Research Article
Functional State of Liver Mitochondria During One Treatment with Benzonidazole-Rochagan®
Laboratory of Biophysics, Faculty of Sciences, National University of Colombia-Medell�n Branch, AA 3840 Medellin-Colombia
At the present the benzonidazole-Rochagan® is the unique drug used against Chagas’ disease (Turrens et al., 1996; Marr and Docampo, 1986), a parasitic disease afflicting several millions of Latin Americans (Weir, 2006). The clinic treatment with benzonidazole has to be prolonged and has very toxic effects for different organs (Rodrigues and de Castro, 2002; Castro and Díaz de Toranzo, 1988). The cellular mechanisms of different tissue lesions during benzonidazole treatment are not well known (Castro and Díaz de Toranzo, 1988).
Mitochondria are the cellular organels that transforme and store a considerable part of the energy, which is utilized by the cell for its functions; for this reason mitochondria play a crucial role at the cellular physiology. Several investigations have already showed that mitochondria from different organs are the target of the action of several drugs and toxic agents (Amacher, 2005). Based on these facts, it is very important to know if benzonidazole administration implies negative alterations on mitochondrial functionality and so explaining part of the toxic effect of benzonidazole treatment. Then, the current investigation was aimed at studying the functional state of liver mitochondria during one treatment with benzonidazole-Rochagan®. The studied mitochondrial bioenergetic parameters were the state 4, the state 3, the respiratory control (RC), the efficiency of oxidative phosphorylation (ADP:O), the mitochondrial transmembrane electrical potential (ΔΨ) and the activity of the mitochondrial ATPsynthase.
Benzonidazole administration: Benzonidazole ( N-benzyl-2-nitroimidazoyl acetamide, Rochagan®, Roche) was prepared daily by trituration and suspension of the tablet in distilled water. The therapeutic scheme with benzonidazole was according to (de Souza et al., 2000), in which the drug was given orally (100 mg benzonidazole/kg body weight) male adult Sprague-Dawley rats for nine consecutive days. Control animals receive only distilled water (the same daily volume than treated animals) for nine consecutive days. All animals were cared for in accordance with the Guide for the Care and Use of Laboratory Animals (1996, published by National Academy Press, 2101 Constitution Ave. NW, Washington, DC 20055, USA).
Preparation of mitochondria and assay of mitochondrial bioenergetic parameters: Rat liver mitochondria were isolated from adult male Sprague-Dawley rats, fasted overnight according to Lemeshko (2002). State 4, state 3, RC and ADP:O were assayed by means of the rates of oxygen consumption, which were determined with a Clark oxygen electrode in the basal medium 250 mM Sucrose, 5 mM KH2PO4, 10 mM HEPES, 0.1 mM EGTA, pH 7.2; this medium was supplemented with 5 mM succinate and 2.5 μM rotenone. ΔΨ was measured by monitoring the fluorescence of safranine O (520 /580 nm) according to Lemeshko (2002), using the above mentioned basal medium. The activity of mitochondrial ATPsynthase was estimated by a potentiometric method according to the Nishimura et al. (1962), using the assay buffer 250 mM sucrose, 5 mM KH2PO4, 1 mM HEPES, 0.1 mM EGTA, pH 7.2. All experiments were carried out at 30°C using 1 mg of mitochondrial protein/mL. Protein was determined by the biuret method (Gornall et al., 1949). All reagents were of analytical grade and were purchased from Sigma.
Statistical analysis: All values were means±SEM of eight independent experiments and their statistical significance (p<0.05) was evaluated using Student�s t-test. Statistical analysis was performed by GraphPad PRISM-version 2.0 software (San Diego-CA, USA).
At the present benzonidazole-Rochagan® is the only drug that is used in Latin America for the treatment of Chagas disease, a parasitic disease that is endemic in Latin America, affecting 16-18 million people, with more than 100 million exposed to the risk of infection. The parasite Trypanosoma cruzi is the etiological agent of Chagas disease. The mode action of benzonidazole could involve covalent bond or other interactions of nitroreduction intermediates with parasite components (Polak and Richle, 1978), or binding to DNA, lipids and proteins (Diaz de Toranzo et al., 1988). Benzonidazole treatment has very toxic effects (Rodrigues and de Castro, 2002; Castro and Díaz de Toranzo, 1988) and the cellular mechanisms of different tissue lesions during benzonidazole administration are not well known (Castro and Díaz de Toranzo, 1988). To our knowledge, at the present, no studies have investigated the mitochondrial energy-converting system upon benzonidazole administration.
The current benzonidazole therapeutic scheme was selected because this scheme of benzonidazole treatment, when it was used in one experimental mice model of T. cruzi infection, prevents the detection of parasites in the blood and controls parasite growth, favouring animal survival (de Souza et al., 2000).
In Fig. 1, it was showed the ratio of the liver weight, divided by the body weight from normal and benzonidazole-treated rats after the 9th day of the treatment; this ratio was 28% greater in treated rats than in control ones, reflecting that during this benzonidazole treatment the liver is a physiological target.
Table 1 showed the current studied mitochondrial bioenergetic parameters of liver mitochondria during one treatment with benzonidazole-Rochagan®.
Table 1 showed that benzonidazole administration did not decrease ΔΨ with respect to that of the control, indicating that it did not induce an increase in the passive membrane permeability to protons.
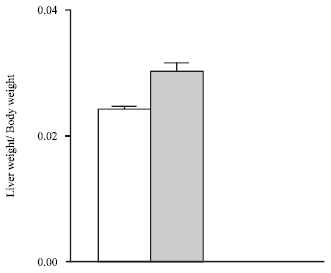 | |
| Fig. 1: | The ratio of liver weight, divided by body weight from normal and benzonidazole-treated rats after the 9th day of the treatment. Normal animals (white bar) and treated animals (black bar); all data were means±SEM (n = 8). There is a statistical difference between groups (p<0.001) |
| Table 1: | Functional state of liver mitochondria during one treatment with benzonidazole-Rochagan® |
 | |
| State 3 and 4 represent the oxygen consumption rates measured in the presence of 300 μM of exogenous ADP and in its absence, respectively. Oxygen consumption rates expressed as nmol O/min x mg of mitochondrial protein. RC is the ratio between the oxygen consumption rates measured in the presence and in the absence of exogenous ADP. ΔΨ is the mitochondrial transmembrane electrical potential expressed in fluorescence a.u. ATPsynthase indicates the activity of mitochondrial ATPsynthase expressed as nmol Pi / min x mg of mitochondrial protein. The oxidative substrate was 5 mM succinate. Values are the means (±SEM) of eight independent experiments in duplicate. Key: (*) statistically different (p<0.03) from control | |
The activity of the mitochondrial ATPsynthase did not decrease upon benzonidazole administration, i.e., the mitochondria after benzonidazole treatment were able to rephosphorylate ADP with a rate sufficient to preserve the cellular mitochondrial energy supply. Indeed, the ADP/O ratio did not statistically decrease after benzonidazole administration, reflecting any important energy dissipation. Taken together, these data indicated that functional state of liver mitochondria was not negatively altered upon the current benzonidazole administration.
The data in the Table 1, display a statistical increase in RC after benzonidazole treatment (11.8% of statistical difference). The RC is the ratio between state 3 and 4, it is therefore a hybrid parameter depending on a number of primary parameters (state 4 and 3). To know more about this mentioned dependence it is useful to look more carefully at the data of state 4 and 3. The Table 1 shows that the means of state 3 from control and treated groups are practically equal (1.9% of not statistical difference); for this reason the primary parameter of state 3 is not the main cause in the statistical increase of RC after benzonidazole treatment. On the contrary, the Table 1 shows that the mean of state 4 from control group is considerable higher than this one�s from treated group (9.2% of not statistical difference). Taken together, the primary parameter of state 4 is the main cause in the statistical increase of RC upon benzonidazole administration. In spite that state 4 and 3 are primary parameters, to know one of them or both (in one independent mitochondrial preparation) is not so informative; the most important thing, physiological speaking, is to know the state 3 with respect to the state 4 (i.e., RC) for one independent mitochondrial preparation. It is in this sense that RC is a mitochondrial parameter used for assessing the functional integrity of a mitochondria preparation and based on this fact, the current results indicate that upon the present benzonidazole-Rochagan® administration liver mitochondria from treated animals maintenance better functionality than this one�s from control animals.
The mitochondrial respiration when no substrate is present (i.e., at state 4) is only because the inner mitochondrial membrane is not completely impermeable to protons, which can therefore slowly leak back across the inner mitochondrial membrane even in the absence of net ATP synthesis. This leak mostly is due to an endogenous proton leak, or to the presence of a specific uncoupling protein (Brooker et al., 1998; Adams et al., 2001). In spite that there was no statistical alteration of state 4 upon benzonidazole administration, but there was a tendency of this mitochondrial parameter to decrease, this primer parameter was the main cause in the statistical increase of RC after benzonidazole treatment; for this reason it was
possible that during the current benzonidazole administration it could take changes in the composition of the inner mitochondrial membrane, bioenergetically affecting only the factors that contribute to mitochondrial respiration at state 4 and implicating a statistical increase in RC.
It is possible that the above suggested inner mitochondrial membrane alterations take place in other cellular liver membranes, implicating some toxic effects of the benzonidazole administration. In this sense, it is important to note that it was observed (de Castro et al., 1992) lipid accumulation in the cytoplasm of rat agrenal cortex cells� during one benzonidazole treatment (a single dose of 100 mg kg-1), implicating possible alterations of lipid metabolism and of biomembrane composition too.
Taken together the last conclusion of the current investigation and the observation of de Castro et al. (1992), it will be very worthy to focus future investigations on the effect of benzonidazole treatment on diverse biological membranes from different organs. For example, the investigators (Marra et al., 2002) have shown alterations in the fatty-acid pattern of liver microsomal membranes from Trypanosoma cruzi infected mice by benzonidazole treatment (100 mg kg-1 day, during 30 days); unfortunately, these investigators did not study the effect of benzonidazole treatment on the fatty-acid pattern of liver microsomal membranes from uninfected mice; exactly this kind of study will be worthy to carry out and studying diverse biomembranes from different organs.
This study was supported by the financial contribution of COLCIENCIAS-Grant No. 1118-04-16552 and the National University of Colombia. The author wish to thank Dr. Armando Galeano, Coordinador de Vectores-Dirección Nacional de Salud de Antioquia, for his generous gift of the benzonidazole-Rochagan® tables.