Research Article
Metformin Ameliorates Infiltration of Inflammatory Cells and Pancreatic Injury Biomarkers Induced by L-Arginine
Department of Physiology, College of Medicine, King Khalid University, Abha, Saudi Arabia
LiveDNA: 966.36872
The inflammatory disease, Acute Pancreatitis (AP) is characterized by severe abdominal pain that lasts for days to a few weeks and represents a significant challenge to the health community since it can develop to systemic inflammation and multiple organs failure causing a high mortality rate that can reach of up to 50% in the severe form of AP1,2. The pathogenesis of AP is caused by many factors such as the gallstone blocking the common bile duct, heavy alcohol consumption, adverse drug effects, direct trauma, virus and sepsis and shock3. Activation of digestive proteinases like trypsin can lead, in severe cases, to diffuse pancreatic necrosis and haemorrhage, leukocyte infiltration and necrosis and apoptosis of pancreatic acinar cells, which lead to self-digestion and inflammation of the pancreas4. Experimental AP using animal models of the disease is very useful to thoroughly understand the pathophysiology of the disease and to test potential drugs and compounds to treat AP5.
It is well documented that L-arginine can induce AP in rats and mice following intraperitoneal injections of 2 doses (2.5-4 g kg–1) of the amino acid2,5. L-arginine is converted to L-citrulline and nitric oxide by the enzyme nitric oxide synthase and the fast reaction of nitric oxide with oxygen radicals yield a highly cytotoxic compound, peroxynitrite that induces nitrosative stress leading to tissue damage6. In addition, L-arginine is hydrolyzed to L-ornithine and urea by the enzyme arginase and L-ornithine is reported to induce a severe type of AP in rats7. Furthermore, the analogue of the hormone cholecystokinin, cerulean8 and ethanol9 were successfully used to induce AP in animal models, which is characterized by pancreatic inflammatory cell infiltration, activation of intracellular digestive enzymes, apoptosis in pancreatic acinar cells, cell death and fibrosis.
The antidiabetic drug metformin10 is emerging as a pleiotropic drug that has many benefits. For example, metformin is reported to ameliorate several types of liver diseases such as nonalcoholic fatty liver disease, improving liver injury in diabetes with hyperlipidaemia11 and protection of primary rat hepatocytes against oxidative stress-induced apoptosis12. In addition to the anti-inflammatory13 and antioxidant14 effects of metformin, metformin was also reported to inhibit the progression of pancreatic cancer15.
We recently reported inhibition of L-arginine-induced acute pancreatitis by vitamin E via the modulation of TNF-α-AMPK axis16 and in cell signalling, AMPK activation is the most well-known mechanism of action of the drug metformin17.
Therefore, we speculated that modulation of the TNF-α-CD45 axis and AMPK by acute pancreatitis in a rat model of the disease could be inhibited with metformin.
Study area: This study was carried out at the Research Centre, College of Medicine, King Khalid University, Abha, Saudi Arabia from March-June, 2020.
Animals: All experimental procedures were approved by the medical research ethical committee at King Khalid University and according to the guide for the care and use of laboratory animals published by the US National Institutes of Health (NIH publication No. 85-23, revised 1996). Male Wistar rats (total 24 rats) weighing 150-200 g were used for these studies. All rats were bred and housed in the research centre of King Khalid University, College of Medicine (Abha, Saudi Arabia), at temperatures of 23±1°C and a 12 hrs light: 12 hrs dark cycle. Rats had free access to tap water and fed standard laboratory chow during the acclimatization period.
Experimental design: After a 1 week adaptation period, rats were randomly assigned into 4 groups (n = 6, each) and were distributed in their corresponding cages and classified as follows: Control group (Control): Nontreated rats that were injected intraperitoneally with the vehicle, metformin group (Met): Rats treated with metformin (50 mg kg–1) daily for 17 days, L-arginine-treated group, the model group (L-arg): Rats were injected intraperitoneally on day 15 with 2 doses of L-arginine (2.5 g kg–1) at 1 hr intervals2. They received no treatment (vehicle) in the 1st 2 weeks, the protective group (Met +L-arg): Rats were treated with metformin from 1-17 days and injected on day 15 with 2.5 g kg–1 L-arginine, 2 doses within 1 hr. At the end of the experimental period (on day 17), blood samples were collected by cardiac puncture under anaesthesia (sodium thiopental at 40 mg kg–1 body weight) and animals were then culled by cervical dislocation under anaesthesia and pancreatic tissues were harvested. Blood samples were collected without anticoagulation, left for 10 min, then centrifuged for 10 min at 4000 r min–1 to obtain serum, which was stored at -20°C until further biochemical analysis.
Determination of blood levels of amylase and lactate dehydrogenase (LDH): On day 17, animals were sacrificed and serum levels of amylase and LDH were assessed (colourimetric) by assay kits obtained from Abcam, Cambridge, UK. All parameters were determined according to the manufacturer’s instructions.
Quantitative real-time polymerase chain reaction (qRT-PCR) of TNF-α and IL-10: qRT-PCR was performed as previously described13. In brief, Total RNAs were isolated from freshly dissected rats’ pancreases using the RNeasy Mini Kit (Qiagen Pty, Victoria, Australia) and 1 mg RNA was reverse-transcribed with the cDNA synthesis kit (Fermentas, USA). Triplicate cDNA samples and standards were amplified in Master Mix containing SYBR green using an Applied Biosystems (Thermo Fisher Scientific Inc, MA, USA) with primers specific for TNF-α (sense, 5-GATCTCAAAGACAACCAACATGTG-3, antisense, 5-CTC CAGCTGGAAGACTCCTCCCAG-3), IL-10 (sense, 5-GCAGGACTT TAAGGGTTACTTGG-3, antisense, 5-GGGGAGAAATCGATG ACAGC-3) and β-actin (sense, 5-GGTCGGTGTGAACGGATTTGG-3, antisense, 5-ATGTAGGCCATGAGGTCCACC-3). The relative expression was calculated according to the manufacturer software.
Western blotting analysis of AMPK: Proteins were extracted from pancreas tissues and 25 μg of protein per sample were immunoblotted as we described previously18. Membranes were probed with anti-AMPK-phospho-Thr172 (1:1000 Cell Signalling Technology, Beverly, MA, USA) at 4°C overnight. Proteins were visualized using the ECL detection kit (Amersham-Pharmacia, UK). Relative expression was determined using Image analysis software to read the band intensity of the target proteins against the control sample after normalization by β-actin on the Chemi Doc MP imager.
Histological analysis: Pancreas specimens were immediately fixed in 10% formal saline for 24 hrs. Paraffin blocks were prepared and 5 μm thick sections were stained with hematoxylin and eosin (H and E) stain to elucidate the status of pancreas architecture and the structural changes.
Immunohistochemistry of leukocyte common antigen (CD45): The pancreas from all rats were collected and fixed in 10% formal saline for 24 hrs before dehydration with alcohols and paraffin embedding using standard methods. Blocks were processed, sectioned in 5 μm thickness and deparaffinized. Antigen retrieval was made by boiling the sections in 10 Mm citrate buffer pH 6 for 10 min, then the sections were left to cool at room temperature for 20 min. Sections were incubated in a humidity chamber with the primary antibody, anti-cluster of differentiation (CD) 45 (Cat# ab10558, Abcam, Cambridge, UK) as a marker for cells of haematopoietic origin. Sections were then counter stained with Meyer hematoxylin.
Statistical and morphometric analysis: The data were expressed as Mean±Standard Deviation (SD). Data were processed and analyzed using the SPSS version 10.0 (SPSS, Inc., Chicago, Ill., USA). One-way ANOVA was performed followed by Tukey’s post hoc test. Pearson correlation statistical analysis was done for the detection of a probable significance between 2 different parameters. Results were considered significant if p<0.05.
Using the "Leica Qwin 500 C" image analyzer (Cambridge, UK), the areas (%) of CD45 immunostaining were obtained in 10 non-overlapping high power fields/rat of immunostained sections. Quantitative data were tabulated as a means and Standard Deviations (SD) and compared using analysis of variance (ANOVA) followed by post hoc analysis (Tukey test). A significant difference was considered when p<0.05. Calculations were made on SPSS software (version 19).
L-arg induces acute pancreatitis in rats: We first modelled acute pancreatitis in rats to test our working hypothesis mentioned above. Injection of the model group of rats with 2 doses of L-arginine (2.5 g kg–1) at 1 hr intervals caused after 2 days a sharp increase in biomarkers of pancreas injury and abnormal changes in pancreatic tissue histology (Fig. 1). Significant (p<0.001) high serum levels of amylase and LDH were observed in the model group (L-arg) compared to normal levels in the control group (Fig. 1a-b). Pancreas sections prepared for H and E staining of L-arg injected rats revealed disorganized lobular architecture with inflammatory infiltration within the Connective Tissue (CT) septa. The acini appear with multiple cytoplasmic vacuolations and pyknotic nuclei. In addition, the presence of congested blood vessels and extravasated blood in between the acini compared to normal structures in the control group (Fig. 1c-d).
Metformin inhibits L-arg-induced biomarkers of inflammation and acute pancreatic injury: To determine whether metformin treatment can inhibit the release of biomarkers of inflammation and pancreas injury, we measured tissue gene expression of TNF-α and IL-10 and serum levels of amylase and LDH in all animal groups (Fig. 2). Metformin treatment significantly (p<0.0265) reduced L-arg-induced the inflammatory biomarker TNF-α (Fig. 2a) and augmented the pancreatic tissue gene expression of the anti-inflammatory biomarker, IL-10 (Fig. 2b). Metformin also ameliorated serum amylase (Fig. 2c) and LDH (Fig. 2d). In addition, the tissue and blood levels of these parameters in the control metformin group were comparable to the control untreated group. However, the level of TNF-α, IL-10, amylase and LDH in the treated group (Met+L-arg) was significantly (p<0.0424) elevated compared with the control group of rats. This means partial protection by metformin was achieved.
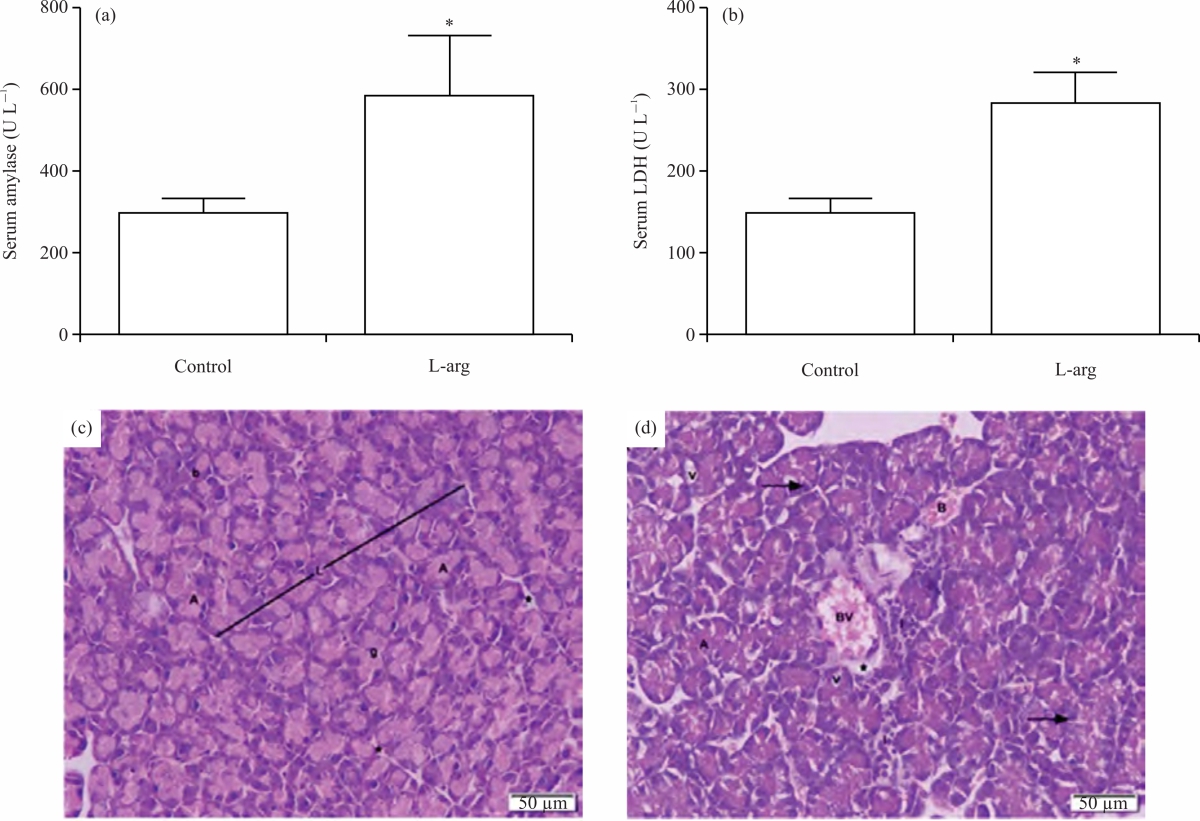 |
| Fig. 1(a-d): | Induction of acute pancreatitis in rats by L-arg, (a) Blood levels of amylase, (b) LDH were measured at the end of the experiment in the model group (L-arg) compared to the control group rats, (c) H&E stained images (x200) of harvested tissues of rats from the control group and (d) the model group are visualized using light microscopy Results represent the (Mean±SD), *p = 0.001 versus control, (c and d) H&E stained images (x200) of harvested tissues obtained from the pancreas. Arrows in (D) point to pyknotic nuclei and the (*) points to the CT septa, H and E: Hematoxylin and eosin, L: Lobules, A: Acini, I: Infiltrated inflammatory cells, b: Basophilic cytoplasm, g: Acidophilic granules, v: Vacuoles, BV: Congested blood vessels and B: Extravasated blood |
Metformin increases AMPK T172 phosphorylation inhibited by L-arg: To investigate whether the observed inhibition of TNF-α with metformin treatment shown above was also associated with the modulation of AMPK, we evaluated the pancreatic tissue levels of phosphorylated AMPK (p-AMPK) at T172 in all animal groups (Fig. 3). A sharp decrease in the expression of the p-AMPK protein was observed in immunoblots of pancreatic tissue samples prepared from the model group (L-arg), which was significantly (p<0.0001) augmented by metformin treatment (Fig. 3a-b).
We further determined the correlation between AMPK scorning and TNF-α and amylase in all animal groups. The data in (Fig. 3c-d) show a negative correlation between AMPK protein expression and these biomarkers, TNF-α mRNA (r = -0.9033, p<0.0001) and amylase (r =-0.7418, p = 0.0004).
Metformin inhibits leukocyte infiltration induced by L-arg in pancreas tissue: To determine whether the activator of AMPK, metformin can effectively reduce pancreatic leukocyte infiltration induced by L-arg intoxication in rats, we assessed the infiltration of inflammatory cells (CD45+ leukocytes) into the pancreas tissue (Fig. 4). Compared to a weak CD45 positive immunostained cells in control groups (Fig. 4a-b), immunohistochemical staining for this biomarker in pancreas sections of the L-arg group showed numerous CD45 positive cells (Fig. 4c-d), which were significantly (p<0.0001) inhibited by metformin in the metformin-treated groups (Met+L-arg) (Fig. 4e-f). Furthermore, quantification of CD45 positive immunostaining cells (Fig. 4g) showed effective inhibition of CD45 by metformin.
Correlation between pancreatic leukocyte infiltration score and inflammation, AMPK and tissue injury biomarkers: The link between CD45 score and inflammatory and anti-inflammatory biomarkers, AMPK and pancreatic injury biomarker in L-arg-induced acute pancreatitis is shown in Fig. 5.
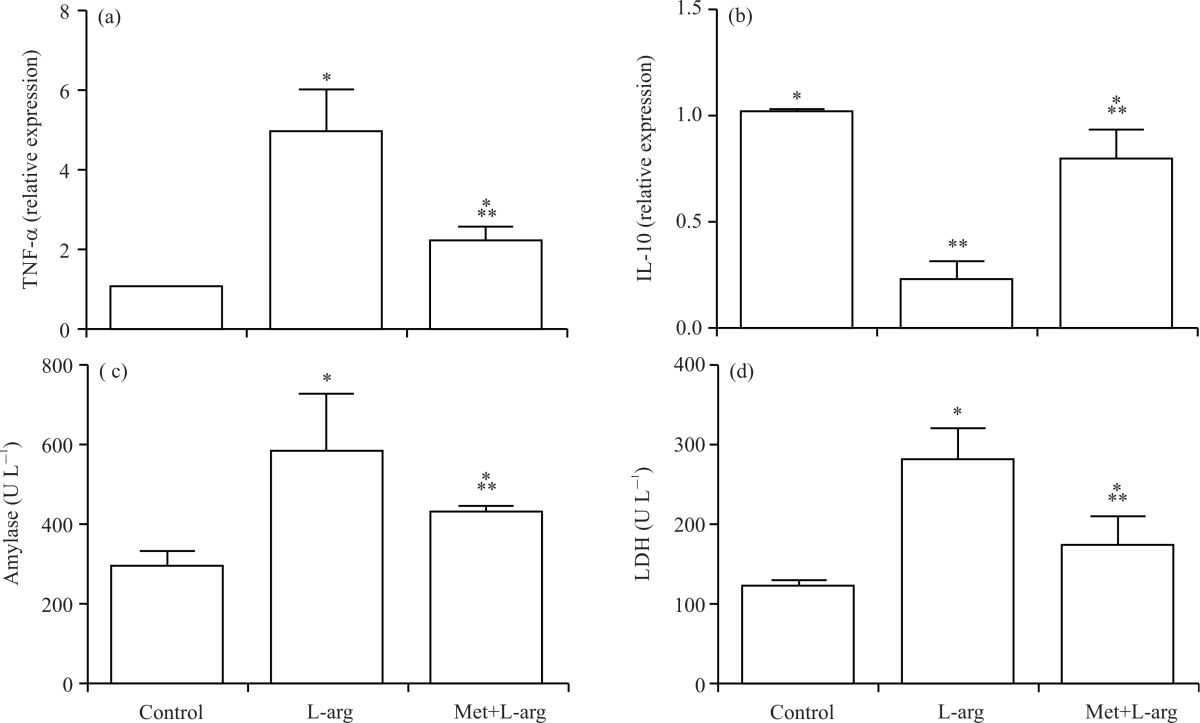 |
| Fig. 2(a-d): | Inhibition of L-arg-induced biomarkers of inflammation and acute pancreatic injury by metformin, (a) pancreas tissue levels of TNF-a, (b) pancreas tissue levels of IL-10, (c) blood levels of amylase, (d) blood LDH were measured at the end of the experiment in all groups of rats Results represent the Mean±SD, presented p-values are all significant, *p < 0.0001 versus control and **p < 0.0265 versus L-arg |
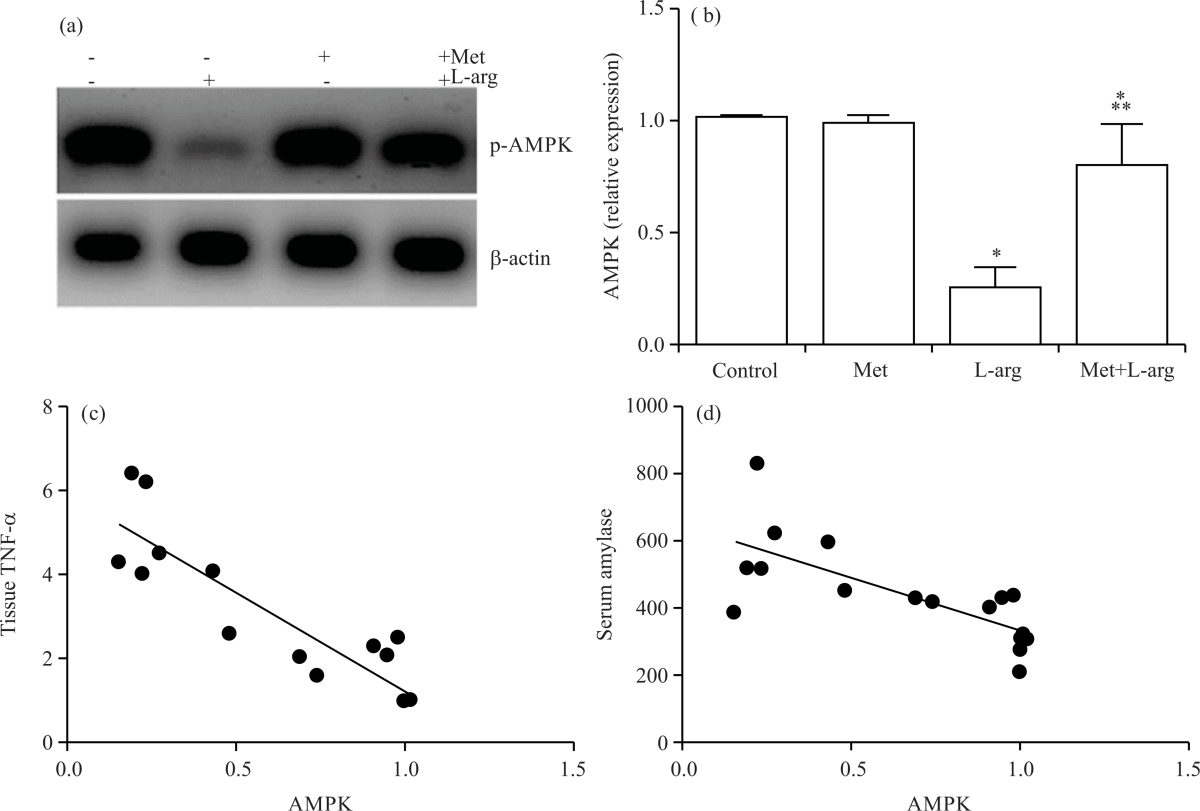 |
| Fig. 3(a-d): | Metformin protects against AMPK inhibition caused by acute pancreatitis, pancreas tissue lysates prepared from all the groups of rats were examined by Western blotting for phospho-AMPK (p-AMPK) (a and b) and β-actin as a loading control (a), Histograms represent the relative expression of p-AMPK signalling protein is shown (b), (c) significant correlation between p-AMPK versus TNF-amRNA, (d) p-AMPK versus serum amylase Results represent the (Mean±SD), Presented p-values are all significant, *p < 0.0001 versus control, **p < 0.0245 versus L-arg |
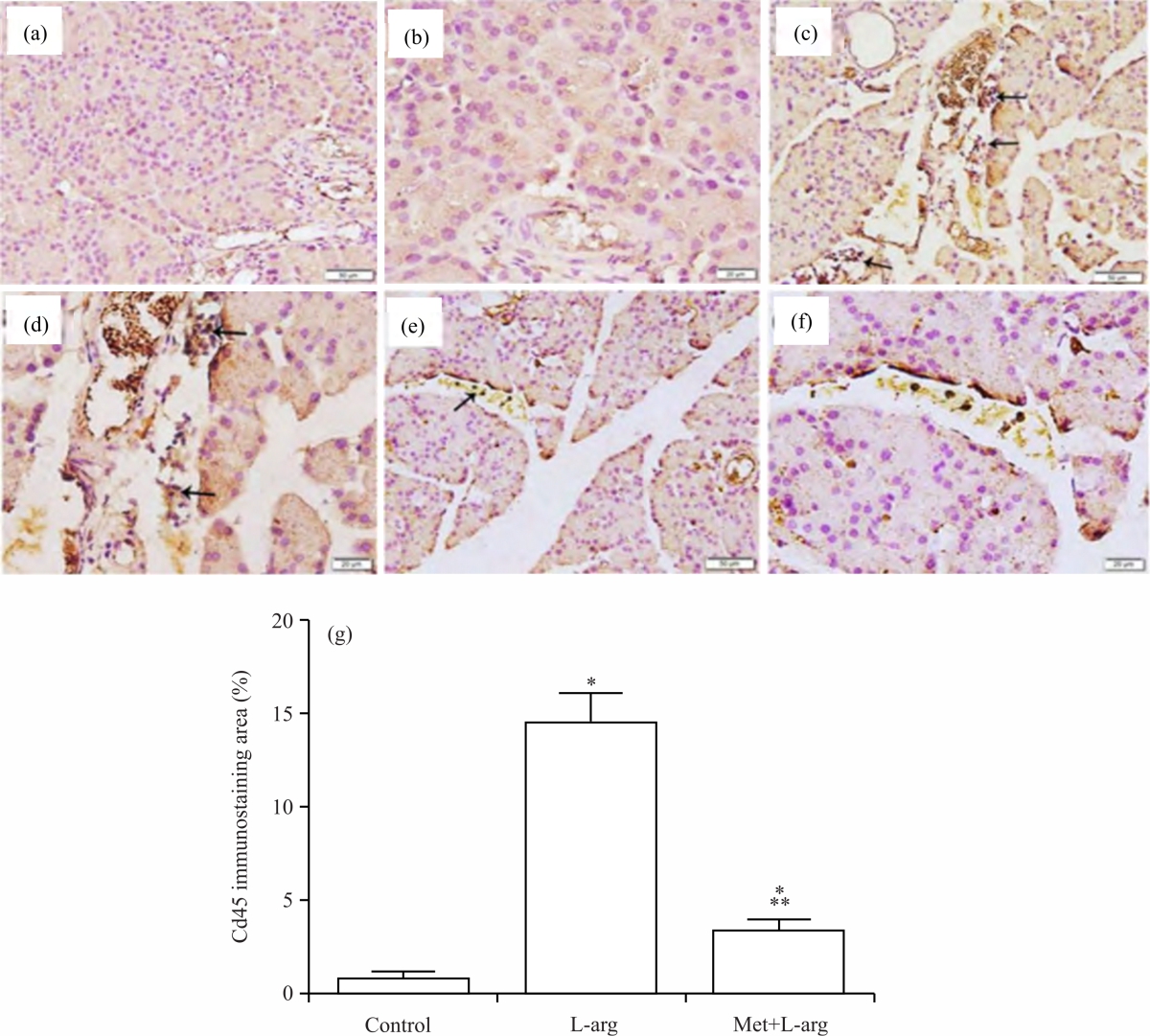 |
| Fig. 4(a-g): | Metformin inhibits L-arg-induced CD45 expression, (a and b) Immunohistochemistry of CD45 of pancreas sections (a,c,e, x200; b,d,f, x400) from the control, (c and d) L-arg, (e and f) Met+L-arg groups of rats are depicted. (g) histograms represent the quantitative analysis of CD45 immunostaining area % in pancreas sections from the above groups Arrows point to positive CD45-immunostained cells, Presented p values are all significant, *p<0.0001 versus control and **p<0.0245 versus L-arg, CD45: Cluster of differentiation 45 (leukocyte common antigen) and L-arg: L-arginine and Met: Metformin |
A significant correlation between these parameters was observed, CD45 versus TNF-α (r = 0.911, p<0.0001) (Fig. 5a), CD45 versus IL-10 (r = -0.968, p<0.0001) (Fig. 5b), CD45 versus AMPK (r = -0.954, p<0.0001) (Fig. 5c) and CD45 versus amylase (r = 0.792, p<0.0001) (Fig. 5d).
This study investigated the TNF-α-CD45 axis mediated acute pancreatitis and AMPK in a rat model of L-arg-induced acute pancreatitis in the presence and absence of metformin. Also, we investigated the link between these parameters as well as pancreatic injury biomarkers. Therefore, we modelled acute pancreatic injury in rats and here we have shown, using western blots, qRT-PCR, blood chemistry and immunohistochemistry staining methods that induction of acute pancreatitis in rats by L-arg caused after 2 days a substantial augmentation of inflammation and leukocyte infiltration (CD45) and inhibition of pancreatic p-AMPK and anti-inflammatory biomarker IL-10 that was modulated by metformin (Fig. 2-4). In addition, our data demonstrate a significant correlation between these parameters which support the link between the pathophysiology of acute pancreatitis with the TNF-α-CD45 axis and confirm that metformin is an appropriate drug in pancreatic injury rats. Our results were thus consistent with our working hypothesis mentioned above. These are in agreement with previous reports that demonstrated (i) High serum levels of amylase and LDH are well-known biomarkers of pancreatic injury19, (ii) Inflammation is a crucial step in the pathogenesis of acute pancreatitis18, (iii) Pancreatitis decreases the tissue levels of AMPK20 and in cell signalling, AMPK is located downstream of TNF-α21 and (iv) TNF-α induces leukocyte transmigration22 and leukocyte infiltration is documented in acute pancreatitis23.
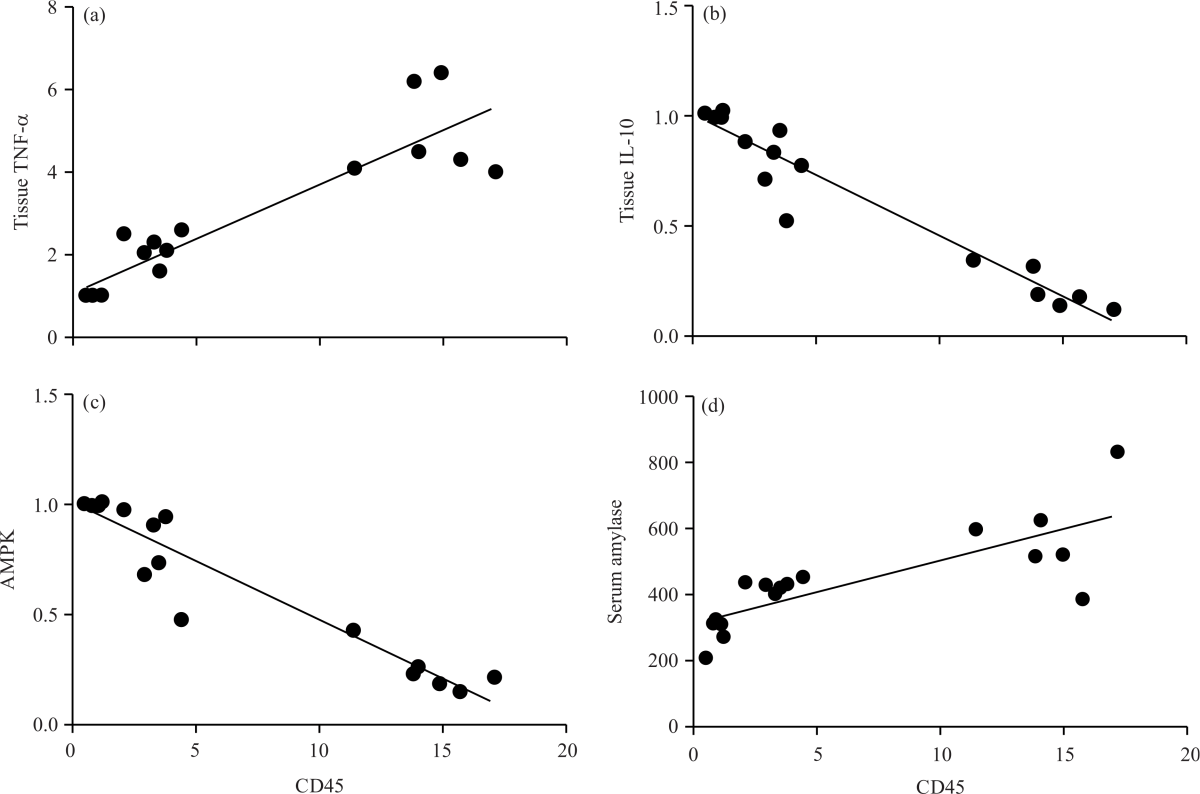 |
| Fig. 5(a-d): | Correlation between pancreatic leukocyte infiltration score and inflammation, AMPK and tissue injury biomarkers, degree of leukocyte infiltration measured as CD45 protein expression in all groups of rats after experiment completion. Positive correlation between CD5 and tissue TNF-α and serum amylase are shown in (a) and (d), respectively. Whereas, negative correlation between CD5 and tissue IL-10 and AMPK are shown in (b) and (c), respectively |
Beneficial effects of metformin were reported to inhibit the progression of pancreatic cancer in diabetic24 and non-diabetic patients15 and in sitagliptin-induced pancreatitis in diabetic rats25. These reports are consistent with our data that showed the ability of metformin to treat pancreatic injury.
However, contrasting reports are in disagreement with our findings. For example, a therapeutic dose of metformin caused acute pancreatitis in (i) An old female patient who had a history of Type 2 Diabetes Mellitus (T2DM), ischemic heart disease and previous myocardial infarction26 and (ii) A young “healthy” male with T2DM27.
The direct damaging effects of pancreatic proteinases such as trypsin “overflow” and inflammation are known to be involved in the pathology of acute pancreatitis in humans3 and animals injected with L-arg8,28 that lead to tissue necrosis. A link between these parameters, inflammation, necrosis and severe acute pancreatitis was documented as demonstrated by the augmentation of the inflammatory cytokine TNF-α and biomarkers of tissue necrosis, amylase and LDH in acute pancreatic injury29,30. On the other hand, AMPK and the anti-inflammatory cytokine IL-10 were reported to be ameliorated in patients with severe acute pancreatitis and pancreatic cancer20,31,32. Also, the severity of acute pancreatitis is higher in knock-out mice for IL-1033. In addition, bile-pancreatic duct obstruction-induced acute pancreatitis caused an elevation of pancreatic CD45 expression and TNF-α production, which were inhibited by the antioxidant, N-acetylcysteine34. Furthermore, the anti-inflammatory compound resveratrol was reported to inhibit L-arg-induced acute necrotizing pancreatitis30. These reports are in agreement with our findings of elevated levels of TNF-α, amylase and LDH, CD45 and inhibiting levels of AMPK and IL-10 in L-arg-induced AP, which were all protected with metformin. These results demonstrate in animals that metformin can overcome several of the pathogenic mechanisms associated with AP, which show the possibility that metformin has the potential to be further investigated as a therapeutic agent for AP.
Collectively, our data support the conclusion that the antidiabetic and anti-inflammatory drug, metformin inhibits the infiltration of inflammatory cells into the pancreas and also ameliorates biomarkers of pancreatic injury for 17 days in a rat model of acute pancreatitis induced by L-arg. In addition, metformin augments AMPK levels in pancreatic tissue. We also demonstrated an association between these parameters as well as the pancreas injury biomarker.
This study represents a significant contribution in the study of acute pancreatic injury induced by L-arginine (L-arg) in rats since it demonstrates that (i) L-arg-induced Acute Pancreatitis (AP) modulated pancreatic TNF-α-CD45 axis that was protected with metformin, (ii) Metformin protects against AMPK inhibition induced by L-arg and (iii) A significant correlation between leukocyte infiltrations measured as CD5 protein expression and inflammation, AMPK and pancreatic injury.
This work was supported by the Research Deanship of King Khalid University, Abha, Saudi Arabia; Grant number KKU-Project No. R.G.P1./44/42. The author would like to thank Professor Bahjat Al-Ani from the Department of Physiology, College of Medicine, King Khalid University, Abha, Saudi Arabia, Professor Mohamed A. Haidara and Dr. Samaa S. Kamar from the Physiology and Medical Histology Departments, Kasr Al-Aini Faculty of Medicine, Cairo University, Cairo, Egypt for their input and help during the preparation of this manuscript. We are also grateful to Dr. Mariam Al-Ani from Face Studio Clinic, 273 Hagley Road, Birmingham, B16 9NB, UK for proofreading the manuscript.