Research Article
Asiatic Acid Ameliorates Cognitive Impairment in Diabetic Rats
Institute of Physical Culture, Hunan University of Arts and Science, Changde, Hunan, China
LiveDNA: 86.35761
Diabetes mellitus (DM) is an anomalous carbohydrate metabolism characterized by poorly controlled high blood sugar1. Hyperglycemia is mainly harmful to blood insulin concentrations or the sensitivity of organs and tissues to insulin. Long-term irregular hyperglycemia is closely related to many serious complications. The hippocampus plays an important role in cognitive performance. Moreover, the hippocampus is one of the vulnerable tissues of the brain which is affected severely in long-term irregular hyperglycemia2. Hence, diabetes-associated cognitive decline (DACD) is a common chronic complication of uncontrolled hyperglycemia. With the increasing worldwide epidemic of diabetes, the number of patients who suffer from cognitive impairment also rises greatly.
The mechanism of DACD is complicated. Blood glucose is a double-edged sword interrelated with the development of physiological changes in humans3. Moderate glucose in the blood has been increasingly recognized by people, for its validity in providing energy to maintain normal life activities in a healthy body. However, hypoglycemia disturbs central nervous system regulation function to cause fatigue, while excessive glucose is one of the risk factors in diabetes-metabolic syndrome4. Diabetic cognitive dysfunction belongs to neuropathy syndrome in metabolic disturbance5. The abnormalities of oxidative stress, inflammatory response and apoptosis are identified as crucial and main factors linked with the majority of these disorders. So natural products with anti-oxidative, anti-inflammatory and anti-apoptosis properties are potential approaches to attenuate cognitive dysfunction in diabetes mellitus.
Asiatic acid (AA) is a kind of pentacyclic triterpene, which possesses multiple effects on the physiologic function of an organism. In hyperglycemia, AA was demonstrated to enhance the capability of relieving diabetic symptoms, including decreasing plasma glucose and increasing high-density lipoprotein6. The protective action of AA was also investigated against STZ-induced oxidative injury and inflammation response in diabetic complications7. Moreover, AA played an important role in performing cognitive competence in many experiments, such as novel object location, passive avoidance and the Morris water maze test. It was proved that AA attenuated glutamate-induced apoptosis, which was involved in neuronal damage of the pyramidal layer and cognitive capability8.
However, it remains unknown if AA could protect the hippocampus against DACD. The major aim of this research was to explore the improvement of AA in the cognitive ability of diabetic rats.
Study area: The study was carried out at the Human Movement Science Laboratory, Hunan University of Arts and Science (January to July, 2021).
Animals and experimental design: A total of 45 Sprague-Dawley rats (male, 230±20 g) were raised in a stable environment (23±2°C, 50±10% humidity, 12 hrs light cycle). STZ (50 mg kg–1) was injected intraperitoneally to establish a diabetic rat model. The blood sugar level was above 235 mg dL–1 in rats, which could be selected as a practicable diabetic model. The purity of asiatic acid was >98%. AA was orally fed at 30 mg/kg/day for 45 days.
Morris water maze: Rats were measured by morris water maze (MWM) after administration of AA to appraise learning and memory performance. The rat was allowed to swim freely to adapt to the environment. In the escape latency experiment, the average time of looking for the hidden platform was checked to appraise learning ability for 4 consecutive days. In the probe trial experiment, the rat was measured to locate the former platform, which was removed from the water on the 5th day.
Biochemical assay: After MWM tests, rats were anaesthetized by chloral hydrate. Hippocampus was excised rapidly and washed with chilled isotonic saline. The sample was centrifuged (12000×g, 20 min, 4) and utilized for various biochemical determinations. The levels of SOD, CAT, GSH, MDA, AChE and ChAT were measured with commercial kits from Nanjing Jiancheng Biotechnology Institute, while TNF-α and IL-1β levels were analyzed by ELISA kits from BOSTER Biological Technology.
qRT-PCR analysis: The RNA was extracted from hippocampus tissues by trizol methods. Total RNA was reverse transcribed to synthesize cDNA which was amplified by PCR. The qRT-PCR was implemented by Bio-Rad real-time PCR system. The primer sequences were reported previously9. The β-actin was an inner control and mRNA expression was normalised to β-actin.
Western blot: The hippocampus was lysed, homogenized and centrifuged (12000×g, 20 min, 4) for the western blot experiment. The extractive supernatant was detected to appraise protein concentration through the BCA method. Then, the protein was boiled to break the stereoscopic structure. The antibodies were reported previously10. The primary and specific antibodies could bind to the particular target protein. Next, the protein binding compounds were incubated with secondary antibodies with an HRP label, which could be detected by the ECL system. The band assay was calculated by ImageJ. The β-actin was an inner control. The signal was demonstrated as a relative quantity.
Statistical analysis: All data were shown as Mean±SD. Statistical difference was demonstrated by the ANOVA test. The p<0.05 was deemed statistically significant.
Properties of AA on DACD: MWM was utilized to appraise the curative effect of AA against DACD. The time of escape latency was significantly augmented in the DM group, while AA remarkably decreased escape latency (Fig. 1a). In probe tests, the frequency of crossing the removed platform was significantly the reduced in DM group. By contrast, AA remarkably aggrandized the frequency of platform crossings (Fig. 1b).
The x-axis of Fig. 1a is expressed during different periods in the escape latency experiment. The x-axis of Fig. 1b is groups: CON: Control group, DM: Diabetes mellitus, DM+AA, Diabetes mellitus treated with asiatic acid (30 mg kg–1).
Properties of AA on diabetic-evoked cholinergic dysfunction in the hippocampus: To appraise the properties of AA on the diabetic-evoked cholinergic disorder, the activities of AChE and ChAT were measured in the hippocampus. The AChE activity was significantly elevated in the DM group, while AA remarkably inhibited diabetic-evoked AChE activity in the hippocampus (Fig. 2a). However, the ChAT activity was significantly decreased in the DM group, while AA remarkably elevated ChAT activity in the hippocampus (Fig. 2b).
Properties of AA on modulations of BDNF and GFAP in the hippocampus: To appraise the properties of AA on diabetic-evoked central nervous system dysfunction, BDNF and GFAP expressions were measured in the hippocampus. The BDNF expression was significantly restricted in the DM group, while AA remarkably strengthened BDNF expression in the hippocampus (Fig. 3a-b).
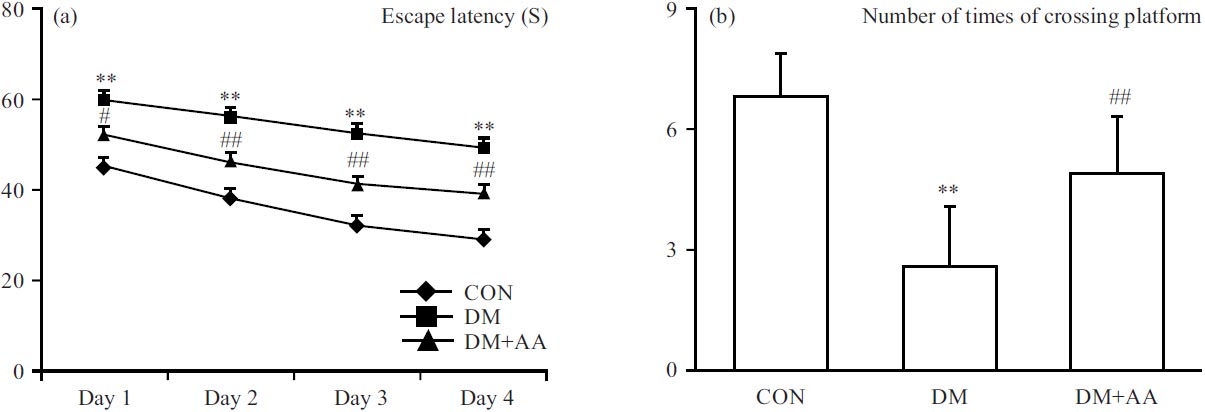 |
| Fig. 1(a-b): | Properties of (a) AA on escape latency and (b) Frequency of platform crossings in STZ-induced diabetic rat model **p<0.01 vs. CON group, #p<0.05 and ##p<0.01 vs. DM group |
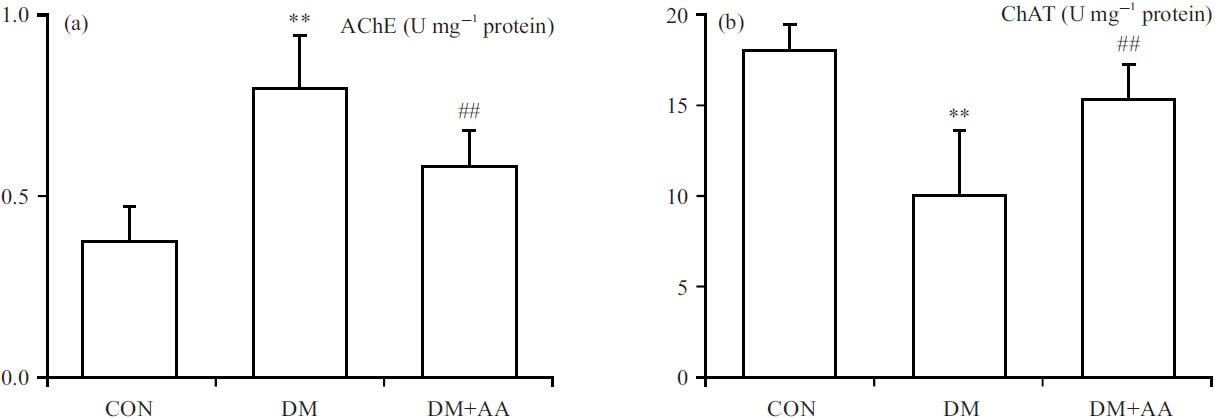 |
| Fig. 2(a-b): | Properties of AA on the regulations of (a) AChE and (b) ChAT concentrations **p<0.01 vs. CON group, ##p<0.01 vs. DM group, the X-axis of both parts are groups: CON: Control group, DM: Diabetes mellitus, DM+AA and Diabetes mellitus treated with asiatic acid (30 mg kg–1) |
However, GFAP expression was significantly elevated in the DM group, while AA remarkably inhibited diabetic-evoked GFAP expression in the hippocampus (Fig. 3c-d).
Properties of AA on modulations of Nrf-2/HO-1 pathway in the hippocampus: To appraise the properties of AA against DACD via the Nrf-2/HO-1 pathway, which is involved in process of hyperglycemic-relevant complications, Nrf-2 and HO-1 expressions were measured in the hippocampus. The Nrf-2 and HO-1 expressions were significantly relieved in the DM group, while AA remarkably strengthened Nrf-2 and HO-1expressions in the hippocampus (Fig. 4a-d).
Properties of AA on diabetic-evoked oxidative stress: To appraise the anti-oxidation properties of AA in the STZ-induced diabetes model, the contents of SOD, CAT, GSH and MDA were measured in the hippocampus. The SOD, CAT and GSH levels were significantly reduced in the DM group, while AA remarkably elevated these antioxidant factors towards normalcy in the hippocampus (Fig. 5a-c). In contrast, MDA concentration was increased significantly, while AA remarkably restricted diabetic-evoked MDA concentration in the hippocampus (Fig. 5d).
Properties of AA on diabetic-evoked inflammatory response: To appraise the anti-inflammatory properties of AA in the STZ-induced diabetes model, TNF-α and IL-1β levels were measured in the hippocampus. TNF-α and IL-1β levels were significantly increased in the DM group. By contrast, AA remarkably inhibited these inflammatory indicators in the hippocampus (Fig. 6a-d).
Properties of AA on diabetic-evoked apoptosis: To appraise the anti-apoptosis properties of AA in the STZ-induced diabetes model, Bax and Bcl-2 expressions were measured in the hippocampus. Bax expression was dramatically elevated in the DM group, while AA remarkably relieved diabetic-evoked Bax expression in the hippocampus (Fig. 7a-b). However, Bcl-2 expression was significantly suppressed in the DM group, while AA remarkably strengthened Bcl-2 expression in the hippocampus (Fig. 7c-d).
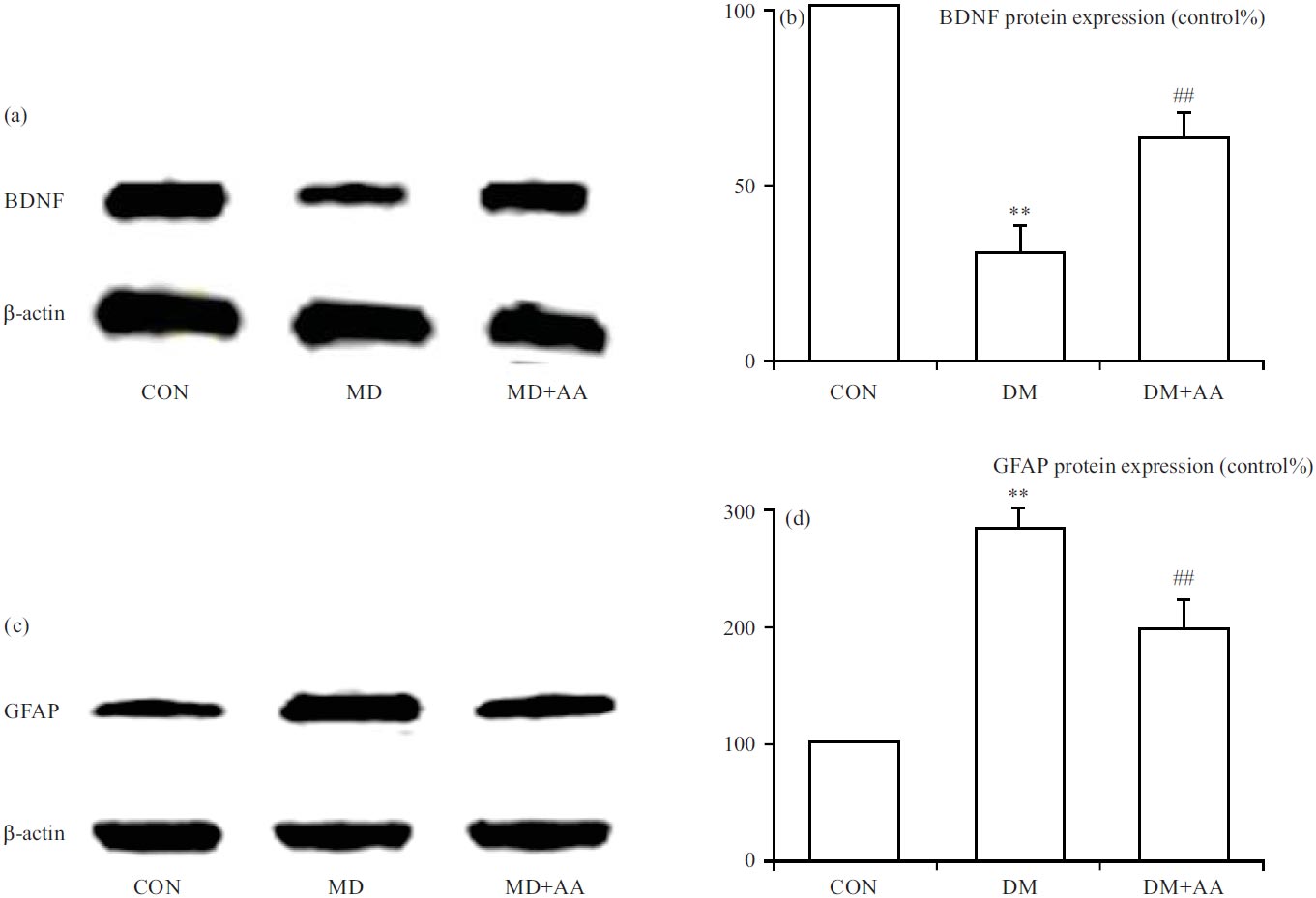 |
| Fig. 3(a-d): | Properties of AA on the modulations of (a, b) BDNF and (c, d) GFAP expressions **p<0.01 vs. CON group, ##p<0.01 vs. DM group, the x-axis of both parts are groups: CON: Control group, DM: Diabetes mellitus, DM+AA and Diabetes mellitus treated with asiatic acid (30 mg kg–1) |
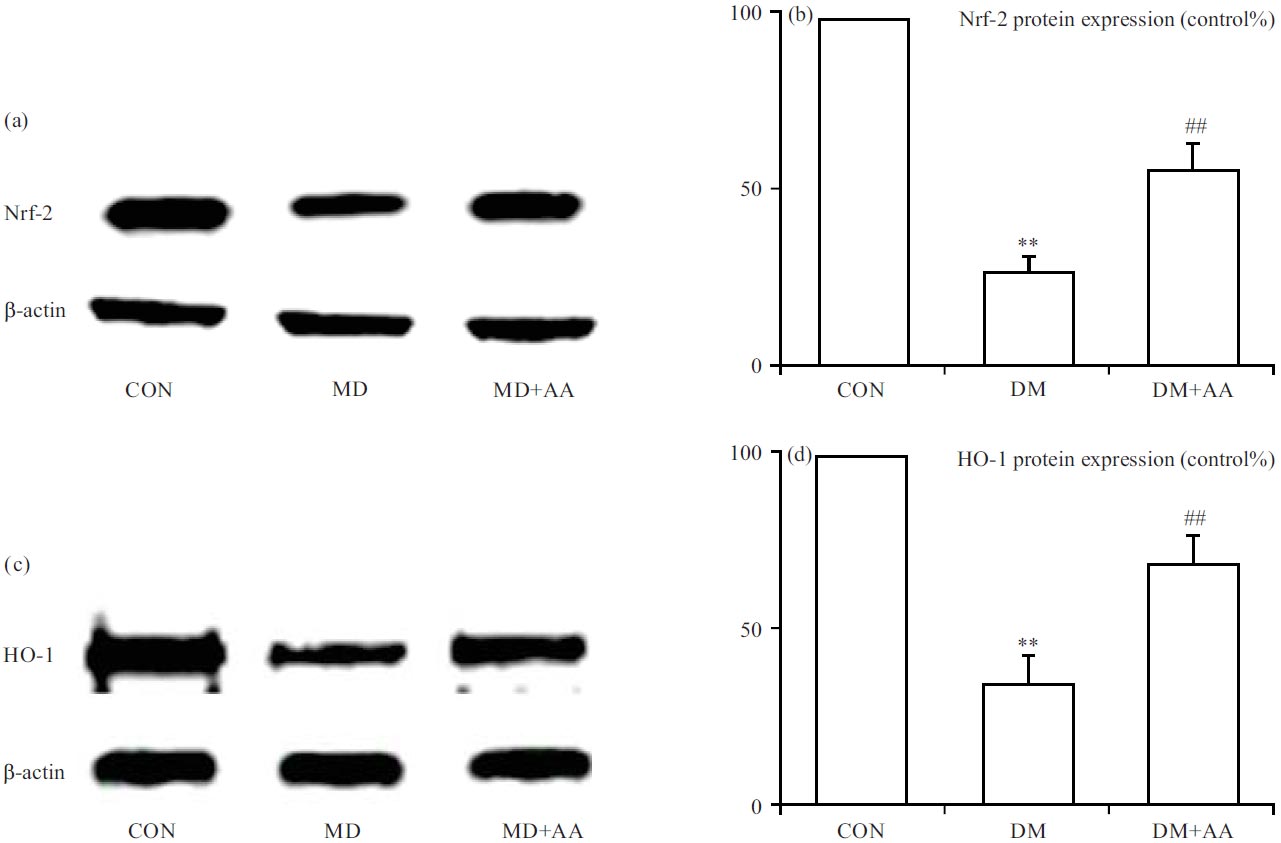 |
| Fig. 4(a-d): | Properties of AA on modulations of (a, b) Nrf-2 and (c, d) HO-1 expressions **p<0.01 vs. CON group, ##p<0.01 vs. DM group, the x-axis of both parts are groups: CON: Control group, DM: Diabetes mellitus, DM+AA and Diabetes mellitus treated with asiatic acid (30 mg kg–1) |
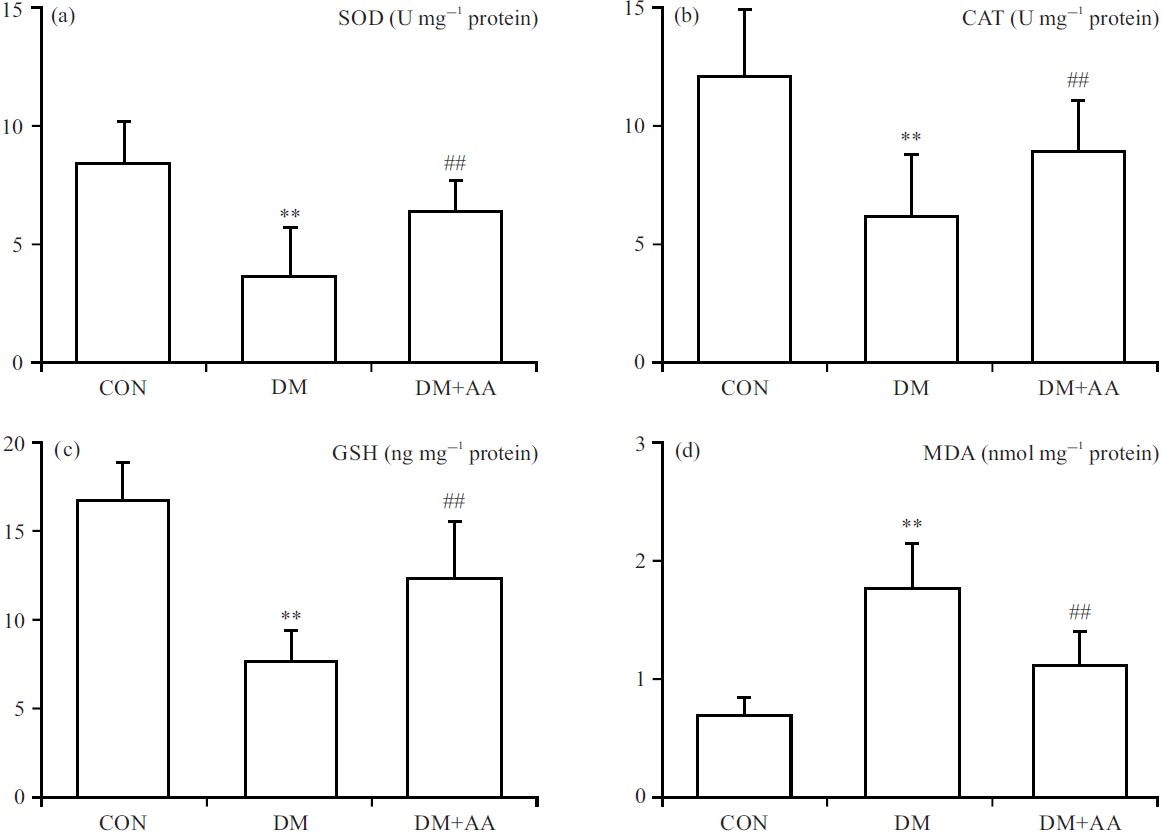 |
| Fig. 5(a-d): | Properties of AA on oxidation factors including (a) SOD, (b) CAT, (c) GSH and (d) MDA contents **p<0.01 vs. CON group, #p<0.05, ##p<0.01 vs. DM group, the x-axis of all the parts are groups: CON: Control group, DM: Diabetes mellitus, DM+AA and Diabetes mellitus treated with asiatic acid (30 mg kg–1) |
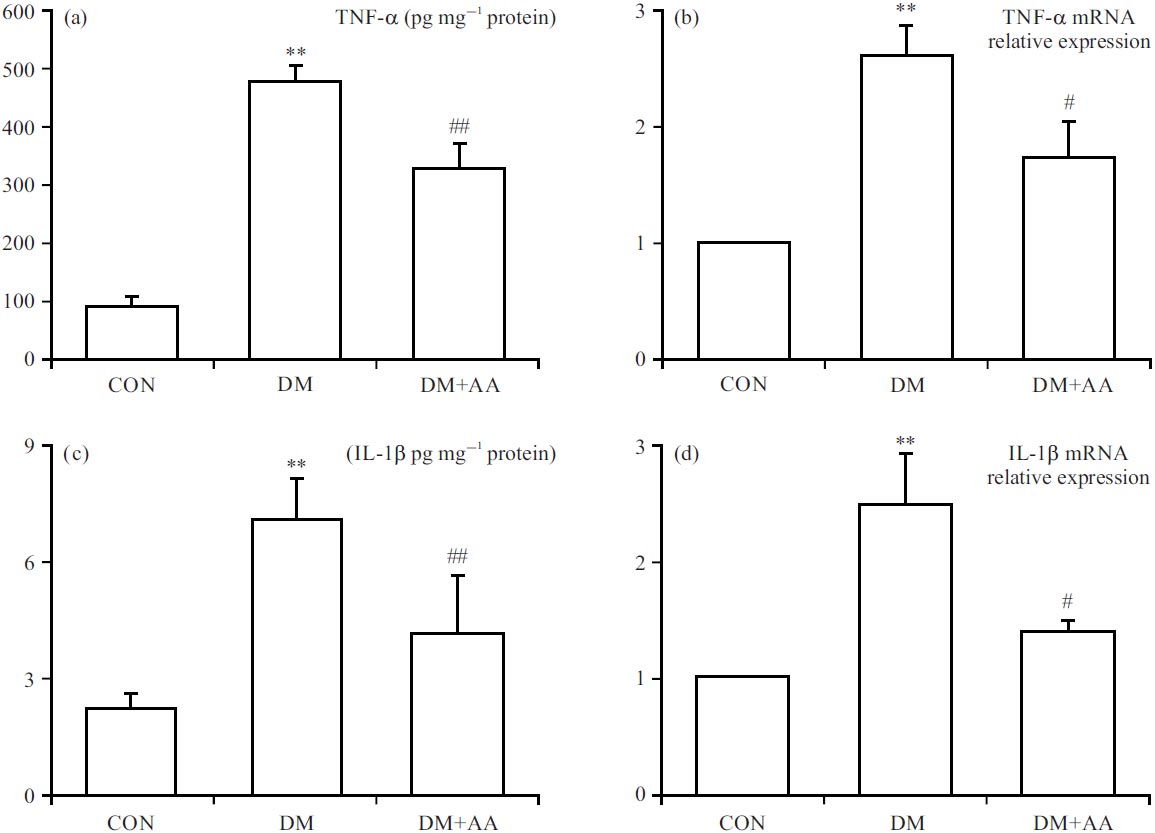 |
| Fig. 6(a-d): | Properties of AA on inflammation indicators including (a, b) TNF-α and (c, d) IL-1β levels **p<0.01 vs. CON group, #p<0.05, ##p<0.01 vs. DM group, the x-axis of all the parts are groups: CON: Control group, DM: Diabetes mellitus, DM+AA and Diabetes mellitus treated with asiatic acid (30 mg kg–1) |
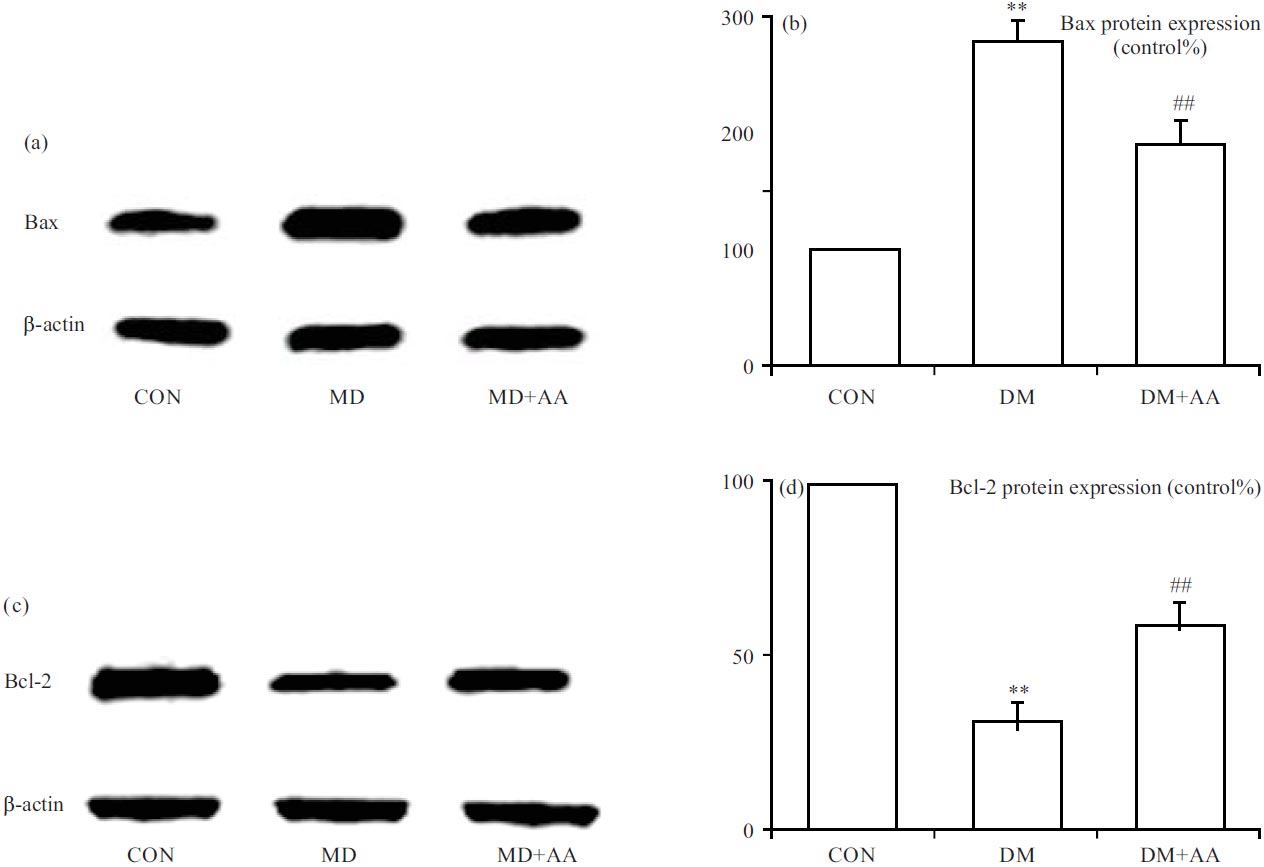 |
| Fig. 7(a-d): | Properties of AA on apoptosis parameters including (a, b) Bax and (c, d) Bcl-2 expression **p<0.01 vs. CON group, ##p<0.01 vs. DM group, the x-axis of both parts are groups: CON: Control group, DM: Diabetes mellitus, DM+AA and Diabetes mellitus treated with asiatic acid (30 mg kg–1) |
DACD is described as learning and memory impairments, which are closely related to hyperglycemia. The AA with anti-oxidation, anti-inflammation and anti-apoptosis could protect against both diabetes and cognitive defect11,12. Previous studies showed that AA prevented organs against hyperglycemia-related injuries, such as diabetic nephropathy and diabetic cardiopathy13. In addition, AA was involved in the improvement of quinolinic acid, kainic acid and aluminium chloride-induced toxicology, which were risk factors for cognitive impairments14-16. Thus, it is highly reasonable that AA possessed biological effects in the treatment of DACD. In the present study, results demonstrated AA mitigated hyperglycemia-induced cognitive impairments by MWM.
Cholinergic dysfunction is closely related to cognitive decline and poorly controlled hyperglycemia is harmful to the modulation of synaptic function. AChE and ChAT are key marker enzymes in the regulation of the cholinergic system that modulates neurotransmitter products to alter learning and memory processes17. There are various results that hyperglycemia perturbs cholinergic dysfunction on DACD. Previous studies showed that diabetes was involved in nervous system disorder by decreasing AChE and increasing ChAT. In AlCl3-induced amyloid pathology, AA increased AChE activity to improve spatial memory capacity and motor function18. In this study, AA modulated the cholinergic system by mitigating AChE activity and enhancing ChAT activity on DACD.
BDNF is a neurotrophin and is mainly expressed in the central nervous system, especially in the hippocampus, which contributes to learning and memory processes. The GFAP is an astrocyte-specific marker which gets activated in response to dysfunction of the central nervous system. In diabetes, hyperglycemia disturbs the original normal expression of BDNF and GFAP in the hippocampus19. In addition, AA strengthened BDNF expression and weakened GFAP expression to prevent or alleviate D-galactose-induced brain injury20. In this study, AA coordinated the central nervous system by raising BDNF expression and declining GFAP expression to prevent DACD.
Nrf-2 and HO-1 expressions were inhibited in hyperglycemia-induced hippocampus toxicology. Previous research showed Nrf-2/HO-1 pathway possessed multiple bioactivities which were involved in the regulation of oxidative, inflammation and apoptosis21. In spinal cord injury, AA promotes Nrf-2/HO-1 levels and suppressed NLRP3 expression to ameliorate oxidative stress and inflammation22. Additionally, AA increased HO-1 expression in cigarette smoke-induced pulmonary inflammation23. Besides, AA promoted Nrf-2 signalling to protect against oxidative injury and apoptosis in tert-butyl hydroperoxide-stimulated HepG2 cell24. In this study, AA ameliorated DACD by activating Nrf-2 and HO-1 expressions.
AA is a potent anti-oxidation agent. In DACD, long-term hyperglycemia facilitates disequilibrium of oxidation and antioxidation. In hyperglycemia, AA was demonstrated to enhance the oxidation resistance by retaining cardiac GSH content to attenuate glycative damage and coagulatory disorder7. In addition, AA enhanced GSH content and reduced the MDA level of the brain to alleviate cognitive deficits in monosodium glutamate-induced dementia8. In this study, AA offered potent beneficial functions against STZ-induced cognitive disorder by elevating SOD, CAT and GSH levels and restoring MDA contents.
Uncontrolled hyperglycemia induces an inflammatory response, which is involved in the development of cognitive decline. The TNF-α and IL1-β are common inflammatory factors. In the hippocampus, hyperglycemia promoted TNF-α and IL1-β levels to cause more serious cognitive deficits and that anti-inflammatories may be effective at attenuating this malfunction. In AlCl3-evoked cognitive impairment, AA ameliorated inflammation by attenuating TNF-α and IL1-β expressions25. In this study, AA ameliorated STZ-influenced inflammation against cognitive impairment via shifting TNF-α and IL-1β levels towards normalcy.
Abnormality of apoptosis is one of the central characteristics of DACD. Moreover, oxidative stress and inflammatory response are important processes in cell apoptosis. Bax and Bcl-2 are marks of apoptosis. The AA is the main compound of chloroform fraction from Actinidia arguta which inhibited apoptosis by preventing Bax expression against high-fat diet-induced cognitive dysfunction26. In the high glucose-induced toxicant experiment, AA improved cell viability by increasing Bcl-2 mRNA expression and reducing mRNA expression27. In this study, AA exhibited its inhibition of apoptosis by reducing Bax expression and enhancing Bcl-2 expression.
This study implied the alleviative effect of AA against cognitive impairment in STZ-evoked diabetic rats. The AA decreased escape latency and enhanced times of crossing platform. In the hippocampus, AA restricted AChE and GFAP expressions, while promoting ChAT and BDNF expressions to attenuate brain injury. Additionally, AA inhibited oxidative stress, inflammatory response and apoptosis in the treatment of cognitive dysfunction in diabetic rats. Therefore, these possible mechanisms revealed AA had plentiful biological and pharmacological functions and could be developed as an available agent for ameliorating DACD.
Current research identified AA improved diabetic-evoked cognitive dysfunctions which were connected to its bioactivities of anti-oxidation, anti-inflammation and anti-apoptosis. The above research exhibited AA may be a powerful remedy approach to mitigating the cognitive decline in diabetes.