Research Article
Molecular Modelling Analysis of the Metabolism of Topotecan
School of Biomedical Sciences, Faculty of Health Sciences, C42, The University of Sydney, P.O. Box 170, Lid com be, NSW 1825, Australia
Topotecan (TOP, HycaminTM) is a water-soluble analogue of camptothecin, approved for use in second line therapy of patients with ovarian carcinoma and small-cell lung cancer (Arbuck and Takimoto, 1998; Herben et al., 2002). In addition, it has also been used to treat many forms of childhood cancer, including neuroblastoma, medulloblastoma, rhabodomyosarcoma and leukaemia (Furman et al., 2002; Galindo-Rodriguez et al., 2002). TOP inhibits DNA replication and RNA transcription by stabilizing the cleavable complexes formed between nuclear enzyme topoisomerase I and DNA (Rosing et al., 1999). There is an increased interest in topoisomerases as they are found to be targets for naturally occurring anticancer drugs (Tian et al., 2005). Topoisomerases are a group of enzymes, present in all organisms including bacteria, viruses, yeast and humans, that alter topology of DNA (Wang, 1996). There are two general types of topoisomerases: type I and type II. Type I cleaves and separates a single strand of DNA whereas type II cleaves both the DNA strands. Mammalian topoisomerase I is particularly important for supporting movement of replication fork during DNA replication and for relaxing supercoils formed during DNA transcription (Wang, 1996).
The lactone ring undergoes reversible hydrolysis that is found to be pH-dependent. The ring form (TOPR) is more stable at low pH whereas the open ring acid form (TOPO) is more stable at high pH. The drug needs to be in lactone ring form for the stabilization of topoisomerase I complex. TOP undergoes both renal and hepatic elimination (Bai et al., 2003). About 49% of an intravenously administered dose and 20% of an oral dose are excreted in the urine as parent drug. The primary hepatic metabolite of TOP is N-desmethyltopotecan. The primary metabolite of TOP is N-desmethyltopotecan (NDM-TOP). Like TOP, N-desmethyltopotecan also exists in the lactone ring form (NDM-TOPR) at low pH and in the open ring acid form (NDM-TOPO) at high pH.
In this study molecular modelling analyses have been carried out of TOPR, TOPO and the metabolites NDM-TOPR and NDM-TOPO, in order to obtain a better understanding of toxicity due to TPT and its metabolites. The study was carried out in the School of Biomedical Sciences, The University of Sydney during February to June 2006.
Computational Methods
The geometries of TOPR, TOPO and the metabolites NDM-TOPR and NDM-TOPO have been optimized based on molecular mechanics (Fig. 1), semi-empirical and DFT calculations, using the molecular modelling program Spartan ’04 (Spartan, 2004). Molecular mechanics calculations were carried out using MMFF force field. Semi-empirical calculations were carried out using the routine PM3. DFT calculations were carried out at B3LYP/6-31G* level. In optimization calculations, a RMS gradient of 0.001 was set as the terminating condition. For the optimized structures, single point calculations were carried to give heat of formation, enthalpy, entropy, free energy, dipole moment, solvation energy, energies for HOMO and LUMO. The order of calculations: molecular mechanics followed by semi-empirical followed by DFT ensured that the structure was not embedded in a local minimum.
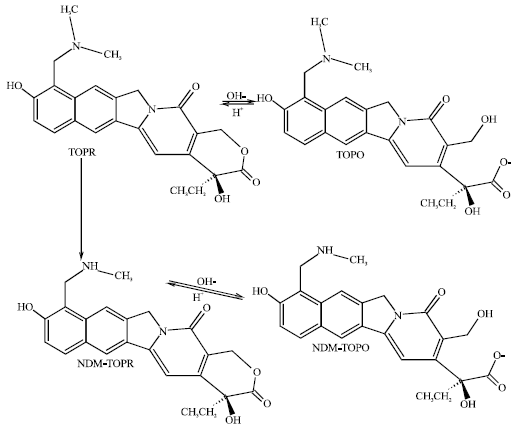 | |
| Fig. 1: | Metabolic pathways of toptotecan |
To further check whether the global minimum was reached, some calculations were carried out with improvable structures. It was found that when the stated order was followed, structure corresponding to the global minimum or close to that could ultimately be reached in all cases. Although RMS gradient of 0.001 may not be sufficiently low for vibrational analysis, it is believed to be sufficient for calculations associated with electronic energy levels.
Table 1 gives the total energy, heat of formation as per PM3 calculation, enthalpy, entropy, free energy, surface area, volume, dipole moment, energies of HOMO and LUMO as per both PM3 and DFT calculations for TOPR and TOPO and their metabolites NDM-TOPR and NDM-TOPO. Figure 2-5 give the regions of negative electrostatic potential (greyish-white envelopes) in (a) and density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) in (b) as applied to the optimized structures of TOPR, TOPO, NDM-TOPR and NDM-TOPO. The calculated solvation energies of TOPR, TOPO and their metabolites NDM-TOPR and NDM-TOPO from PM3 calculations in kcal mol-1 are respectively -18.21, -29.65, -20.38 and -36.68 and their dipole moments from DFT calculations are 8.8, 8.2, 8.9 and 8.7, respectively.
Large or moderately large solvation energies and dipole moments for TOPR, TOPO, NDM-TOPR and NDM-TOPO indicate that the compounds will be soluble in water so that they would have a high clearance rate.
The calculated LUMO-HOMO energy differences for TOPR, TOPO, NDM-TOPR and NDM-TOPO are found to be low indicating that the compounds would all be kinetically labile.
The molecular surface of TOPR, TOPO, NDM-TOPR and NDM-TOPO are found to abound in neutral (green), negative (yellow and red) regions, indicating the two compounds are more likely to subject to hydrophobic interaction and electrophilic attack.
| Table 1: | Calculated thermodynamic and other parameters of toptotecan and its metabolites |
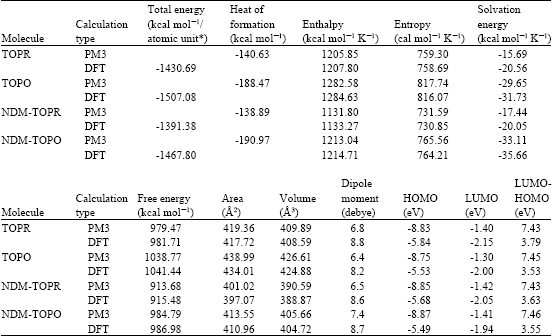 | |
| * in atomic units from DFT calculations | |
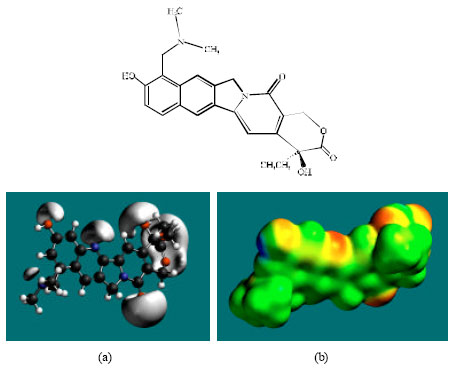 | |
| Fig. 2: | Structure of TOPR giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential) and in (b) surface electric charges (where red indicates negative, blue indicates positive and green indicates neutral) |
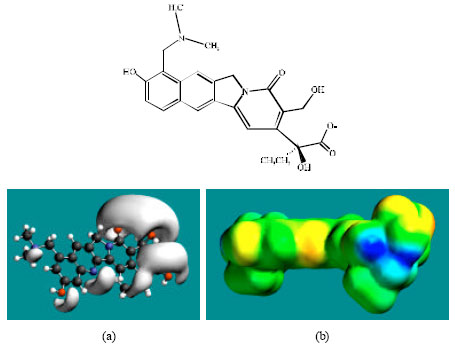 | |
| Fig. 3: | Structure of TOPO giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential) and in (b) surface electric charges (where red indicates negative, blue indicates positive and green indicates neutral) |
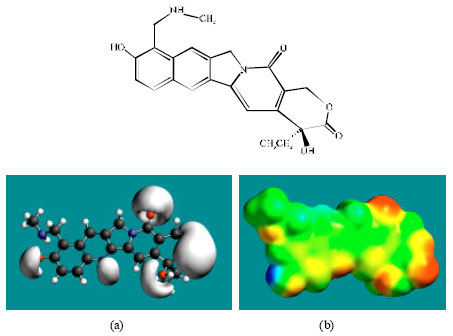 | |
| Fig. 4: | Structure of NDM-TOPR giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential) and in (b) surface electric charges (where red indicates negative, blue indicates positive and green indicates neutral) |
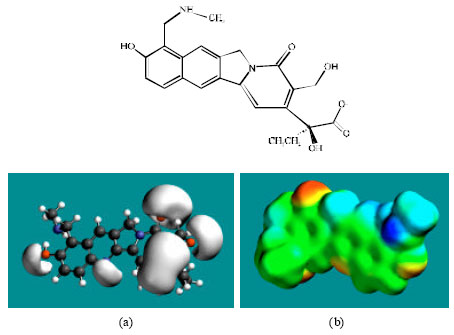 | |
| Fig. 5: | Structure of NDM-TOPO giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential) and in (b) surface electric charges (where red indicates negative, blue indicates positive and green indicates neutral) |
The presence of one or more positively charged lobes (blue) on the molecular surfaces of TOPO, NDM-TOPR and NDM-TOPO indicates that the compounds may also be subject to nucleophilic attack such as that by glutathione and nucleobases in DNA. Reaction with glutathione results into glutathione depletion that introduces oxidative stress which in turn induces cellular toxicity. Oxidation of nucleobases in DNA causes DNA damage.
When the surface area and volume of TOPR are compared with those of TOPO, NDM-TOPR and NDM-TOPO, it is found that the values for TOPR are distinctly different from those of other compounds, giving support to the idea that TOPO, NDM-TOPR and NDM-TOPO may not associate with topoisomerase I. It was noted earlier that toptotecan needs to be in lactone ring form for the stabilization of topoisomerase I complex that is involved in inhibiting DNA replication.
In the case of TOPR, TOPO, NDM-TOPR and NDM-TOPO, the electrostatic potential is found to be more negative around the various oxygen and nitrogen centers indicating that the positions may be subject to electrophilic attack.
Molecular modelling analyses based on semi-empirical and DFT calculations show that TOPR, TOPO, NDM-TOPR and NDM-TOPO all have small LUMO-HOMO energy differences so that they would be kinetically labile. The molecular surface of all the compounds abound in neutral and negative regions so they would readily undergo hydrophobic interaction and electrophilic attack, NDM-TOPR and NDM-TOPO also have some positively charged lobe so that they can be subject to some nucleophilic attack. This means that NDM-TOPR and NDM-TOPO can cause glutathione depletion and oxidation of nucleobases in DNA, thus inducing cellular toxicity and DNA damage.
Abbreviations
| TOP | : | Topotecan, |
| NDMTOP | : | N-desmethyltopotecan |
| DFT | : | Density functional theory |
| LUMO | : | Lowest unoccupied molecular orbital |
| HOMO | Highest occupied molecular orbital |
Fazlul Huq is grateful to the School of Biomedical Sciences, The University of Sydney for the time release from teaching.