this study.
It appears from the results observed in this study that light intensity is only important in the managing of fear in broilers when using incandescent lighting and not LED lighting. At the early time point (10 days of age) all the broilers were at the same light intensity but the broilers housed under incandescent lighting exhibited more fear during the isolation test than those housed under LED lighting. Furthermore, at 45 days of age the LED treatments did not differ in tonic immobility response from each other or the INCAN-dim treatment. So light intensity did not matter when using LEDs but did matter when using incandescent lighting as the broilers raised under high intensity incandescent lighting were the most fearful. This agrees with Robles24 which also saw that high intensity incandescent lighting resulted in more fearful birds than low intensity incandescent lighting. It also agrees with previous research that observed birds raised under LEDs showing less fear than those raised under other lighting types3,4.
Light intensity did not affect the stress susceptibility of broilers in this study. It was thought that possible the light intensity would affect stress as it has been observed that light intensity can affect resting ability21, however, in this study the dark period was total darkness unlike previous research which had 1 lux of light at night. This may explain why there was no effect of light intensity on stress. These results of this study do agree though with other studies which saw no effect of light intensity on stress measures of broilers22,23.
Light type did affect stress susceptibility in this study. The LED treatments had lower composite asymmetry scores and lower corticosterone concentrations when compared to the incandescent treatments. This is similar to previous research which saw the same effect of lighting type on broilers3. This effect is likely do to the type of light that each bulb type is emitting. Spectral outputs vary greatly between lighting types and it is theorized that the LEDs emit a less stressful light for poultry than other types of lighting20. Therefore, by using LEDs in poultry production birds can be raised under less stressful and more animal welfare friendly conditions.
Based on the results of this study the current management practices of rearing broilers under dim light (<10 lux) appears to be outdated when using LED lights. The LED lights had previously been observed to result in calmer, less stress susceptible birds and this study furthers that demonstrated even under brighter lighting this still holds true when compared to incandescent lighting. This is an important finding as it allows producers to raise birds under brighter light which is more accepted by the public, animal welfare groups and even the European Union. As it was hypothesized the current scientific literature on light intensity and its effect on broiler chickens does not apply when using LEDs. This opens the question on whether other concepts such as photoperiod or timing of lighting need to be reevaluated as well. These questions merit future study to ensure that the industry is maximizing management of lighting to improve production and animal welfare.
CONCLUSION
The LED lighting differs greatly from previous technology and therefore requires study like this study to optimize it use in poultry production.
SIGNIFICANCE STATEMENTS
| • | LED lighting at 20 lux did not have detrimental effects on fear or stress |
| • | Incandescent lighting at 20 lux and in general resulted in more stress than LED lighting or dim lighting |
| • | Broilers can be reared under brighter lighting using LEDs without reduced production or welfare |
REFERENCES
- Benson, E.R., D.P. Hougentogler, J. McGurk, E. Herrman and R.L. Alphin, 2013. Durability of incandescent, compact fluorescent and light emitting diode lamps in poultry conditions. Applied Eng. Agric., 29: 103-111.
CrossRefDirect Link - Archer, G.S., 2015. Comparison of incandescent, CFL, LED and bird level LED lighting: Growth, fear and stress. Int. J. Poult. Sci., 14: 449-455.
CrossRefDirect Link - Huth, J.C. and G.S. Archer, 2015. Comparison of two LED light bulbs to a dimmable CFL and their effects on broiler chicken growth, stress and fear. Poult. Sci., 94: 2027-2036.
CrossRefDirect Link - Prescott, N.B., C.M. Wathes and J.R. Jarvis, 2003. Light, vision and the welfare of poultry. Anim. Welfare, 12: 269-288.
Direct Link - Charles, R.G., F.E. Robinson, R.T. Hardin, M.W. Yu, J. Feddes and H.L. Classen, 1992. Growth, body composition and plasma androgen concentration of male broiler chickens subjected to different regimens of photoperiod and light intensity. Poult. Sci., 71: 1595-1605.
PubMedDirect Link - Lien, R.J., J.B. Hess, S.R. McKee and S.F. Bilgili, 2008. Effect of light intensity on live performance and processing characteristics of broilers. Poult. Sci., 87: 853-857.
CrossRefPubMedDirect Link - Deaton, J.W., F.N. Reece, L.F. Kubena and J.D. May, 1976. Effect of varying light intensity on broiler performance. Poult. Sci., 55: 515-519.
Direct Link - Downs, K.M., R.J. Lien, J.B. Hess, S.F. Bilgili and W.A. Dozier III, 2006. The effects of photoperiod length, light intensity and feed energy on growth responses and meat yield of broilers. J. Applied Poult. Res., 15: 406-416.
CrossRefDirect Link - Kristensen, H.H., G.C. Perry, N.B. Prescott, J. Ladewig, A.K. Ersboll and C.M. Wathes, 2006. Leg health and performance of broiler chickens reared in different light environments. Br. Poult. Sci., 47: 257-263.
CrossRefPubMedDirect Link - Newberry, R.C., J.R. Hunt and E.E. Gardiner, 1988. Influence of light intensity on behavior and performance of broiler chickens. Poult. Sci., 67: 1020-1025.
CrossRefPubMedDirect Link - Blatchford, R.A., K.C. Klasing, H.L. Shivaprasad, P.S. Wakenell, G.S. Archer and J.A. Mench, 2009. The effect of light intensity on the behavior, eye and leg health and immune function of broiler chickens. Poult. Sci., 88: 20-28.
CrossRefDirect Link - Rozenboim, I., B. Robinzon and A. Rosenstrauch, 1999. Effect of light source and regimen on growing broilers. Br. Poult. Sci., 40: 452-457.
CrossRefDirect Link - Zimmermann, N.G., 1988. Broiler performance when reared under various light sources. Poult. Sci., 67: 43-51.
CrossRefDirect Link - Mendes, A.S., S.J. Paixao, R. Restelatto, G.M. Morello, D.J. de Moura and J.C. Possenti, 2013. Performance and preference of broiler chickens exposed to different lighting sources. J. Applied Poult. Res., 22: 62-70.
CrossRefDirect Link - Bayraktar, H., A. Altan and C. Seremet, 2012. The effects of spot lighting on broiler performance and welfare. J. Anim. Vet. Adv., 11: 1139-1144.
Direct Link - Sultana, S., M.R. Hassan, H.S. Choe and K.S. Ryu, 2013. The effect of monochromatic and mixed LED light colour on the behaviour and fear responses of broiler chicken. Avian Biol. Res., 6: 207-214.
CrossRefDirect Link - Alvino, G.M., R.A. Blatchford, G.S. Archer and J.A. Mench, 2009. Light intensity during rearing affects the behavioural synchrony and resting patterns of broiler chickens. Br. Poult. Sci., 50: 275-283.
CrossRefDirect Link - Lien, R.J., J.B. Hess, S.R. McKee, S.F. Bilgili and J.C. Townsend, 2007. Effect of light intensity and photoperiod on live performance, heterophil-to-lymphocyte ratio and processing yields of broilers. Poult. Sci., 86: 1287-1293.
PubMedDirect Link - Mahmood, S., F. Ahmad, Ahsan ul Haq, G. Abbas and R.A. Qureshi et al., 2014. Comparative effect of light intensity on behavior and blood profile in broilers. Sch. Adv. Anim. Vet. Sci., 1: 25-29.
Direct Link - FASS., 2010. Guide for the Care and Use of Agricultural Animals in Research and Teaching. 3rd Edn., Federation of Animal Science Societies, Champaign, IL., USA.
Direct Link - Archer, G.S. and J.A. Mench, 2014. Natural incubation patterns and the effects of exposing eggs to light at various times during incubation on post-hatch fear and stress responses in broiler (meat) chickens. Applied Anim. Behav. Sci., 152: 44-51.
CrossRefDirect Link - Forkman, B., A. Boissy, M.C. Meunier-Salaun, E. Canali and R.B. Jones, 2007. A critical review of fear tests used on cattle, pigs, sheep, poultry and horses. Physiol. Behav., 92: 340-374.
Direct Link - Jones, R.B., 1986. The tonic immobility reaction of the domestic fowl: A review. Worlds Poult. Sci. Assoc. J., 42: 82-96.
CrossRefDirect Link - Archer, G.S. and J.A. Mench, 2013. The effects of light stimulation during incubation on indicators of stress susceptibility in broilers. Poult. Sci., 92: 3103-3108.
CrossRefDirect Link








