ABSTRACT
Systemic Acquired Resistance (SAR) is a mechanism of induced defense in unaffected parts of the plant by inoculating with microorganisms or other stress as an inducer that confers long-lasting protection against a broad spectrum of microorganisms. This experiment tries to detect the presence of SAR to Puccinia sorghi causing common rust in corn. Common rust susceptible corn plants, in the greenhouse condition, were inoculated by Pseudomonas bacteria; Exserohilum turcicum causing northern corn leaf blight (NCLB); toxin produced by Pyrenophora tritici-repentis (PTR), causing tan spot in wheat; Actigard® 50WG, a selective, systemic compound inducing host-plant resistance (AGD) and a control treatment using water as an inducers. Challenger (P. sorghi) was inoculated one week after inducer. Disease assessment was carried out 13th day after inoculation with P. sorghi by enumerating pustules numbers. Percent area leaf death of inducer-injected leaf was 100% in plants injected with PTR toxin and more than 80% in NCLB treatments. In AGD, PST and water injected plants percentage of dead leaf area was below 55% except two plants in Actigard® treated which were above 80%. Unlike previous findings, PTR toxin significantly decreased the pustules number by 70.52% than control. AGD and PST did not develop systemic resistance and developed pustules numbers comparable to control (PST:~105, AGD: ~104, Control: ~138). Based on this experiment, it can be concluded that SAR can be activated in corn, however, widely adapted commercial like Benzothiadiazole may not be efficient against P. sorghi.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ijpp.2011.43.50
URL: https://scialert.net/abstract/?doi=ijpp.2011.43.50
INTRODUCTION
Common rust is one of the worldwide spread foliar diseases of corn. It is caused by the fungus Puccinia sorghi. Corn cultivation is spread all over the world, so the fungus P. sorghi. It reduces the grain yield. Yield loss varies depending on the percentage of leaf area infected and the host growth stages. Estimates of reduction in grain weight range from about 3-8% for each 10% of the total leaf area infected (White, 1999). Epidemics of this disease can cause serious losses in yield and quality of sweet corn. High rust susceptibility of many popular sweet corn hybrids is a major factor contributing to rust epidemics (Cornell University, 2004).
Systemic Acquired Resistance (SAR) is a broad spectrum plant defense responses that can be induced biologically by challenging a plant with a weaker strain of a specific pathogen or exposing a plant to natural and/or synthetic chemical compounds (Percival, 2001). Put in simple, SAR is the activation of defense mechanism in uninfected parts of the plants. As the result the entire plant is more resistant to a secondary infection (USDA, 2008). Thus, a first infection presdisposes that plant to resist further attacks. The natural phenomenon of resistance development in response to pathogen infection was initially recognized by Ray (1901) and Beauvene (1901) working with Botrytis cinerea. However, laboratory study of SAR was performed by Ross (1961), who demonstrated that inoculation of a single leaf of tobacco with Tobacco Mosaic Virus (TMV) reduced the severity of subsequent infections on the other leaves and coined the term SAR. Similarly, the after researchers have detected SAR in different crops like cucumber (Galal, 2006), tomato (Esmailzadeh et al., 2008), pepper (Yusuf and Sally, 2004), wheat (Gorlach et al., 1996) etc.
SAR is associated with the systemic expression of a subset of defense gene, e.g; the acidic forms of pathogenesis related (PR1-5) proteins. Search for a signal that may be mobilized from the lesion to elicit systemic resistance has led to the identification of Salicylic Acid (SA) as the most likely candidate. Because SA could be recovered from phloem extracts and because SA treatment of plants leaves could induce both resistance to pathogen and SAR gene expression, it was proposed that SA could serve as an endogenous SAR signal molecule. However, SA is not itself signal and only a compound in signal chain. Rest of the signal chain is yet to be identified. SAR can also be induced chemically by the application of SA (Bi et al., 1997), 2,6-dichloroisonicotinic acid and its methylester called as INA in common, and 1,2,3-benzothiadiazole-7-thiocarboxylic acid-S-methyl-ester (BTH) (Gorlach et al., 1996).
Sneh and Auster (1998) found that among 153 isolates of Rhizoctonia spp 13.3% isolates protected more than 60% of the cabbage seedlings against R. solani and the best eight isolates protected 78-95% of the cabbage seedlings. Similarly, Gorlach et al. (1996) worked on wheat demonstrated that treatment of wheat plants with 30 g of BTH per hectare in repeated field trials led to an average of 35% reduction in disease symptoms of leaf rust caused by P. recondita and leaf spot caused by Septoria compared to the control treatment. Further, BTH induces resistance also in corn. Morris et al. (1998) found that BTH was effective in reducing downy-mildew disease. At 0.25 g of BTH per kg of seed it was significantly different only with control. However, application of 1 or 2 g of BTH per kg of seed induced a significant increase in disease resistance similar to the level of resistance induced by the Apron fungicide treatment. Chemical induction and pathogen infection increases resistance to downy mildew and activates expression of the corn PR-1 and PR-5 genes. They also mentioned that incompatible interaction with rust, disease resistance is correlated with a rapid and strong induction of the defense-related genes.
The objective of this study was to determine the induction of SAR in greenhouse-grown corn to common rust caused by basidiomycete Puccinia sorghi. Specifically we are interested to identify if the, non-host pathogen (Pseudomonas syringiae), northern corn leaf blight pathogen (Exserohilum turcicum) and chemicals (BTH and Pyrenophora tritici-repentis toxin) are effective in inducing SAR in corn.
MATERIALS AND METHODS
Experimental design: Greenhouse experiments were established in summer 2006 in a randomized complete block design with four replications. A corn variety susceptible to common rust, ND 246 (obtained as a courtesy of Dr. Marcelo J. Carena, North Dakota State University) was used in the experiment. Seeds, eight per pot, were planted in sterilized soil in magnum square pots (15x15x16.5 cm, Belden Plastics Co., St. Paul, MN). Plants were maintained in the greenhouse at 20-23°C. In addition to natural daylight, 14 h of supplemental light, starting at 1800 and ending at 0800, was provided. When the plants were at 3-4 leaf stage pots were thinned to six plants per pot and fertilized with Osmocote (14-14-14 N-P-K; 5.8 g per pot, The Scotts Company, Marysvile, OH). Inducer treatments included Pseudomonas bacteria (PST); Exserohilum turcicum causing Northern Corn Leaf Blight (NCLB); toxin produced by Pyrenophora tritici-repentis, a causal agent of tan spot in wheat (PTR); Actigard® 50WG, a selective, systemic compound inducing host-plant resistance (AGD) and a control treatment using water (H2O).
Collection of P. sorghi spores: During summer urediniospore of P. sorghi was collected from the standing sweet corn in Community Garden at Brookings, South Dakota. A vacuum machine designed for rust spore collection was utilized for the collection. Collected spores were hand cleaned to remove plants parts and stored at -20°C.
Multiplication and purification of inoculum of P. sorgi: Corn seeds were sown in 10 pots with six seeds in each. Plants were grown for twenty days in the greenhouse as mentioned earlier. On twentieth day, plants were inoculated with rust. For inoculation inoculum was prepared by thawing frozen spores at 2°C and mixing 20 mg rust spore in 1 mL Isoparaffin solvent (Saltrol 170, Chevron Phillips Chemical Company, Woodlands, TX). Twenty days old plants were inoculated using sprayer powered by CO2 and inoculated plants were kept in gentle breeze of air for 10 min to dry off isoparaffin. After which inoculated plants were kept in the mist chamber with 100% moisture for 24 h. After the dew period, pots were returned to the greenhouse. Thirteen days after inoculation newly developed fresh rust spores were collected. Collected spores were used as an inoculum for experimental plants.
Pseudomonas syringae inoculum (PST) preparation: Culture of Pseudomonas syringae was prepared according to Schaad (1998). P. syringae was cultured in the common media (K2HPO4-0.2 g, KH2PO4-0.05 g, Yeast Extract-0.2 g, Nutrient broth-0.8 g, Sucrose-0.25 g, Water-100 mL, sterilized at 121°C for 30 minutes). P. syringae was added to the culture and shaken overnight at 150 rpm at 27°C for 20 h.
Northern Corn Leaf Blight inoculum (NCLB) preparation: Isolates of E. turcicum was isolated from infected corn leaf tissue collected in different corn growing fields in Brookings, SD. Isolation was done following Ogliari et al. (2005) with slight modifications. Briefly, infected tissue was surface disinfected with 70% (v/v) aqueous alcohol followed by two minutes dip in 1% sodium hypochloride (v/v) and rinsed with sterile distilled water. Leaf pieces were incubated in water agar medium for 30 h at 20°C for sporulation. Single conidium from leaf was transferred to potato dextrose medium (PDA) for monoconidial culture development. Inoculum was produced by inoculating sorghum seeds, previously embedded in water (1.5 ssed:1 water; v/v) with monocoidial isolates followed by incubation in the dark for three weeks at 24°C. Inoculum at the rate of 1000 CFU was used as an inducers.
Pyrenophora tritici-repentis (PTR) toxin extraction: Conidia of Pyrenophora tritici-repentis (PTR) were produced following Lamari and Bernier (1989). Three plugs of clear PTR isolate collected from commercial wheat field in South Dakota were placed onto V8-PDA media (V8-tomato juice-200 mL, potato dextrose-, agar-, water-800 mL) and incubated for 72 h in dark at 22°C to produce colonies of ca. 2 cm in diameter. When the colony was about 2 cm in diameter, plate was flooded with sterile ddH2O and colony flattened using glass rod. After decanting the liquid, plates were incubated at room temperature (22-24°C) under fluorescent light for 24 h for conidiophore development.
| Table 1: | Recipe for modified frie’s medium |
 | |
After 24 h under light, plates were again incubated in dark at 15°C for 24 h for conidia development. Conidia were harvested by flooding plates with sterile ddH2O and gently scrapping with a rubber policeman. Conidial suspension was decanted into sterile beaker and enumerated. One milliliter of conidial suspension (104cfu mL-1) was added to 50 mL modified Frie's media (Table 1) in a 250-mL Erlenmeyer flask and incubated without agitation in dark at 20°C for 3 weeks. On 21st day culture was filtered through Whatman No.1 filter paper followed by filtration through 0.45 μm Millipore membrane, adjusted to pH 6.5 with 1 N NaOH and stored at -20°C.
Inoculation: Inoculation by inducer (PST, NCLB, PTR, AGD and H2O) was done when the plants were 26 days old, grown under condition described earlier. Inoculation was carried out in five of the six plants in each pot by puncturing leaf and injecting inducers through puncture by syringe. To insure the entry of inducers, injection was carried out until a watermark of about 3-4 cm long became visible. Pseudomonas was inoculated at the concentration of 106 cfu.
Seven days after inoculation with inducer, plants were challenged by P. sorghi. Freshly collected spores from greenhouse multiplied P. sorgi were used for inoculation. Spores (20 mg) were mixed with 1 mL isoparaffin solvent and sprayed onto the leaf just above the inducer injected leaf. Inoculated pots were kept in mist chamber (100% RH) for 24 h. After dew period, pots were brought back to the greenhouse.
Data collection and analysis: Data were collected two weeks after inoculation with P. sorghi. Number of pustules developed in the inoculated leaf just above the inducer-injected leaf was enumerated. Occurrence of necrosis on inducer-injected area was noted. Percentage of dead leaf area of inducer injected leaf was also estimated visually. Data were subjected to two way Analysis of Variance (ANOVA), followed by Least Significant Difference (LSD) test at p = 0.05 using SAS ver. 9.0 software. Graph was created using Sigma Plot v 10.0 software.
RESULTS
Statistical analysis was conducted on combined data of two runs of experiment as runs were not statistically significant source of variation (Table 2). The effect of treatments was highly significant both for rust pustule numbers and percent area leaf death of inducer-injected leaves. Necrosis occurred at the point of inducer injection in treatments NCLB, PST and PTR toxin. In plants injected with AGD and water control, necrosis was not visible. Percent area leaf death of inducer-injected leaf was 100% in plants injected with PTR toxin and more than 80% in NCLB treatments. In AGD, PST and water injected plants percentage of dead leaf area was below 55% except two plants in Actigard® treated which were above 80%.
When rust pustules number was analyzed for correlation with percent leaf death of inducer-injected leaves, correlation was negative and highly significant (r = -0.70, p≤0.0001). Sixty percent of plants had more than 50% of the leaf area dead in inducer-injected leaf. Though plants with higher percent area of leaf death had wide variation in terms of number of rust pustules developed, most of them were concentrated in the lower pustules region (Fig. 1). In contrast, plants with lower percent area of leaf death in inducer injected leaves developed higher number of rust pustules.
Numbers of rust pustules developed in PTR injected plants were significantly lower than the rest of the inducer treatments (Table 3). On an average PTR treatment reduced pustules by 70.52% than the water control, 70.72% less pustules than PST, 70% less than AGD and 61% less than NCLB. Though the NCLB injection significantly reduced pustules by 23.58% than water control, it was not significantly different than PST and AGD treatments. Number of rust pustules developed in AGD and PST treatments was not statistically different than the control. Further, contrast analysis revealed that the difference between the average of the expected pustules from chemical treatment (PTR and AGD) and biological treatment (NCLB and PST) were not statistically different (data not shown).
| Table 2: | Analysis of Variance (ANOVA) of percent area leaf death on inducer injected leaf and pustules developed in P. sorghi inoculated leaf |
 | |
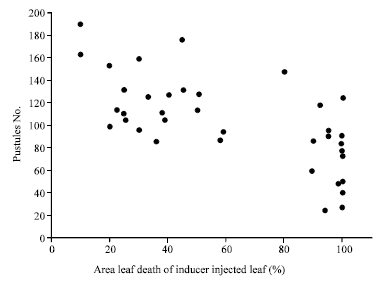 | |
| Fig. 1: | Scatter plot diagram of percent area leaf death of inducer-injected leaf against rust pustules developed in leaf inoculated with P. sorghi1 |
| Table 3: | Means of pustule numbers on observed in leaf inoculated with P. sorghi in four different treatments including water control |
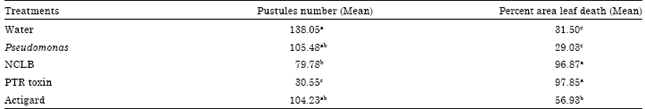 | |
| Means followed by the same letter within a column are not different at p≤0.05 by Tukey multiple comparisons of means. NCLB: Northern corn leaf blight | |
DISCUSSION
Induction of SAR is coordinated with the Hypersensitive Response (HR) through the expression of necrosis and other defense mechanisms in the plant (Devadas and Raina, 2001). Reduction in pustule numbers in PTR and NCLB treatments, that we observed in the current study, can be explained based on this necrosis development or HR. However, in case of PST treatment, pustules numbers were slightly higher than control despite the development of necrosis in the inducer injected area. In our current study, while the necrosis-developed plants had wide variation in pustule numbers, most of the plants in which necrosis was not developed at the point of inducer injection showed more numbers of rust pustules. Therefore, from this experiment it is evident that necrosis development in HR induces systemic resistance to P. sorghi.
Similarly, percent area of leaf death of inducer injected leaf is negatively correlated with the pustules numbers development and is statistically significant. Therefore, it can be inferred that the increased percent of area of leaf death leads to the increased systemic resistance to P. sorghi in corn.
Induction of systemic resistance can be acquired in corn similar to some other crops like, tomato, pepper, cucumber and wheat (Esmailzadeh et al., 2008; Galal, 2006; Yusuf and Sally, 2004; Gorlach et al., 1996). This is also proved in this study as there was significant decrease in pustules numbers in some of the treatments than in control. However, the surprising result in the study was the induction of SAR to P. sorghi by PTR toxin. Though, NCLB at spore concentration of 1000 CFU, also reduced pustules, PTR treatment was the most significant which has not been studied yet. Therefore, further study on this aspect is needed.
Morris et al. (1998) found that the BTH is as effective as commercially available fungicide, Apron, in inducing resistance to downy mildew of corn. Similarly Yusuf and Sally (2004) also reported induction systemic resistance when a BTH derivative was used against white mold disease of pepper caused by Sclerotinia sclerotiorum. In contrary, in our experiment commercially available BTH, Actigard® not only failed to induce systemic resistance but also the development of HR. Similarly, Pseudomonas, which has been used as non-host pathogen and expected to induce resistance, also did not induce SAR. The reason behind such results might be due to the timing between inducer and challenger inoculation. The protection obtained against the pathogen can be local or systemic depending on the time between the inducer treatment and challenger inoculation (Resende et al., 2002). Further, set of genes induced in different plants during the onset of chemically induced resistance are different; this results in spectrum of defense responses against different pathogen in the different plant species (Gorlach et al., 1996).
Results from the current study suggest that SAR can be activated in corn against common rust caused by P. sorghi. However, widely adapted commercial products for SAR induction like BTH may not be efficient against P. sorghi. Therefore, further study with diverse range of products is warranted.
REFERENCES
- Bi, J.L., J.B. Murphy and G.W. Felton, 1997. Does salicylic acid act as a signal in cotton for induced resistance to Helicoverpa zea? J. Chem. Ecol., 23: 1805-1818.
Direct Link - Esmailzadeh, M., M.J. Soleimani and H. Rouhani, 2008. Exogenous applications of salicylic acid for inducing systemic acquired resistance against tomato stem canker disease. J. Biol. Sci., 8: 1039-1044.
CrossRefDirect Link - Galal, A.M.M., 2006. Induction of systemic acquired resistance in cucumber plant against cucumber mosaic cucumovirus by local Streptomyces strains. Plant Pathol. J., 5: 343-349.
CrossRefDirect Link - Gorlach, J., S. Volrath, G. Knauf-Beiter, G. Hengy and U. Beckhove et al., 1996. Benzothiadiazole, a novel class of inducers of systemic acquired resistance, activates gene expression and disease resistance in wheat. Plant Cell, 8: 629-643.
CrossRefDirect Link - Morris, S.W., B. Vernooij, S. Titatarn, M. Starrett and S. Thomas et al., 1998. Induced resistance responses in maize. Mol. Plant Microbes Interaction, 11: 643-658.
PubMed - Ogliari, J.B., M.A. Guimaraes, I.O. Geraldi and L.E.A. Camargo, 2005. New resistance genes in the Zea mays-Exserohilum turcicum pathosystem. Genet. Mol. Biol., 28: 435-439.
Direct Link - Percival, G.C., 2001. Induction of systemic acquired resistance in plants: Potential implications for disease management in urban forestry. J. Arboric., 27: 181-192.
Direct Link - Resende, M.L.V., G.B.A. Nojosa, L.S. Cavalcanti, M.A.G. Aguilar and L.H.C.P. Silva et al., 2002. Induction of resistance in cocoa against Crinipellis perniciosa and Verticillium dahliae by acibenzolar-S-methyl (ASM). Plant Pathol., 51: 621-628.
CrossRef - Sneh, B. and M.I. Auster, 1998. induced resistance of cucumber seedlings caused by some non-pathogenic Rhizoctonia (np-R) isolates. Phytoparasitica, 26: 27-38.
CrossRef - Yusuf, Y. and M. Sally, 2004. Effect of treating pepper with benzothiadiazole derivative and non-pathogenic Xanthomonas campestris pathovars on the level of disease caused by Sclerotinia sclerotiorum. Plant Pathol. J., 3: 52-55.
CrossRefDirect Link - Ross, A.F., 1961. Localized acquired resistance to plant virus infection in hypersensitive hosts. Virology, 14: 329-339.
CrossRef









Getachew Zeleke Reply
Dear All,
Thank you in advance for sharing your experiences. I'm plant protection expert helping small, medium and large scale farmers in Ethiopia. It is of high value to my career. Please send to me any thing that you think will be helpful to me in the field of plant protection.
With Best Regards