ABSTRACT
Concurrent therapy of herbs with conventional regimen has been extensively reported for alleviating morbidities such as diabetes and cardiac manifestations. One of unique natural remedy that carry abundant potential for ameliorating wide varieties of ailment is garlic. Present research was designed to explore the role of garlic in obviating myocardial dysfunction in diabetic animals, when combined with ramipril and/or gliclazide. Diabetic Sprague Dawley rats received garlic (250 mg kg-1), ramipril (1 mg kg-1) and gliclazide (10 mg kg-1) orally either alone or in combination in their respective groups. At the end of treatment, heart was excised; mounted on modified Langendorff’s set-up; perfused with Krebs–Henseleit solution and subjected to ischemia reperfusion injury. Significantly increased percentage recovery was recorded in heart rate and developed tension during post-ischemia in all treated animals, when compared to diabetic group. Further, depleted Superoxide Dismutase (SOD) and catalase activities were substantially inclined in treated groups. Furthermore, combination of garlic with ramipril was most effective in reverting the ischemic damage, whereas, garlic with gliclazide prevent beta cell degeneration. Moreover, histopathological observations validated biochemical and hemodynamic findings. To conclude, this preliminary observation in animals reveals the beneficial role of adding garlic to conventional regimen of gliclazide and ramipril. Further, a careful clinical evaluation of above combination would provide us an opportunity to standardize and implement in cardiovascular disease management in diabetic patient.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ijp.2015.579.587
URL: https://scialert.net/abstract/?doi=ijp.2015.579.587
INTRODUCTION
Diabetes mellitus is a complex metabolic disorder characterized by hyperglycemia often accompanied by glycosuria, polydipsia and polyuria (Celik et al., 2002; Sailaja et al., 2003). Cardiovascular disease is a major complication of diabetes and the leading cause of early death among people with diabetes. The United Kingdom Prospective Diabetes Study (Adler et al., 2000), the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications (Nathan et al., 2005) study and other clinical trials have demonstrated the benefits of optimal control of the blood glucose, blood pressure and cholesterol to reduce the risk of mortality. Loss of glycemic control is detrimental for prevention of cardiovascular diseases. Thus it is worthwhile to elucidate methods and strategies that can curtail uncontrolled hyperglycemia and thereby prevent cardiovascular mortalities.
The use of herbs and herbal therapies has been documented globally especially in patients with chronic ailments including diabetes. The folklore use of herbs for various therapeutic claims and advance access to the electronic and cyber technology could be attributed for addition of herbal remedies to the conventional drugs with or without consent of their clinicians (Eisenberg et al., 1998). Therefore, these patients are exposed to the possible adverse herb-drug interaction. Thus it is imperative to promote credible research to elucidate interaction of most popularly herbal therapies with conventional drugs, when used concomitantly.
In addition to its nutritional values, garlic (Allium sativum) has numerous applications in medicine (Asdaq and Inamdar, 2009). Garlic can reduce blood sugar levels (Sheela et al., 1995; Augusti and Sheela, 1996). It is richer than any other food in adenosine, a nucleic acid which is a building block of DNA and RNA. The primary anti-platelet constituent found in garlic appears to be adenosine (Makheja and Bailey, 1990) which attributes a major role in myocardial infarction. Based on the above reports and other data from recent literature, garlic is placed as unique substance exhibiting both antidiabetic and cardioprotective potencies. Therefore garlic is considered as possible traditional remedy for cardiovascular diseases in diabetic conditions.
Gliclazide, a second generation sulfonylurea has been reported to scavenge free radicals in in vitro analysis (Scott et al., 1991). It decreases blood glucose level by accelerating insulin release from pancreatic islets. Apart from having a hypoglycemic potential, it has also observed to carry anti-oxidant and platelet inhibiting activities (Jennings and Belch, 2000).
Large number of recent discoveries demonstrated the influence of renin-angiotensin system in the mechanism of cardiovascular morbidities that occur due to ischemia reperfusion damage (Linz et al., 1987; Pabla and Curtis, 1995; Neves et al., 1997). The cardiotoxic nature of angiotensin II especially in diabetic cardiovascular diseases are well understood (Hardy et al., 2001). Also, beneficial role of angiotensin converting enzyme inhibitors are well documented in ameliorating ischemia-reperfusion induced cardiovascular injury (Martinelli and Ceriello, 2003).
As evident from above, both gliclazide and ramipril are reported to alleviate cardiovascular morbidities in diabetic subjects and garlic is claimed and confirmed for its potency against cardiotoxicity in diabetic conditions. Thus the research envisaged was done to elucidate the impact of gliclazide and ramipril on chronic administration of garlic in diabetic animals subjected to ischemia reperfusion induced myocardial dysfunction.
MATERIALS AND METHODS
Chemicals: Garlic was purchased from local vegetable market of Bangalore, India. While, gliclazide and ramipril were procured from Dr. Reddys laboratory Ltd. and Micro labs company Ltd, respectively. All other chemicals and reagents were of analytical grade and were purchased from standard companies.
Preparation of garlic homogenate: The cloves of garlic were peeled, sliced and ground into a paste and suspended in distilled water. The Garlic Homogenate (GH) was prepared, 0.1 g mL–1, corresponding to 250 mg kg–1 body weight of animal (Asdaq and Inamdar, 2009). The Aqueous GH were administered by oral gavage everyday within 30 min of preparation.
Animals: Male Sprague Dawley rats weighing around 200-250 g were housed at 25±5°C in a well-ventilated animal house under 12 h light:12 h dark cycle, where they had free access to food (std pellet diet), water ad libitum. The animals were maintained under standard conditions in an animal house as per the guidelines of Committee for the Purpose of Control and Supervision on Experiments on Animals (CPCSEA). The Institutional ethical committee approved the experimental protocol (KCP/IAEC-27/2009-10).
Induction of diabetes: The Male Sprague Dawley albino rats weighing 200-250 g were allowed for fasting for 24 h prior to experimentation. A baseline fasting blood glucose levels were measured before injecting alloxan with Accu Chek Active Glucose Meter (Made in Germany) and made diabetic by injecting alloxan monohydrate 85 mg kg–1 body weight (single dose, intraperitoneally). A blood sugar level more than 300 mg dL–1 were considered diabetic at 18 h post injection of Alloxan monohydrate. Subsequently, animals were monitored for four days for establishing diabetes and taken for experimental purpose.
Experimental design: Once diabetes is induced, rats were randomly allocated into nine groups and treated orally as per the following protocol. Group I and II received normal vehicle and kept as normal and diabetic control, respectively. Group III was treated with GH (250 mg kg–1) (Asdaq and Inamdar, 2010) for 30 days, Group IV was given gliclazide (GLI-10 mg kg–1) for 10 days, Group V was administered ramipril (RAM-1 mg kg–1) (Linz et al., 1992) for 10 days, Group-VI and VII were administered with GH for 30 days with gliclazide and ramipril for the last 10 days respectively, Group-VIII received gliclazide as well as ramipril for 10 days concurrently and Group-IX was treated with GH for 30 days together with gliclazide and ramipril for the last 10 days.
Oral glucose tolerance test: At the end of treatment, rats were subjected to Oral Glucose Tolerance Test (OGTT). Glucose tolerance test was carried out using the method described elsewhere (Babu et al., 2002). Glucose (2 g kg–1) was administered to 8 h fasted rats. Blood samples were collected from the tail vein at 0, 15, 30, 60 and 120 min after oral glucose administration. Blood glucose levels were measured using Glucometer (Accuchek-Active).
Ischemic reperfusion injury: An animal model of myocardial infarction was done according to the method previously described with slight modifications (Walter et al., 2004). At the end of treatment, the diabetic rat heart were excised from deeply anesthetized rat (35 mg kg–1 sodium pentobarbitone, i.p) and perfused with Krebs-Henseleit solution and mounted on modified Langendorff’s preparation. After 15 min equilibration, records were taken for a control period of 15 min and then followed 15 min global ischemia and reperfusion. The normal flow rate (5 mL min–1) was subsequently restored and perfusion was continued for a further 30 min after reperfusion. Recovery in terms of heart rate and developed tension were recorded. Cardiac markers such as CKMB and LDH were estimated by a method described by Asdaq and Inamdar (2010). Thiobarbituric Acid Reactive Substances (TBARS) (Wolf et al., 1986; Sedlakova et al., 2009) were measured as a marker of lipid peroxidation and endogenous antioxidants, such as Superoxide Dismutase (SOD) (Singh and Athar, 2003) and catalase (Lowry et al., 1951) were estimated.
Histopathological studies: On the last day of the experiment, rats were anesthetized and then heart as well as pancreas were isolated and kept in 10% formaldehyde solution. Dehydration and clearing of the tissue is done automatically. The myocardial damage was determined by giving scores depending on the intensity as follows (Karthikeyan et al., 2007), (a) no changes-score, (b) mild-score, (focal myocytes damage or small multifocal degeneration with slight degree of inflammatory process), (c) moderate-score, (extensive myofibrillar degeneration and/or diffuse inflammatory process), marked-score and (d) (necrosis with diffuse inflammatory process).
Statistical analysis: The statistical significance was assessed using one-way analysis of variance (ANOVA) followed by Bonferoni comparison test. The values were expressed as Mean±SEM and p<0.05 was considered significant.
RESULTS
Oral glucose tolerance test: Treatment with GH significantly (p<0.001) improved oral glucose tolerance in diabetic rats. Under fasting conditions, administration of the GH did not significantly affect blood glucose concentrations within 30 min. After oral glucose loading, blood glucose concentrations were significantly (p<0.001) enhanced in all groups when compared to normal control. However, at the end of 120 min, a significant (p<0.001) decrease in blood glucose level was seen in all groups, which received gliclazide alone or in combination with ramipril or GH. The best decline was shown by group treated with GH with gliclazide and ramipril (Table 1).
Effect on SOD, Catalase, TBARS, developed tension and heart rate: As evident from Table 2, significant (p<0.001) decline in SOD and Catalase activities were observed in diabetic control, when compared to normal control. The administration of GH prophylactically helps in elevating these antioxidant activities, when compared to diabetic control. Concurrent treatment of GH with gliclazide and ramipril provided best inclination in antioxidant potentials. The TBARS level was depleted significantly (p<0.001) in all treated groups when compared to diabetic control and normal control.
A significant (p<0.001) recovery in developed tension and heart rate were recorded during post-ischemia in animals subjected to prior treatment of GH, gliclazide and ramipril.
| Table 1: | Effects of treatments on oral glucose tolerance test |
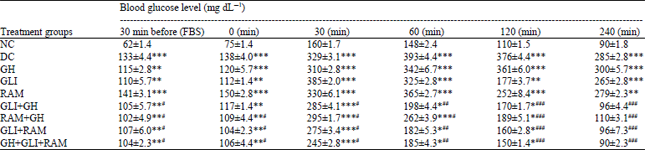 | |
All values are Mean±SEM, n = 6, *p<0.05, **p <0.01, ***p <0.001, when compared to NC: Normal control, #p<0.05, ##p <0.01, ###p <0.001, when compared to DC: Diabetic control, GH: Garlic homogenate (250 mg kg–1), GLI: Gliclazide (10 mg kg–1), RAM: Ramipril (1 mg kg–1) | |
| Table 2: | Effects of treatments on SOD, Catalase, TBARS, developed tension and heart rate |
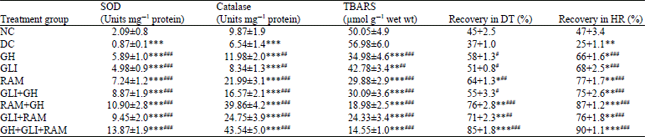 | |
All values are Mean±SEM, n = 6, *p<0.05, **p <0.01, ***p <0.001, when compared to NC: Normal control, #p<0.05, ##p <0.01, ###p <0.001, when compared to DC: Diabetic control, GH: Garlic homogenate (250 mg kg–1), GLI: Gliclazide (10 mg kg–1), RAM: Ramipril (1 mg kg–1), DT: Developed tension and HR: Heart rate | |
Similar to earlier findings of cardiac function, best recovery was found in groups of animals treated with ramipril alone or in conjunction with GH or both GH as well as gliclazide (Table 2).
Effect on LDH and CK-MB: As shown in Table 3, significant (p<0.001) increase in LDH and CK-MB activities were noted in the perfusate and vice versa in heart tissue homogenate in diabetic control group in comparison with normal control group. Chronic garlic administration with addition of ramipril in the last 10 days of GH treatment produced significant (p<0.001) recovery and decrease in biomarker activities in perfusate. Moreover, in groups subjected to triple therapy of GH with concurrent administration of gliclazide and ramipril, there were substantially enhanced activities of LDH and CK-MB activities in heart tissue homogenate were observed.
Effect on histological parameters: The microscopic section of heart muscles of diabetic control showed intervening fibrovascular septae (Fig. 1a). Chronic GH administration caused reduction in intervening fibrovascular septae and the cardiac muscle appears to be within normal limits (Fig. 1b). Further, administration of gliclazide demonstrated few scattered mononuclear inflammatory cells between the cardiac muscle fibres (Fig. 1c), while ramipril treatment showed only few areas of few areas of coagulative necrosis and scattered mononuclear inflammatory cells amidst the cardiac muscle (Fig. 1d).
| Table 3: | Effects of treatments on LDH and CK-MB |
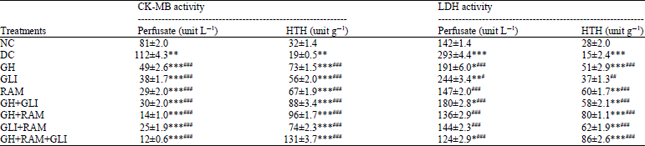 | |
| All values are Mean±SEM, n = 6, *p<0.05, **p <0.01, ***p <0.001, when compared to NC: Normal control, #p<0.05, ##p <0.01, ###p <0.001, when compared to DC: Diabetic control, GH: Garlic homogenate (250 mg kg–1), GLI: Gliclazide (10 mg kg–1), RAM: Ramipril (1 mg kg–1) | |
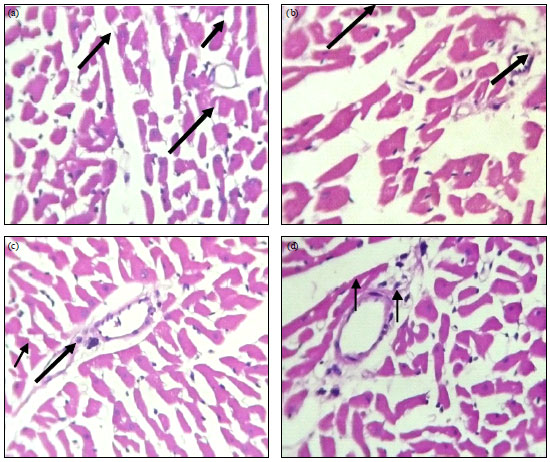 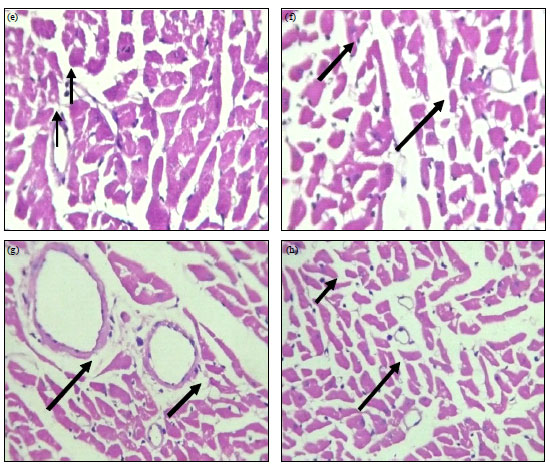 | |
| Fig. 1(a-h): | Haematoxylin and eosin stained section of heart (400X), (a) Diabetic control: Cardiac muscle with intervening fibrovascular septae. Also seen are scanty scattered mononuclear inflammatory cells between the cardiac muscle fibers, (b) GH: Myocardium comprising of cardiac muscle with intervening fibrovascular septae. The cardiac muscle appears to be within normal limits. Also seen are scanty scattered mononuclear inflammatory cells between the cardiac muscle fibers, (c) GLI 10 mg kg–1: Myocardium comprising of cardiac muscle with intervening fibrovascular septae. Also seen are scattered mononuclear inflammatory cells between the cardiac muscle fibers (Arrow), (d) RAM 1 mg kg–1: Myocardium shows cardiac muscle with intervening fibrovascular septae with coagulative necrosis (Arrow), few scattered mononuclear inflammatory cells (Arrow) amidst the cardiac muscle, (e) GH+GLI: Few cardiocytes show coagulative necrosis (Arrow). Also seen are scattered mononuclear inflammatory cells amidst the cardiac muscle, (f) GH+RAM: The cardiac muscle appears to be within normal limits (Arrow). Also seen are scanty scattered mononuclear inflammatory cells between the cardiac muscle fibers, (g) GLI+RAM: There are seen some areas of necrosis (arrow) and scattered mononuclear inflammatory cells (Arrow) amidst the cardiac muscle and (h) AGH+GLI+RAM: Section studied from the myocardium shows cardiac restoration of cardiac architecture with no areas of necrosis |
The combined therapy of GH with gliclazide or ramipril was more cardioprotective with substantially reduced coagulative necrosis (Fig. 1e and f). Upon combined administration of GH with gliclazide or ramipril, there was noticeable alleviation of coagulative necrosis and scattered mononuclear inflammatory cells (Fig. 1g). Best results were found with combined therapy of gliclazide and ramipril with chronic GH administration with restoration of normal architecture of heart (Fig. 1h).
The microscopic section studies from the pancreas of normal control (Fig. 2a) showed pancreatic lobules separated by connective tissue septa. The center of islet cells consist of small Beta-cells (80%, long-arrow) having basophilic granules, while the periphery comprises of large Alpha-cells (20%, short-arrow) having eosinophilic granules. The Diabetic control group (Fig. 2b) shows few degenerated Beta cells, center of islet cells consist of Beta-cells (60%, long-arrow) having basophilic granules while the periphery comprises of Alpha-cells (35%, long-arrow) having eosinophilic granules indicating the decrease of Beta cells when compared with normal control. On treatment with GH (Fig. 2c) and gliclazide together (Fig. 2d and f) or individually, rise in beta cells were found. Further, few degenerated beta cells are seen in groups treated with ramipril (Fig. 2e) that were reduced further in groups having combined administration of GH and ramipril (Fig. 2g). Moreover, concurrent administration of gliclazide with ramipril in animals under chronic GH treatment showed best protection against degeneration of Beta cell (80:15%) (Fig. 2i).
DISCUSSION
The purpose of the present study was to relate the cardioprotective potential and anti diabetic effect of Garlic Homogenate (GH) in diabetic animal subjected to ischemia reperfusion injury. The study was further extended to elucidate the pharmacodynamic interaction of GH in presence of conventional antidiabetic drug, gliclazide (GLI) and commonly prescribed cardioprotective agent, ramipril (RAM), in diabetic and nondiabetic rats. The GH ameliorated the myocardial damage in diabetic animal more significantly than in nondiabetic animals during ischemia reperfusion injury. Moreover, addition of RAM on chronic administration of GH, substantially augmented GH mediated cardioprotection. However, by incorporating GLI in the therapy of GH, a comparatively lesser protection to the myocardium was achieved. Corroborating with this, GH induced antidiabetic potency was augmented when combined with GLI or RAM at 2 h post glucose load. Concurrent administration of GH with GLI and RAM has profound effect on blood glucose lowering after 4 h.
GH has been believed to possess a hypoglycemic effect (Augusti and Sheela, 1996; Agarwal, 1996). Additionally, GH juice is reported to reverse hyperglycemia and alleviated oxidative stress and damage in liver and kidney in alloxan-induced diabetic rats (El-Demerdash et al., 2005). Moreover, it has been reported that GH acts as an insulin secretagogue in diabetic rats (Agarwal, 1996) and hence it can be combine with GLI to enhance the lowering of blood glucose level. The GH can effectively combine with compounds like cysteine and enhance serum insulin (Mathew and Augusti, 1973) and it acts as an antidiabetic agent by increasing either the pancreatic secretion of insulin from the beta cells or its release from bound insulin (Jain and Vyas, 1975). The enhanced therapeutic potential of combined therapy of GH with GLI might be attributed to synergistic beta cell stimulatory potential for insulin release. This finding emphasized the need for validating the dose of GLI when given to patient, who concurrently take garlic therapy. Garlic dose selected for the current study was moderate based on our earlier findings, hence, no significant hypoglycemia was noted.
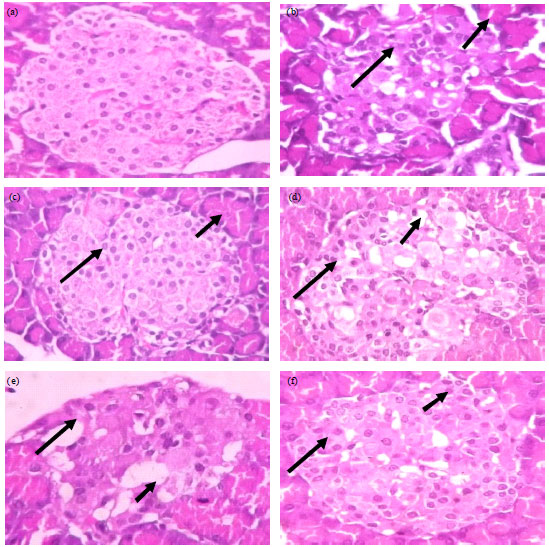 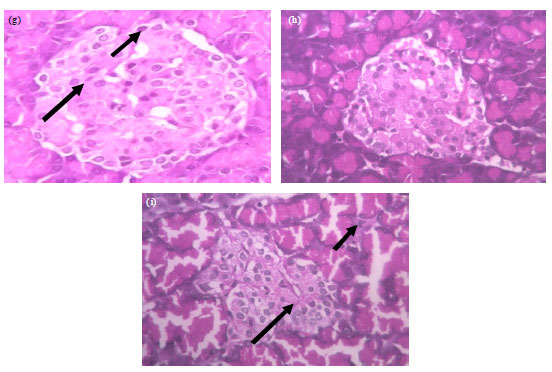 | |
| Fig. 2(a-i): | Haematoxylin and eosin stained section of pancreas (400X), (a) Normal control-section studied shows pancreatic lobules separated by connective tissue septa. The center of islet cells consist of small beta-cells (65%, long-arrow) having basophilic granules while the periphery comprises of large alpha-cells (30%, short-arrow) having eosinophilic granules, (b) Diabetic control-section studied shows pancreatic lobules separated by connective tissue septa. Most of the lobules show large areas of light-staining islets of langerhans. The center of islet cells consist of beta-cells (60%, long-arrow) having basophilic granules while the periphery comprises of alpha-cells (35%, long-arrow) having eosinophilic granules. Also seen are few degenerated beta cells, (c) GH: Center of islet cells consist of small beta-cells (70%, long-arrow) having basophilic granules while the periphery comprises of large alpha-cells (25%, short-arrow) having eosinophilic granules, (d) GLI 10 mg kg–1: The center of islet cells consist of aggregates of small beta-cells (70%, long-arrow) having basophilic granules while the periphery comprises of large alpha-cells (25%, short-arrow) having eosinophilic granules. Intervening these cells are seen thin walled congested capillaries, (e) RAM 1 mg kg–1: The center of islet cells consist of small beta-cells (55%, long-arrow) having basophilic granules while the periphery comprises of large alpha-cells (40%, short-arrow) having eosinophilic granules. Few degenerated beta cells are seen, (f) GH+GLI: The center of islet cells consist of beta-cells (80%, long-arrow) having basophilic granules while the periphery comprises of alpha-cells (15%, long-arrow) having eosinophilic granules, (g) GH+RAM: The center of islet cells consist of beta-cells (60%, Long-arrow) having basophilic granules while the periphery comprises of alpha-cells (45%, Short-arrow) having eosinophilic granules, (h) RAM+GLI: The pancreatic lobules consist largely of the exocrine acini and their intralobular ducts. Most of the lobules show small, round, light-staining islets of langerhans. The center of islet cells consist of aggregates of small beta-cells (65%) having basophilic granules while the periphery comprises of large alpha-cells (30%) having eosinophilic granules. Intervening these cells are seen thin walled capillaries and (i) GH+RAM+GLI: Some of the lobules show small, round, light-staining islets of langerhans. The center of islet cells consist of quantitative decrease in beta-cells (80%) having basophilic granules while the periphery comprises of large alpha-cells (15%) having eosinophilic granules. Also seen are scattered lymphocytes within the islet cells |
However, without substantial clinical trial for dose confirmation of garlic in human, this combination should be avoided for patient use.
The chronic administration of GH along with GLI and RAM caused a significant recovery in heart rate in post-ischemia. The GH was able to maintain the integrity of myocardium indicating its cardioprotective potential which has been previously reported to reduces a multitude of risk factors that play a decisive role in the genesis and progression of IHD (ischemic heart disease), e.g., hypolipidemic effect, lowering of arterial blood pressure and inhibition of platelet aggregation (Agarwal, 1996). In another study on the cardioprotective actions of GH in ischemia and reperfusion, GH augments ACE inhibiting efficacy of ACE inhibitor (Isensee et al., 1993). The renin-angiotensin system is present both in the circulation and in different tissues. Both the cardiac renin-angiotensin system and ACE, which catalyses the final step in the enzymatic cascade of renin-angiotensin system, appear to be activated in various conditions including myocardial ischemia (Hansen, 1995). The ACE activity elevation can act through decrease in bradykinin, nitric oxide and increase in free radicals, vascular hypertrophy, glomerular hypertrophy and renal fibrosis to produce diabetic disorders (Hansen, 1995). In diabetic rats the serum ACE activity decreased by GH, the compounds derived from the aqueous phase of the GH extract has attributed for ACE inhibitory effect (Suetsuna, 1998). Thus it might be possible that the active constituents such as s-allyl cysteine and other sulfur containing substance of GH may contribute for alleviating ACE activity and augments ramipril induced cardioprotection.
Diabetes mellitus causes many disorders such as cardiovascular diseases, nephropathy, neuropathy and retinopathy. Many factors can be involved in these disorders such as an increase of free radicals and decrease in antioxidants. During diabetes, the physiological response to combat oxidative stress is overwhelmed, resulting in an imbalance between pro-oxidative and anti-oxidative compounds. The augmentation of oxidative stress has been clearly documented in the pathogenesis of diabetic complications, including cardiomyopathy (Baynes and Thorpe, 1999). The ischemia-reperfusion model used in the current study also releases oxidative radicals (King and Loeken, 2004) similar to the oxidative radical formation in diabetes induced cardiovascular morbidities. The oxidative free radicals can be scavenged by elevated activities of catalase and Superoxide Dismutase (SOD). In the current study, chronic garlic administration with ramipril results in enhancing SOD and catalase.
CONCLUSION
To conclude, administration of GH lowered blood glucose level and concurrent administration with gliclazide augments its hypoglycemic potential. Similarly, GH demonstrates cardioprotective potential and concomitant treatment increased its efficacy. The findings support the rationale for intervention of exploring beneficial effects of dietary supplement such as GH along with conventional drugs like GLI and RAM. However, further studies should be carried out to explore the possibility of reducing GLI and RAM doses, while treating for diabetes induced cardiac manifestation in patients under traditional remedy with garlic.
ACKNOWLEDGMENTS
I extend my sincere thanks to Krupanidhi College of Pharmacy, Bangalore, India for the support offered by them in carrying out this study.
REFERENCES
- Adler, A.I., I.M. Stratton, H.A.W. Neil, J.S. Yudkin and D.R. Matthews et al., 2000. Association of systolic blood pressure with macrovascular and microvascular complications of type 2 diabetes (UKPDS 36): Prospective observational study Br. Med. J., 321: 412-419.
CrossRefDirect Link - Agarwal, K.C., 1996. Therapeutic actions of garlic constituents. Med. Res. Rev., 16: 111-124.
PubMedDirect Link - Asdaq, S.M. and M.N. Inamdar, 2010. Pharmacodynamic interaction of captopril with garlic in isoproterenol-induced myocardial damage in rat. Phytother. Res., 24: 720-725.
CrossRefPubMedDirect Link - Asdaq, S.M.B. and M.N. Inamdar, 2009. The potential for interaction of hydrochlorothiazide with garlic in rats. Chemico-Biol. Interactions, 181: 472-479.
CrossRefDirect Link - Augusti, K.T. and C.G. Sheela, 1996. Antiperoxide effect of S-allyl cysteine sulfoxide, an insulin secretagogue, in diabetic rats. Experientia, 52: 115-119.
CrossRef - Babu, V., D.T. Ganga and A. Subramonium, 2002. Anti-Hyperglycaemic activity of Cassia kleinii leaf extract in glucose fed normal rats and alloxan induced diabetic rats. Indian J. Pharmacol., 34: 409-415.
Direct Link - Baynes, J.W. and S.R. Thorpe, 1999. Role of oxidative stress in diabetic complications: A new perspective on an old paradigm. Diabetes, 48: 1-9.
CrossRefDirect Link - Celik, I., E. Yegin and F. Odabasoglu, 2002. Effect of experimental diabetes mellitus on plasma lactate dehydrogenase and glutamic oxaloacetic transaminase levels in rabbits. Turk. J. Biol., 26: 151-154.
Direct Link - Eisenberg, D.M., R.B. Davis, S.L. Ettner, S. Wilkey, M. van Rompay and R.C. Kessler, 1998. Trends in alternative medicine use in the United States, 1990-1997: Results of a follow-up national survey. J. Am. Med. Assoc., 280: 1569-1575.
PubMedDirect Link - El-Demerdash, F.M., M.I. Yousef and N.I.A. El-Naga, 2005. Biochemical study on the hypoglycemic effects of onion and garlic in alloxan-induced diabetic rats. Food Chem. Toxicol., 43: 57-63.
CrossRefPubMedDirect Link - Hansen, P.R., 1995. Myocardial reperfusion injury: Experimental evidence and clinical relevance. Eur. Heart J., 16: 734-740.
PubMed - Hardy, G., F. Stanke-Labesque, M. Peoch, A. Hakim and P. Devillier et al., 2001. Cysteinyl leukotrienes modulate angiotensin II constrictor effects on aortas from streptozotocin-induced diabetic rats. Arterioscler. Thromb. Vasc. Biol., 21: 1751-1758.
Direct Link - Isensee, H., B. Rietz and R. Jacob, 1993. Cardioprotective actions of garlic (Allium sativum). Arzneimittelforschung, 43: 94-98.
PubMed - Jain, R.C. and C.R. Vyas, 1975. Garlic in alloxan-induced diabetic rabbits. Am. J. Clin. Nutr., 28: 684-685.
PubMed - Jennings, P.E. and J.J.F. Belch, 2000. Free radical scavenging activity of sulfonylureas: A clinical assessment of the effect of gliclazide. Metabolism, 49: 23-26.
CrossRef - Karthikeyan, K., B.S. Bai and S.N. Devaraj, 2007. Cardioprotective effect of grape seed proanthocyanidins on isoproterenol-induced myocardial injury in rats. Int. J. Cardiol., 115: 326-333.
CrossRefDirect Link - King, G.L. and M.R. Loeken, 2004. Hyperglycemia-induced oxidative stress in diabetic complications. Histochem. Cell Biol., 122: 333-338.
CrossRefPubMedDirect Link - Linz, W., H.H. Lau, G. Beck and B.A. Schokens, 1987. Influence of the thromboxane synthetase inhibitor HOE 944, prostacyclin and indomethacin on reperfusion arrhythmias, cardiodynamics and metabolism in isolated ischemic rat hearts. Biomed. Biochim. Acta, 47: S23-S26.
Direct Link - Linz, W., J. Schaper, G. Wiemer, U. Albus and B.A. Scholkens, 1992. Ramipril prevents left ventricular hypertrophy with myocardial fibrosis without blood pressure reduction: A one year study in rats. Br. J. Pharmacol., 107: 970-975.
CrossRefDirect Link - Lowry, O.H., N.J. Rosebrough, A.L. Farr and R.J. Randall, 1951. Protein measurement with the folin phenol reagent. J. Biol. Chem., 193: 265-275.
CrossRefPubMedDirect Link - Makheja, A.N. and J.M. Bailey, 1990. Antiplatelet constituents of garlic and onion. Agents Actions, 29: 360-363.
PubMed - Martinelli, L. and A. Ceriello, 2003. [Mechanism of action of ACE-inhibitors and angiotensin receptor antagonists in diabetic nephropathy. Recent insights]. Rec. Prog. Med., 94: 131-135.
PubMed - Mathew, P.T. and K.T. Augusti, 1973. Studies on the effect of allicin (diallyl disulphide-oxide) on alloxan diabetes. I. Hypoglycaemic action and enhancement of serum insulin effect and glycogen synthesis. Indian J. Biochem. Biophys., 10: 209-212.
PubMed - Nathan, D.M., P.A. Cleary, J.Y. Backlund, S.M. Genuth and J.M. Lachin et al., 2005. Intensive diabetes treatment and cardiovascular disease in patients with type 1 diabetes. N. Eng. J. Med., 353: 2643-2653.
CrossRefPubMedDirect Link - Neves, L.A.A., A.P. Almeida, M.C. Khosla, M.J. Campagnole-Santos and R.A.S. Santos, 1997. Effect of angiotensin-(1-7) on reperfusion arrhythmias in isolated rat hearts. Braz. J. Med. Biol. Res., 30: 801-809.
CrossRef - Pabla, R. and M.J. Curtis, 1995. Effects of NO modulation on cardiac arrhythmias in the rat isolated heart. Circ. Res., 77: 984-992.
Direct Link - Sailaja, Y.R., R. Baskar and D. Saralakumari, 2003. The antioxidant status during maturation of reticulocytes to erythrocytes in type 2 diabetics. Free Radic. Biol. Med., 35: 133-139.
CrossRefPubMedDirect Link - Scott, N.A., P.E. Jennings, J. Brown and J.J. Belch, 1991. Gliclazide: A general free radical scavenger. Eur. J. Pharmacol.: Mol. Pharmacol., 208: 175-177.
CrossRef - Sedlakova, E., O. Racz, E. Lovasova, R. Beoaeka and M. Kurpas et al., 2009. Markers of oxidative stress in acute myocardial infarction treated by percutaneous coronary intervention. Central Eur. J. Med., 4: 26-31.
CrossRefDirect Link - Sheela, C.G., K. Kumud and K.T. Augusti, 1995. Anti-diabetic effects of onion and garlic sulfoxide amino acids in rats. Planta Medica, 61: 356-357.
CrossRefPubMedDirect Link - Singh, P.N. and M.S. Athar, 2003. Simlified calculation of mean QRS vector (mean electrical axis of heart) of electrocardiogram. Indian J. Physiol. Pharmacol., 47: 212-216.
Direct Link - Suetsuna, K., 1998. Isolation and characterization of angiotensin I-converting enzyme inhibitor dipeptides derived from Allium sativum L (garlic). J. Nutr. Biochem., 9: 415-419.
CrossRef - Walter, M.F., R.F. Jacob, B. Jeffers, M.M. Ghadanfar, G.M. Preston, J. Buch and R.P. Mason, 2004. Serum levels of thiobarbituric acid reactive substances predict cardiovascular events in patients with stable coronary artery disease: A longitudinal analysis of the prevent study. J. Am. Coll. Cardiol., 44: 1996-2002.
PubMed - Wolf, R.E., G.M. Graeber, J.R. Burge, J.L. DeShong, J.L. MacDonald and R. Zajtchuk, 1986. Evaluation of serum creatine kinase and lactate dehydrogenase in experimental myocardial infarction, atriotomies and thoracotomies. Ann. Thor. Surg., 41: 378-386.
CrossRef








