ABSTRACT
The present study investigated the chemopreventive effects of the water extract of chlorella on the development and growth of DMBA-induced mammary tumors. Female rats were daily administered vehicle control or chlorella either at 0.5 g or at 1.0 g kg-1 body weight starting at age of 35 days and continued to the end of the experiment. At age of 50 days breast tumor was induced by administering DMBA at 25 mg kg-1 body weight. Similar DMBA dose was administered to DMBA-alone group at age of 50 days. As a control for chlorella treatment one group (chlorella-alone) was administered chlorella at 1.0 g kg-1 body weight starting at age of 35 days and continued to the end of the experiment. Animals were then followed for 15 weeks. Effects of chlorella on the expression of proliferating cell nuclear antigen (PCNA), p53 and estrogen receptor (ER) were investigated in mammary tissues of control and experimental groups using immunohistochemistry. Present data demonstrated that chlorella treatment restored the normal expression levels of PCNA and ER. Chlorella also significantly increased cell death as assessed by the terminal deoxynucleotidyl transferase-mediated triphoshate nick-end labeling (TUNEL) analysis. In conclusion, the protective role of chlorella’s water extract against carcinogen-induced breast cancer seems to be mediated through its anti-proliferative and pro-apoptotic properties.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ijcr.2009.12.24
URL: https://scialert.net/abstract/?doi=ijcr.2009.12.24
INTRODUCTION
Breast cancer is the most common cancer and cause of death in women and makes up one tenth of all new cancer diagnoses worldwide (Bray et al., 2004; Ray et al., 2007). Development of mammary tumors requires aberrant accretion of cells caused by excessive proliferation, insufficient apoptosis or dysregulation of cellular differentiation (Kumaraguruparan et al., 2006; Hanahan and Weinberg, 2000). Recently, considerable attention has been focused on identifying naturally occurring chemopreventive substances capable of inhibiting, hindering, or reversing the multi-stage carcinogenesis. The main objective of breast cancer chemoprevention research is to advance knowledge in identifying and characterizing entities that might reduce the risk of human population developing cancer. Therefore, it is of interest to explore the possibility of using phytochemicals or other dietary chemicals as chemopreventive agents. Furthermore, the study of the biological effects of these phytochemicals at cellular level provides the molecular basis of their anti-disease function. It also helps to establish the platform for generating more potent chemopreventive and even chemotherapeutic agents (Gosslau and Yu-Chen, 2004).
Unicellular green alga, Chlorella vulgaris, has been shown to express various pharmacological effects in animals and humans (Hasegawa et al., 2005). Hot water extracts of Chlorella vulgaris contain potent biological modifiers for immune responses against tumors, bacteria, viruses and native antigens such as casein (Hasegawa et al., 1999). Chlorella has also been reported to possess anti-oxidative, anti-inflammatory and anti-tumor properties in vitro (Guzman et al., 2001). In addition, chemopreventive effects of chlorella were reported against cancer such as hepatocarcinogenesis in rats (Takekoshi et al., 2005). Thus, the benefits of the chlorella organism are shown to be very wide-ranging and it has the potential to be used in nutritional fortification (Hasegawa et al., 2005).
Estrogens are known to modulate proliferative activity not only in the classic estrogen responsive tissues (Yager and Liehr, 1996) but also in organs and/or cells of other apparatuses (Fisher et al., 1984; Francavilla et al., 1989; Messa et al., 2000; Barone et al., 2006). They exert their biological activity by binding with two type of receptors: estrogen receptor-alpha (ER-α), the prevalent form in the breast, bone, cardiovascular tissue, urogenital tract and central nervous system and estrogen receptor-beta (ER-β), the prevalent form in the gut (Di Leo et al., 2008; Gustafsson, 1999; Dechering et al., 2000). ER mediates the action of estrogens and plays a major role in the initiation and progression of breast cancer (Pearce and Jordan, 2004).
p53, the protein encoded by the tumour suppressor gene p53, functions as a master regulator of both cell division and apoptosis (Mihara et al., 2003).
The rat mammary gland is a widely used model for studying the pathogenesis, therapy and chemoprevention of breast cancer (Amin and Buratovich, 2007; Nandi et al., 1995; Russo et al., 1990; Welsch, 1985). Rat mammary gland carcinomas resemble human breast cancer in histopathogenesis, pathological features and hormone-dependence. The present study investigated the effect of chlorella on proliferation, apoptosis and expression of estrogen receptors in breast epithelium in a DMBA-induced mammary tumour in rats model system (Amin et al., 2005). Immunohistochemistry was used to detect expressions of cell proliferating marker, PCNA, ER and p53. Apoptosis was also determined by terminal deoxynucleotide transferase dUTP nick labeling (TUNEL) staining.
MATERIALS AND METHODS
Chemicals
DMBA and olive oil were purchased from (Sigma-Aldrich, St. Louis MO, USA). Chlorella was purchased as tablets from Wakunaga of America CO., LTD. Mission Viejo, CA, USA.
Animals and Experimental Protocol
Five-week-old female Wistar rats (100-150 g body weight) were divided into five groups of seven rats each. Rats were housed in a pathogen-free environment and under constant environmental conditions (photoperiod, temperature, air humidity, food) at the animal house of the Faculty of Medicine in UAE University. Female rats were daily administered vehicle control or chlorella either at 0.5 g or at 1.0 g kg-1 body weight starting at age of 35 days and continued to the end of the experiment. Breast tumor was then induced by administering DMBA- dissolved in 1 mL vehicle (0.5 mL of olive oil and 0.5 mL of saline) (Amin et al., 2005; Costa et al., 2002) at 25 mg kg-1 body weight at age of 50 days. As a control for chlorella treatment one group (chlorella-alone) was administered chlorella at 1.0 g kg-1 body weight starting at age of 35 days and continued to the end of the experiment. Similar DMBA dose was administered to DMBA-alone group at age of 50 days. Animals were then followed for 15 weeks post DMBA treatment. The chlorella was obtained in the form of supplement tablets of 100% pure broken cell wall chlorella powder. Before oral administration, chlorella tablets were ground and suspended in warm water. The rats were palpated for tumor detection twice weekly throughout the hundred and twenty-day experimental period. At the end of the experimental period, all rats were alive which were anesthetized with diethyl ether and sacrificed by decapitation. The mammary tissues were dissected out and were then fixed in 10% buffered formalin. Doses of chlorella used in this study were confirmed to be most suitable and effective in tested rats according to preliminary experiments. This study was conducted after the approval of the Animal Research Ethics Committee of UAE University, UAE.
Histology
Histological examination of rat mammary tissues from rats of control, DMBA-alone and chlorella +DMBA treated groups were performed as described by Samy et al. (2006). Briefly, small pieces of mammary tissue, fixed in 10% phosphate-buffered formalin, were embedded in paraffin. The blocks were cut to obtain 5 μm thick sections and stained with hematoxylin-eosin sections were examined using a Leica DMRB/E light microscope (Heerbrugg, Switzerland).
Tunel Assay
Apoptosis was assessed in deparaffinized sections using TUNEL technique as per manufacturer’s protocol (Chemicon International, Temecula, CA, USA). This method detects the DNA fragmentation associated with apoptosis by labeling 3-OH DNA termini with digoxigenin nucleotides, a process facilitated by terminal deoxynucleotidyl transferase. The labeled fragments are then allowed to bind to anti-digoxigenin antibody conjugated with peroxidase. Color was developed by adding sufficient peroxidase substrate to specimens.
Immunohistochemistry
Sections of 5-6μm were cut and mounted for immunostaining. Sections were deparaffinized in xylene, rehydrated and rinsed in 0.1M phosphate buffered saline (PBS, pH 7.2). Endogenous peroxidase was blocked by incubating tissue sections in 3% hydrogen peroxide for 20 min at ambient temperature to diminish non-specific staining. Sections were rinsed in PBS and further nonspecific binding was blocked by additional incubation of tissue sections with normal goat serum for 20 min and diluted in PBS for tenmin at room temperature. With the removal of excess normal sera, the sections were incubated at 4 °C overnight with primary antibodies. All primary antibodies were diluted in 0.1 BSA and 0.01% sodium azide dissolved in PBS. Subsequent to incubation, sections were washed in PBS (3x5min), incubated for 1h at 4 °C with avidin-biotinylated peroxidase complex (1/400; Dako, Glostrup, Denmark), rinsed in PBS as above. Following PBS washes, the sections were rinsed in acetate buffer for tenmin and the peroxidase reaction developed in a 0.05% solution of 3,3’-diaminobenzidine tetrahydrochloride (DAB: (Sigma-Aldrich, St. Louis MO, USA)), 0.03% hydrogen peroxide and imidazole in Tris-HCl buffer (pH 7.6). Sections were counterstained with hematoxylin. The number of immunoreactively stained cells in each section was calculated as explained below.
Semiquantitative Analysis of Immunoreactive Cells
A semiquantitative procedure used to count the number of immunoreactive cells is described by Weaver et al. (2007) and Weaver and Liu (2008). Briefly, the immunocytochemically stained mammary tissues of the four different treatments, including control chlorella-alone, DMBA-alone and chlorella + DMBA treated groups, were assessed for the degree of staining for each primary antibody. A total of 100 cells were scored per site. Three sites per section were randomly selected from each of the sections representative of each parameter. This yielded 21 sampled sites per treatment (n = 7). The total number of immunoreactively stained cells recorded for each treatment is presented as a percentage of the total number of cells counted.
Statistical Analysis
SPSS (version 10) statistical program (SPSS Inc., Chicago, IL, USA) was used for statistical analysis; all data are expressed as group Mean ± SE. ANOVA was used to detect differences between various groups. If a significant difference was found between the means of the treated and control groups, Dunnett’s t test was applied.
RESULTS
All tumors observed at autopsy were encapsulated and of solid consistency. There was no evidence of acute toxicity after the administration of DMBA or chlorella extracts. No mammary tumor development was seen in the control group during the experimental period. First tumors were recorded 100 days post DMBA administration in all groups and then the incidences increased time-dependently throughout the experiment. Histologically, mammary tumors in all DMBA-treated groups represented benign lesions that ranged from florid epithelial hyperplasia to fibroadenomas (Fig. 1). The extent of changes for all tested markers was listed in (Table 1). Breast tissues of the group treated with chlorella-alone appeared normal. Chlorella preparations associated with the prevention and treatment of cancer has been shown to be mediated via the immune system rather than its direct toxicity against tumors (Kralovec et al., 2005).
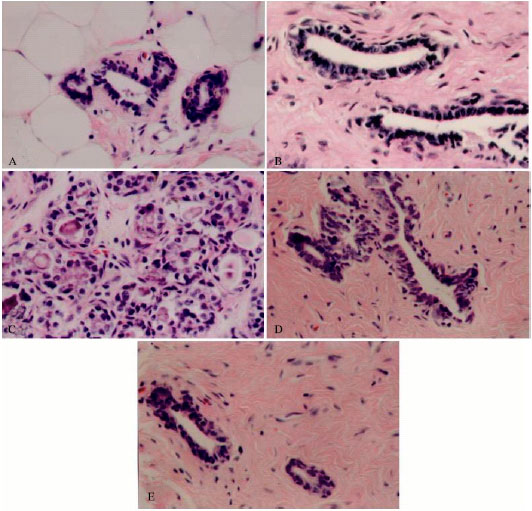 | |
| Fig. 1: | Photomicrographs of mammary tissues of rats treated with vehicle (A, control), chlorella-alone (B, chlorella control), DMBA-alone (C, induced), DMBA + chlorella at 0.5 g kg-1 b. wt. (D), and DMBA + chlorella at 1.0 g kg-1 b. wt. (E). (A-B) Normal ductal structures of the mammary gland. (C) Florid epithelial hyperplasia of DMBA-induced breast tumor with secondary lumina and increased numbers of active cells both at the basal lamina and away from it. (D-E) Mammary ducts with mild hyperplasia after DMBA-treated rats were fed with chlorella extract. Hematoxylin and Eosin staining, 400X |
| Table 1: | The expression of PCNA, ER, TUNEL and p53 in breast tissues of control, chlorella-alone, DMBA-induced and chlorella-protected groups. The percent of PCNA, ER and p53 positive cells in each section was calculated as described in the materials and methods |
 | |
| (Means ± SD; n = 7), ††† p<0.001, † p<0.05 compared with DMBA-alone group, ***p<0.001, *p<0.05, compared with control group | |
 | |
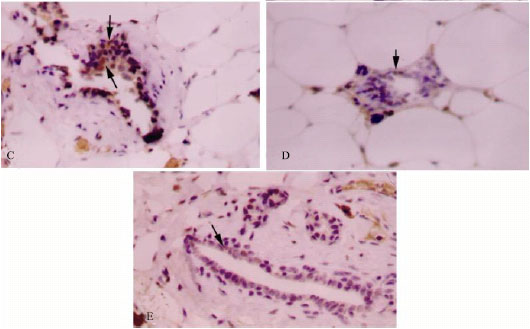
| Fig. 2: | Immunohistochemical staining for PCNA in mammary tissues of rats treated with vehicle (A, control), chlorella-alone (B, chlorella control), DMBA-alone (C, induced), DMBA + chlorella at 0.5 g kg-1 b. wt. (D) and DMBA + chlorella at 1.0 g kg-1 b. wt. (E). Chlorella administration decreases the number of proliferating cells in mammary ducts (C-D). Counter stained with hematoxylin. Mouse monoclonal antibody (Dako, Glostrup, Denmark) was used as per manufacturer’s protocol. All images, 400X |
Antiproliferative Effect of Chlorella
Cell proliferation is regulated by multiple mechanisms. Proliferative cell nuclear antigen (PCNA), a nuclear protein present in proliferating cells, is essential for cell replication and acts as a marker for cellular proliferation (Malkas et al., 2006). Levels of PCNA both in control and chlorella-alone groups did not show any significant variations. DMBA significantly induced (536%) the PCNA expression compared to control PCNA levels. Treatment with chlorella extract significantly decreased (78.6%) the expression of PCNA in the DMBA + chlorella rats (p<0.001) compared to tumor tissue from animals treated with DMBA-alone (Table 1, Fig. 2). Furthermore, the lactiferous ducts in untreated-DMBA tissue were filled with proliferated cells; however, in the chlorella treated-groups these ducts were mostly clear of the proliferating cells.
Chlorella Modulates Expression of Estrogen Receptors
ER is another marker that is used extensively in breast cancer diagnosis and prognosis. Distribution of ERα in mammary tissues of control, chlorella-alone and DMBA-alone groups is shown in Fig. 3A-C, respectively. ER-immunoreactivity was considered positive only when a strong dark brown stain was detected within the nuclei of epithelial cells of mammary ducts. In control and chlorella-alone samples, some epithelial cells showed consistent nuclear immunoreactivity (16 and 14%, respectively) but occasional and weaker intra-cytoplasmic positivity was detectable as well (Table 1, Fig. 3). In DMBA-alone group, a significant increase (81%) of ERα immunoreactivity was observed. Pretreatments with chlorella abolished (51.7-58.6%) the DMBA-induced upregulation of ERα in mammary tissues (Fig. 3D-E).
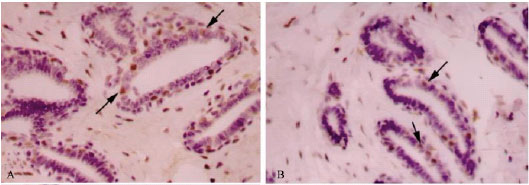 | |
| Fig. 3: | Immunohistochemcal staining of ER in mammary tissues of rats treated with vehicle (A, control), chlorella-alone (B, chlorella control), DMBA (C, induced), DMBA + chlorella at 0.5 g kg-1 b. wt. (D) and DMBA + chlorella at 1.0 g kg-1 b.wt. (E). Chlorella treatments decrease the number of ER-positive cells in tumor masses (D-E). Counter stained with hematoxylin. Rabbit polyclonal antibody (Dako, Glostrup, Denmark) was used as per manufacturer’s protocol. All images, 400X |
Proapoptotic Effects of Chlorella
As there was a significant reduction in the tumor size by the chlorella treatment, effects of different doses of chlorella on the tumor growth were examined using TUNEL assay. TUNEL assay was used to identify apoptotic cells of mammary ducts. Brown staining, indicating TUNEL-positive nuclei was visible in breast tissues of control, chlorella-alone and DMBA-alone animals (Fig. 4). It was interesting to notice a decrease in TUNEL-positive cells in animals treated with chlorella-alone (7% versus 10% in control). However, TUNEL-positive cells were significantly (p<0.001) increased in the DMBA-alone group (120%) compared to the control group (Fig. 4). Pre-administration of chlorella prior to and concomitant with DMBA treatment has retained the high number of TUNEL-positive cells in the DMBA-alone group (Fig. 4). It is worth mentioning here however that compared to DMBA-alone group, lower dose of chlorella caused much less increase of cell death (41%) compared to the higher dose (111%).
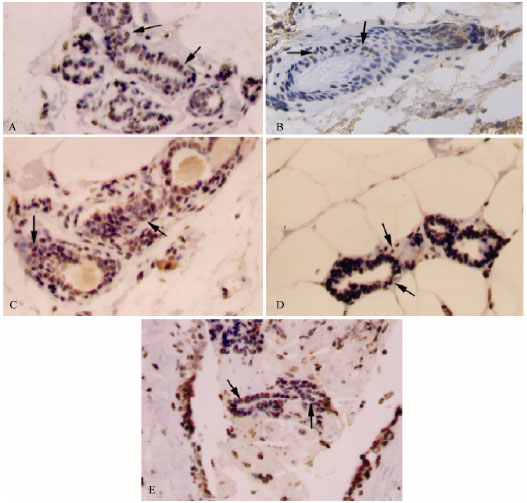 | |
| Fig. 4: | Immunohistochemcal staining for TUNEL in mammary tissues of rats treated with vehicle (A, control), chlorella-alone (B, chlorella control), DMBA-alone (C, induced), DMBA + chlorella at 0.5 g kg-1 b. wt. (D) and DMBA + chlorella at 1.0 g kg-1 b. wt. (E). A greater number of TUNEL positive cells were observed in tumors from chlorella treated animals (D-E). Brown staining indicates TUNEL-positive cells. Counter stained with hematoxylin. All images, 400X |
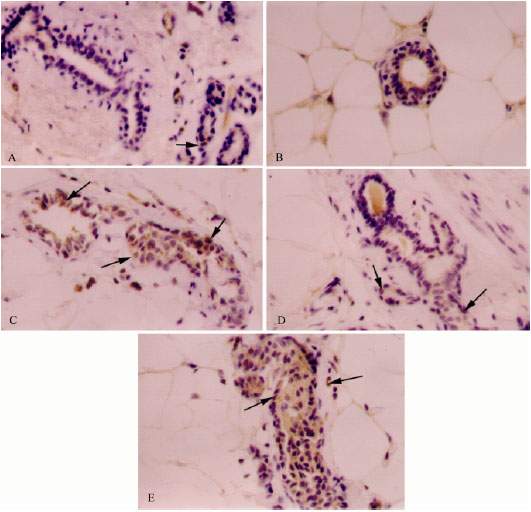 | |
| Fig. 5: | Immunohistochemcal staining for p53 in mammary tissues of rats treated with vehicle (A, control), chlorella-alone (B, chlorella control), DMBA-alone (C, induced), DMBA + chlorella at 0.5 g kg-1 b. wt. (D) and DMBA + chlorella at 1.0 g kg-1 b. wt. (E) Brown staining indicating positive immuno-stained cells increased in the DMBA-alone group (B). The concomitant treatment with chlorella before and after DMBA treatment decreased the number of p53-positive cells compared to the DMBA-alone group (D-E). Counter stained with hematoxylin. Mouse monoclonal antibody (Dako, Glostrup, Denmark) was used as per manufacturer’s protocol. All images, 400X |
Effect of Chlorella on p53 Expression
p53 protein expression was detected in mammary tissues of control, chlorella-alone and DMBA-alone animals (Fig. 5A-C). Similar level of p53 expressing cells were reported in control and chlorella-alone treated animals. Brown staining, indicating positive immunostained cells, was significantly more (144%; p<0.001) in the DMBA-alone group compared to the control group (Fig. 5). Treatment with chlorella before and concomitant with DMBA uptake had a slight effect (8.4-15%) on the number of p53-positive cells compared to the DMBA-alone group (Fig. 5).
DISCUSSION
Despite significant advances in the treatment of breast cancer, this disease remains the leading cause of death and the most commonly diagnosed cancers among women (Jemal et al., 2005, 2008). Epidemiological data from more than 250 case control and cohort studies shows an inverse relationship between the risk of certain types of cancer and consumption of dietary phytochemicals and fibers (Borek, 2004). Multiple mechanisms have been identified for the anti-neoplastic effects of plants, including antioxidant, anti-inflammatory and anti-proliferative activities, inhibition of bio-activating enzymes and induction of detoxifying enzymes (Le Marchand, 2002). Chlorella’s antioxidative, anti-inflammatory and anti-tumor properties were reported by Guzman et al. (2001). In addition, chemopreventive effects of chlorella were reported against hepatocarcinogenesis in rats (Takekoshi et al., 2005).
Despite a large number of studies analyzing chlorella’s chemoprotective effects in vitro, a paucity of data is available on its in vivo mode of action. The present research demonstrates, in vivo, the anti-breast cancer effect of chlorella utilizing DMBA as a cancer inducer. DMBA, a polycyclic aromatic hydrocarbon, is genotoxic and is capable of forming carcinogen-DNA adducts in human or animal tissues. The development and progression of mammary carcinoma is influenced by estrogens that mediate their effects via. ER. ER pathway is involved in proliferation, angiogenesis and metastasis of cancer cells (Wesierska-Gadek et al., 2008). The estrogen–ER complex induces both genetic and epigenetic changes that influence the expression of a number of genes involved in the regulation of cell proliferation and differentiation (Murphy and Watson, 2002; Shaw et al., 2002). In this study, chlorella has significantly restored the control levels of ER. A number of studies including the present one have demonstrated the overexpression of ER in mammary tumours. ER-positive breast cancers have also been reported to be associated with evasion of apoptosis (Alfred et al., 2001; Baccouche et al., 2003; De Las Mulas et al., 2005).
Data presented in this study suggest that chlorella’s water extract is effective in suppressing the proliferation of mammary tumors as shown by growth inhibition and apoptosis induction. Many of the molecular alterations that accompany carcinogenesis lead to uncontrolled proliferation and growth and the ability of the transformed cells to prevaricate apoptosis. Elevated proliferative activity has been shown not only to be responsible for hyperplastic phenomena but also to facilitate tumour development by enhancing the probability of genomic mutations (Barone et al., 2003). Anticancer effects of some potential phytochemicals are known to be mediated through differential regulation of the cell cycle and subsequent events leading to cell death (Pozo-Guisado et al., 2002; Jackson and Singletary, 2004). Chlorella has a significant antiproliferative effect (Table 1, Fig. 2). Immunohistochemical staining showed a decrease in the PCNA and an increase in apoptosis in tumors from animal treated with chlorella. PCNA is synthesized in the G1 to S phase of the cell cycle. Expression of PCNA is known to be increased in rapidly proliferating tumours (Maga and Hubscher, 2003; Paunesku et al., 2001). A positive correlation between the number of PCNA-positive cells and histopathologic malignancy has been reported in different mammary tumours (Franzen et al., 1997; Funakoshi et al., 2000). Neoplastic cells undergo changes that diminish their susceptibility to apoptosis (Hersey and Zhang, 2003; Johnstone et al., 2002; Kaufmann and Vaux, 2003). Results presented herein shows that chlorella effectively suppresses the proliferation as well as induces apoptosis in DMBA-induced mammary tumors. The antiproliferative effect of chlorella extract has been shown to be associated with apoptosis-inducing activity of chlorella in different human cancer cell lines (Cha et al., 2008). Moreover, higher dose of chlorella has shown more cytotoxic effects in DMBA-induced breast cancer. Other studies have also shown that water extracts of chlorella induced antiprolifrative and apoptotic effects on activated hepatic stellate cells (Wu et al., 2005).
p53 is critical for the prevention of the onset and progression of breast cancer, overexpression of p53 can stop the cell cycle and causes DNA repair and apoptosis (Benz, 2008). Carcinogens-induced DNA damage, if not repaired, can lead to mutagenesis and tumor initiation (Sun et al., 2007; Cox, 1998). Following DNA damage, p53 can be activated leading to the repair of DNA damage or apoptosis. Chlorella is widely known as a health supplements (Mallick and Rai, 1999; Hasegawa et al., 2000) and is reported to have anti-tumor properties in vitro (Park et al., 2005). A growing body of evidence indicates a close correlation between the up-regulation of p53 and apoptosis in cancer cells treated with chemotherapeutic drugs (Gosslau et al., 2005). It is therefore quite intriguing to find that p53 is not required for the antiproliferative and pro-apoptotic action of chlorella in the present study (Table 1, Fig. 5). 2-chloroadenosine (a resistant analogue of adenosine) has been shown to induce apoptosis in vitro through the activation of the intrinsic pathway of apoptosis in a p53-independent way (Bastin-Coyette et al., 2008). Resveratrol has also been shown to induce apoptosis independent of p53 (Gosslau et al., 2008). It is therefore possible that p53 may not be the primary target for the proapoptotic action of chlorella extract in this study. Finally, the fact that the proapoptotic action of chlorella is p53-independent, emphasizes that chlorella is potentially useful to be developed into a chemotherapeutic agent against p53-resistant cancer cells.
In conclusion, the present study provides evidence for antiproliferative and pro-apoptotic capacities of chlorella against DMBA-induced-breast cancer in rats. Despite the growing number of studies that suggest a chemopreventive potential of chlorella, human intervention trials will remain to be the ultimate determinant of the aforementioned beneficial nature of this green alga.
ACKNOWLEDGMENTS
This study was financially supported by a grant from the Research Affairs Office at the UAE University; Grant No. 03-04-2-11/06. The author is grateful to Mr. A. Hamza, Mr. S. Daoud and Ms. N. Al-Kahloot for their valuable technical assistance throughout this study. The author also thanks Ms. Karima Mohamed and Ms. Anwar Al-Neaimi for their help formatting this study.
REFERENCES
- Alfred, D., S. Mohsin and S. Fuqua, 2001. Histological and biological evolution of human premalignant breast disease. Endocr-Relat Cancer, 8: 47-61.
CrossRefPubMedDirect Link - Amin, A. and M. Buratovich, 2007. The anti-cancer charm of flavonoids: A cup-of-tea will do. Recent Patents Anti-Cancer Drug Discov., 2: 109-117.
CrossRefPubMedDirect Link - Amin, A., A. Alkaabi, S. Al-Falasi and S.A. Daoud, 2005. Chemopreventive activities of Trigonella foenum graecum (Fenugreek) against breast cancer. Cell Biol. Int., 29: 687-694.
CrossRefPubMedDirect Link - Baccouche, S., J. Daoud, M. Frikha, R. Mokdad-Gargouri, A. Gargouri and R. Jlidi, 2003. Immunohistochemical status of P53, MDM2, bcl-2, bax and ER in invasive ductal breast cacinoma in Tunisian patients. Ann. N. Y. Acad. Sci., 1010: 752-763.
CrossRefPubMedDirect Link - Barone, M., R. Ladisa, A. Di Leo, D. Spano, D. Francioso, V. Aglio, A. Amoruso, A. Francavilla and A. Iolascon, 2006. Estrogen-induced proliferation in cultured hepatocytes involves cyclin D1, p21(Cip1) and p27(Kip1). Dig. Dis. Sci., 51: 580-586.
CrossRefPubMedDirect Link - Barone, M., E. Maiorano, R. Ladisa, R. Cuomo, A. Pece, P. Berloco, M.L. Caruso, A.M. Valentini, A. Iolascon, A. Francavilla, A. Di Leo and E. Ierardi, 2003. Influence of ursodeoxycholate-enriched diet on liver tumor growth in HBV transgenic mice. Hepatology, 37: 880-886.
CrossRefPubMedDirect Link - Bastin-Coyette, L., C. Smal, S. Cardoen, P. Saussoy, E. Van Den Neste and F. Bontemps, 2008. Mechanisms of cell death induced by 2-chloroadenosine in leukemic B-cells. Biochem. Pharmacol., 75: 1451-1460.
CrossRefPubMedDirect Link - Benz, C.C., 2008. Impact of aging on the biology of breast cancer. Crit. Rev. Oncol. Hematol., 66: 65-74.
CrossRefPubMedDirect Link - Bernardes, J., M. Seixas, G. Lima, L. Marinho and L. Gebrim, 2003. The effect of tamoxifen on PCNA expression In reply to: Fibroadenomas. Breast J., 9: 302-306.
CrossRefPubMedDirect Link - Borek, C., 2004. Dietary antioxidants and human cancer. Integr. Cancer Ther., 3: 333-341.
CrossRefPubMedDirect Link - Bray, F., P. McCarron and D.M. Parkin, 2004. The changing global patterns of female breast cancer incidence and mortality. Breast Cancer Res., 6: 229-239.
CrossRefPubMedDirect Link - Costa, I., M. Solanas and E. Escrich, 2002. Histopathologic characterization of mammary neoplastic lesions induced with 7,12 dimethylbenz(α)anthracene in the rat: A comparative analysis with human breast tumor. Arch. Pathol. Lab. Med., 126: 915-927.
PubMedDirect Link - Cox, R., 1998. The multi-step nature of carcinogenesis and the implications for risk analysis. Int. J. Radiat. Biol., 73: 373-376.
CrossRefPubMedDirect Link - Dechering, K., C. Boersma, S. Mosselman, 2000. Estrogen receptors alpha and beta: Two receptors of a kind? Curr. Med. Chem., 7: 561-576.
CrossRefPubMedDirect Link - Di Leo, A., Barone, E. Maiorano, S. Tanzi, D. Piscitelli, S. Marangi, K. Lofano, E. Ierardi, M. Principi and A. Francavilla, 2008. ER-ß expression in large bowel adenomas: Implications in colon carcinogenesis. Dig Liver Dis., 40: 260-266.
CrossRefPubMedDirect Link - Fisher, B., N. Gunduz, E.A. Saffer and S. Zheng, 1984. Relation of estrogen and its receptor to rat liver growth and regeneration. Cancer Res., 44: 2410-2415.
PubMedDirect Link - Francavilla, A., L. Polimeno, A. Dileo, M. Barone, P. Ove, M. Coetzee, P. Eagon, L. Makowka, G. Ambrosino, V. Mazzaferro and T.E. Starzl, 1989. The effect of estrogen and tamoxifen on hepatocyte proliferation in vivo and in vitro. Hepatology, 9: 614-620.
CrossRefPubMedDirect Link - Franzen, B., S. Linder, A.A. Alaiya, E. Eriksson, K. Fujioka, A.C. Bergman, H. Jornvall and Auer, 1997. Analysis of polypeptide expression in benign and malignant human breast lesions. Electrophoresis, 18: 582-587.
CrossRefPubMedDirect Link - Funakoshi, Y., H. Nakayama, K. Uetsuka, R. Nishimura, N. Sasaki and K. Doi, 2000. Cellular proliferative and telomerase activity in canine mammary gland tumours. Vet. Pathol., 37: 177-183.
PubMedDirect Link - Gosslau, A., S. Pabbaraja, S. Knapp and K.Y. Chen, 2008. Trans- and cis-stilbene polyphenols induced rapid perinuclear mitochondrial clustering and p53-independent apoptosis in Eur. J. Pharmacol., 587: 25-34.
CrossRefPubMedDirect Link - Gosslau, A., M. Chen, C.T. Ho and K.Y. Chen, 2005. A methoxy derivative of resveratrol analogue selectively induced activation of the mitochondrial apoptotic pathway in transformed fibroblasts. Br. J. Cancer, 92: 513-521.
CrossRefPubMedDirect Link - Gosslau, A. and K. Yu-Chen, 2004. Nutraceuticals, apoptosis and disease prevention. Nutrition, 20: 95-102.
CrossRefDirect Link - Gustafsson, J.A., 1999. Estrogen receptor beta: A new dimension in estrogen mechanism of action. J. Endocrinol., 163: 379-383.
CrossRefPubMedDirect Link - Guzman, S., A. Gato and J.M. Calleja, 2001. Antiinflammatory, analgesic and free radical scavenging activities of the marine microalgae Chlorella stigmatophora and Phaeodactylum tricornutum. Phytother. Res., 15: 224-230.
CrossRefPubMedDirect Link - Hanahan, D. and R.A. Weinberg, 2000. The hallmarks of cancer. Cell, 100: 57-70.
CrossRefPubMedDirect Link - Hasegawa, T., S. Kumamoto, K. Nomoto and Y. Yoshikai, 2005. Host defensive and pharmacological study of chlorella vulgaris strain CK. Stud. Natl. Prod. Chem., 30: 761-795.
CrossRef - Hasegawa, T., K. Noda, S. Kumamoto, Y. Ando, A. Yamada and Y. Yoshikai, 2000. Chlorella vulgaris culture supernatant (CVS) reduces psychological stress-induced apoptosis in thymocytes of mice. Int. J. Immunopharmacol., 22: 877-885.
CrossRefPubMedDirect Link - Hersey, P. and X.Z. Zhang, 2003. Overcoming resistance of cancer cells to apoptosis. J. Cell. Physiol., 196: 9-18.
CrossRefPubMedDirect Link - Jackson, S.J.T.and K.W. Singletary, 2004. Sulforaphane: A naturally occurring mammary carcinoma mitotic inhibitor, which disrupts tubulin polymerization. Carcinogenesis, 25: 219-227.
CrossRefPubMedDirect Link - Jemal, A., T. Murray, E. Ward, A. Samuels and R.C. Tiwari et al., 2005. Cancer Statistics, 2005. CA. Cancer J. Clin., 55: 10-30.
CrossRefPubMedDirect Link - Johnstone, R.W., A.A. Ruefli and S.W. Lowe, 2002. Apoptosis: A link between cancer genetics and chemotherapy. Cell, 108: 153-164.
CrossRefPubMedDirect Link - Kaufmann, S.H. and D.L. Vaux, 2003. Alterations in the apoptotic machinery and their potential role in anticancer drug resistance. Oncogene, 22: 7414-7430.
CrossRefPubMedDirect Link - Kumaraguruparan, R., D. Prathiba and S. Nagini, 2006. Of humans and canines: Immunohistochemical analysis of PCNA, Bcl-2, p53, cytokeratin and ER in mammary tumours. Res. Vet. Sci., 81: 218-224.
CrossRefPubMedDirect Link - Le Marchand, L., 2002. Cancer preventive effects of flavonoids-a review. Biomed. Pharmacother., 56: 296-301.
CrossRefPubMedDirect Link - Maga, G. and U. Hubscher, 2003. Proliferating cell nuclear antigen (PCNA): A dancer with many partners. J. Cell Sci., 116: 3051-3060.
PubMedDirect Link - Malkas, L.H., B.S. Herbert, W. Abdel-Aziz, L.E. Dobrolecki, Y. Liu and B. Agarwal et al,, 2006. A cancer-associated PCNA expressed in breast cancer has implications as a potential biomarker. Proc. Natl. Acad. Sci. USA., 103: 19472-19477.
CrossRefPubMedDirect Link - Mallick, N. and L.C. Rai, 1999. Response of the antioxidant systems of the nitrogen fixing cyanobacterium Anabaena doliolum to copper. J. Plant Physiol., 155: 146-149.
CrossRefDirect Link - Messa, C., F. Russo, M. Pricci and A. Di Leo, 2000. Epidermal growth factor and 17β-estradiol effects on proliferation of a human gastric cancer cell line (AGS). Scand. J. Gastroenterol., 35: 753-758.
CrossRefPubMedDirect Link - Mihara, M., S. Erster, A. Zaika, O. Petrenko, T. Chittenden, P. Pancoska and U.M. Moll, 2003. P53 has a direct apoptogenic role at the mitochondria. Mol. Cell, 11: 577-590.
CrossRefPubMedDirect Link - De Las Mulas, J.M., Y. Millan and R. Dios, 2005. A prospective analysis of immunohistochemically determined estrogen receptor {alpha} and progesterone receptor expression and host and tumour factors as predictors of disease-free period in mammary tumours of the dog. Vet. Pathol., 42: 200-212.
PubMedDirect Link - Murphy, L.C.and P. Watson, 2002. Steroid receptors in human breast carcinogenesis and breast cancer progression. Biomed. Pharmacother., 56: 65-77.
CrossRefPubMedDirect Link - Nandi, S., R.C. Guzman and J. Yang, 1995. Hormones and mammary carcinogenesis in mice, rats and humans: A unifying hypothesis. Proc. Natl. Acad. Sci. USA., 92: 3650-3657.
PubMedDirect Link - Park, J.Y., H.Y. Cho, J.K. Kim, K.H. Noh and J.R. Yang et al., 2005. Chlorella dichloromethane extract ameliorates no production and inos expression through the down-regulation of NFкB activity mediated by suppressed oxidative stress in raw 264.7 macrophages. Clin. Chem. Acta, 351: 185-196.
CrossRefPubMedDirect Link - Paunesku, T., S. Mittal, M. Protic, J. Oryhon, S.V. Korolev, A. Joachimiak and G.E. Woloschak, 2001. Proliferating cell nuclear antigen (PCNA): Ringmaster of the genome. Int. J. Radiat. Biol., 77: 1007-1021.
CrossRefPubMedDirect Link - Pearce, S.T. and V.C. Jordan, 2004. The biological role of estrogen receptors alpha and beta in cancer. Crit. Rev. Oncol. Hematol., 50: 3-22.
CrossRefPubMedDirect Link - Pozo-Guisado, E., A. Alvarez-Barrientos, S. Mulero-Navarro, B. Santiago-Josefat and P.M. Fernandez-Salguero, 2002. The antiproliferative activity of resveratrol results in apoptosis in MCF-7 but not in MDA-MB-231 human breast cancer cells: Cell-specific alteration of the cell cycle. Biochem. Pharmacol., 64: 1375-1386.
CrossRefPubMedDirect Link - Ray, A., K. Nkhata, J. Grande and M.P. Cleary, 2007. Diet-induced obesity and mammary tumor development in relation to estrogen receptor status. Cancer Lett., 253: 291-300.
CrossRefPubMedDirect Link - Russo, J., B.A. Gusterson, A.E. Rogers, I.H. Russo, S.R. Wellings and M.J. van Zwieten, 1990. Comparative study of human and rat mammary tumorigenesis. Lab. Invest., 62: 244-278.
PubMed - Samy, R.P., P. Gopalakrishnakone and S. Ignacimuthu, 2006. Anti-tumor promoting potential of luteolin against 7,12-dimethylbenz(a)anthracene-induced mammary tumors in rats. Chem. Biol. Interact., 164: 1-14.
CrossRefPubMedDirect Link - Shaw, J.A., K. Udokang, J.M. Mosquera, H. Chauhan, J.L. Jones and R.A. Walker, 2002. Oestrogen receptors alpha and beta in normal human breast and breast carcinomas. Pathology, 198: 450-457.
CrossRefPubMedDirect Link - Sun, Y.W., C.R. Herzog, J. Krzeminski, S. Amin, G. Perdew and K. El-Bayoumy, 2007. Effects of the environmental mammary carcinogen 6-nitrochrysene on p53 and p21Cip1 protein expression and cell cycle regulation in MCF-7 and MCF-10A cells. Chem. Biol. Interact., 170: 31-39.
CrossRefPubMedDirect Link - Takekoshi, H., G. Suzuki, H. Chubachi and M. Nakano, 2005. Effect of Chlorella pyrenoidosa on fecal excretion and liver accumulation of polychlorinated dibenzo-p-dioxin in mice. Chemosphere, 59: 297-304.
CrossRefPubMedDirect Link - Weaver, C.V. and S.P. Liu, 2008. Differentially expressed pro- and anti-apoptogenic genes in response to benzene exposure: Immunohistochemical localization of p53, Bag, Bad, Bax, Bcl-2 and Bcl-w in lung epithelia, Exp. Toxicol. Pathol., 59: 265-272.
CrossRefPubMedDirect Link - Weaver, C.V., S.P. Liu, J.S. Lu and B. Lin, 2007. The effects of benzene exposure on apoptosis in epithelial lung cells: localization by terminal deoxynucleotidyl transferase-mediated dUTP-biotin nick end labeling (TUNEL) and the immunocytochemical localization of apoptosis-related gene products. Cell Biol. Toxicol., 23: 201-220.
CrossRefPubMedDirect Link - Welsch, C.W., 1985. Host factors affecting the growth of carcinogen induced rat mammary carcinomas: A review and tribute to Charles Brenton Huggins. Cancer Res., 45: 3415-3443.
PubMedDirect Link - Wesierska-Gadek, J., M.P. Kramer and M. Maurer, 2008. Resveratrol modulates roscovitine-mediated cell cycle arrest of human MCF-7 breast cancer cells. Food Chem. Toxicol., 46: 1327-1327.
CrossRefPubMedDirect Link - Wu, L.C., J.A. Ho, M.C. Sheih and I.W. Lu, 2005. Antioxidant and antiproliferative activities of Spirulina and Chlorella water extracts. J. Agric. Food Chem., 53: 4207-4212.
PubMedDirect Link - Yager, J.D. and J.G. Liehr, 1996. Molecular mechanisms of estrogen carcinogenesis. Annu. Rev. Pharmacol. Toxicol., 36: 203-232.
CrossRefPubMedDirect Link - Cha, K.H., S.Y. Koo and D.U. Lee, 2008. Antiproliferative effects of carotenoids extracted from Chlorella ellipsoidea and Chlorella vulgaris on human colon cancer cells. J. Agric. Food Chem., 56: 10521-10526.
CrossRefPubMedDirect Link - Hasegawa, T., K. Ito, S. Ueno, S. Kumamoto, Y. Ando, A. Yamada, K. Nomoto and Y. Yasunobu, 1999. Oral administration of hot water extracts of Chlorella vulgaris reduces IgE production against milk casein in mice. Int. J. Immunopharmcol., 21: 311-323.
CrossRef









ashrafullah khan Reply
the paper is well written with good results