ABSTRACT
Seeds of Phaseolus vulgaris cv. Limburgs vroege were sown in peat moss supplemented with ZnSO4 (600 mg kg–1) and irrigated with distilled water for 14 days. In the Zn-treated plants, significant reductions were shown in the capacities of glutathione reductase (GR) and ascorbate peroxidase (APOD) in the primary leaves as well as enhancement of the two main antioxidants in roots syringaldazine peroxidase (SPOD) and guaiacol peroxidase (GPOD). Zinc also caused reduced activities of superoxide dismutase (SOD), catalase (CAT) and isocitrate dehydrogenase (ICDH) nearly equally in leaves and roots. The Zn-induced increase in the content of individual amino acids (except cysteine) in primary leaves and roots was associated with a decrease in their total protein content. SDS-PAGE revealed de novo synthesis of possibly a phytochelatin synthase (MM 23.49 kDa) and two Zn-binding metallothionine-like proteins (MM 10.71 and 10.16 kDa) only in the root. These proteins might be responsible for the amelioration of the impact of Zn on the root. Another protein (MM 20.30 kDa) was induced in leaves which might be assumed to interfere with the reduction in enlargement of their cells.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ijb.2007.33.39
URL: https://scialert.net/abstract/?doi=ijb.2007.33.39
INTRODUCTION
Heavy metal stress is known to trigger a variety of defense mechanisms in plants including the biosynthesis of the metal-binding polypeptides phytochelatins [(γ-L-glutamyl-L-cysteinyl)n-glycine], where n ranges mostly between 2 and 5 but can be as high as 11 (Reddy and Prasad 1990). Phytochelatins (PCS) have molecular masses (MM) of 1.5-4 kDa and are synthesized directly from the tripeptide glutathione (GSH; γ-L-glutamyl-L-cyteinyl-glycine) through the activity of PC synthase with MM of approximately 25 kDa (Prasad, 1995; Cobbett et al., 1998). As the metal-binding metallothionine-like proteins (MM 7-18 kDa) contain a number of amino acids with glutamic acid, cysteine and glycine as the major constituents, they are thought to be aggregates of phytochelatins (Prasad, 1995). In maize, about 72 and 18% of Cd were bound to a low MM phytochelatin and a metallothionine-like (11.35 kDa) protein, respectively and these percentages were closely similar in Datura innoxia (Prasad, 1995).
Glutathione reductase (GR) is activated by heavy metal stress and catalyzes the reduction of glutathione disulphide (GSSG) in the chloroplasts into reduced glutathione (GSH) (Cuypers et al., 1999), deriving the required H2 from the strong reducing agent NADPH2. This reduced glutathione is a powerful scavenger of the Zn-induced reactive oxygen species (O2¯):
GSSG + NADPH2 → 2 GSH + NADP ………………… (GR) 2 GSH + O2¯ → GSSG + H2O
Another advantage of GR activity is the production of NADP in the processes of photosynthetic electron transport and ATP synthesis. Through the activity of isocitrate dehydrogense (ICDH), NADP may also be converted into the hydrogen donor NADPH2 needed for the production of GSH:
Isocitrate + NADP → α-ketoglutarate + CO2+NADPH2 ……. (ICDH)
Dehydroascorbate (DHA) can be reduced by GSH into ascorbate (AsA) through the activity of dehydroascorbate reductase (DHAR), thus providing a link between the glutathione and ascorbate pathways which play crucial roles in photosynthesis (Noctor and Foyer, 1998). Ascorbate peroxidase (APOD) catalyzes the oxidation of AsA in the chloroplasts into DHA, deriving the required O2 from the Zn-induced production of H2O2:
DHA + GSH → AsA + GSSG ……………………. (DHAR)
AsA + H2O2 → DHA + 2 H2O …………………… (APOD)
Zn-induced formation of H2O2 is involved in the β-oxidation of fatty acids of membrane lipids (Clijsters et al., 1999; Cuypers et al., 2001; Candan and Tarhan, 2003). The two peroxidases guaiacol peroxidase (GPOD) and syringaldazine peroxidase (SPOD) perform similar functions in protecting cells against the oxidative damage caused by H2O2:
Guaiacol (reduced) + H2O2 → guaiacol (oxidized) + 2 H2O …….. (GPOD)
Syringaldazine (reduced) + H2O2 → syringaldazine (oxidized) + 2 H2O ……………………….. (SPOD)
The two substrates guaiacol and syringaldazine are essential ingredients for lignin biosynthesis (Schreiber et al., 1999; Cuypers et al., 2002).
In a previous research (Kasim, 2006), the shoots and roots of 14-day-old seedlings of Phaseolus vulgaris L. cv. Limburgs vroege subjected to 600 mg ZnSO4 kg–1 peat moss soil responded in strikingly different ways: while growth parameters of the shoot were drastically reduced, those of the root remained unchanged. These reductions were associated with significant decreases in cell enlargement, stomatal frequency, substomatal CO2 concentration, photosynthetic pigments and disorganization of grana and thylakoids, which led to a severe reduction in the photosynthetic rate. Thus, the present study is an attempt to explore a possible relationship between these Zn-induced structural, ultra-structural and photosynthetic changes in Phaseolus vulgaris seedlings (Kasim, 2006) and the consequences of the outlined changes in their enzymes, amino acid pool and protein pattern.
MATERIALS AND METHODS
Seeds of Phaseolus vulgaris cv. Limburgs vroege were sown in peat moss either with or without a supplement of ZnSO4 (600 mg kg–1), irrigated with distilled water and kept for 14 days at 22°C ± 2, 62-65% relative humidity and daily illumination for 16 h with white fluorescent lamps at a photon flux density of 160 μmol m–2 s–1.
Enzyme capacities (potential activity measured in vitro under non-limiting reaction conditions) were assayed spectrophotometrically at 25°C in primary leaves and roots of three replicate seedlings and expressed as mU g–1 fresh weight since Zn did not affect the tissue water content under the experimental conditions (Cuypers et al., 2002). The enzymes assayed, wave lengths used for their capacity measurement and the used methods are shown in the following Table.
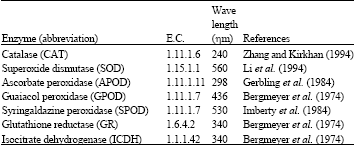 |
The contents of 16 amino acids (alanine, argenine, aspartic acid, cysteine, glutamic acid, glycine, histidine, isoleucine, leucine, lysine methionine, phenylalanine, proline, serine, threonine and valine) were measured in the extracts of primary leaves and roots of three replicate seedlings according to the method of Anderson et al. (1977) using HPLC (Beckman, System Gold, Programmable Solvent Module 126). Total protein contents of the same extracts were determined according to Bradford (1976).
SDS-PAGE of total protein of primary leaves and roots was performed using the method of Laemmli (1971) as modified by Studier (1973). The resulting gel was scanned and molecular masses (MM) of bands were determined using the program Gel-Pro Analyzer v. 3.1 for Windows.
Data based on replicates were subjected to one-way ANOVA analysis to estimate the least significant differences (LSD) at p ≤ 0.01 using the SPSS v. 12 package for Windows.
RESULTS
Enzymes: ZnSO4 treatment caused significant reductions in the capacities of CAT and ICDH but increased that of SPOD in the primary leaves and roots as compared with their respective control values; reductions in SOD capacities were non-significant (Fig. 1). The capacities of GR, APOD and GPOD were reduced with ZnSO4 in the primary leaves but increased in the root.
In the control and Zn-treated primary leaves, the capacities of APOD and GR were relatively much higher than corresponding values in roots, while the opposite was the case with GPOD and SPOD (Table 1). Zinc nearly halved the leaf/root capacity ratio of APOD and GR, but almost doubled the root/leaf capacity ratio of GPOD; the latter ratio of SPOD remained almost unaffected by Zn. The capacities of ICDH, CAT and SOD were nearly equal in the Zn-treated and untreated primary leaves and roots.
Amino acids and total protein: Total protein content of primary leaves and roots was significantly reduced by ZnSO4 (Fig. 1). Except for cysteine, the content of individual amino acids in the primary leaves was consistently higher than in the roots of the Zn-treated and untreated seedlings (Fig. 2).
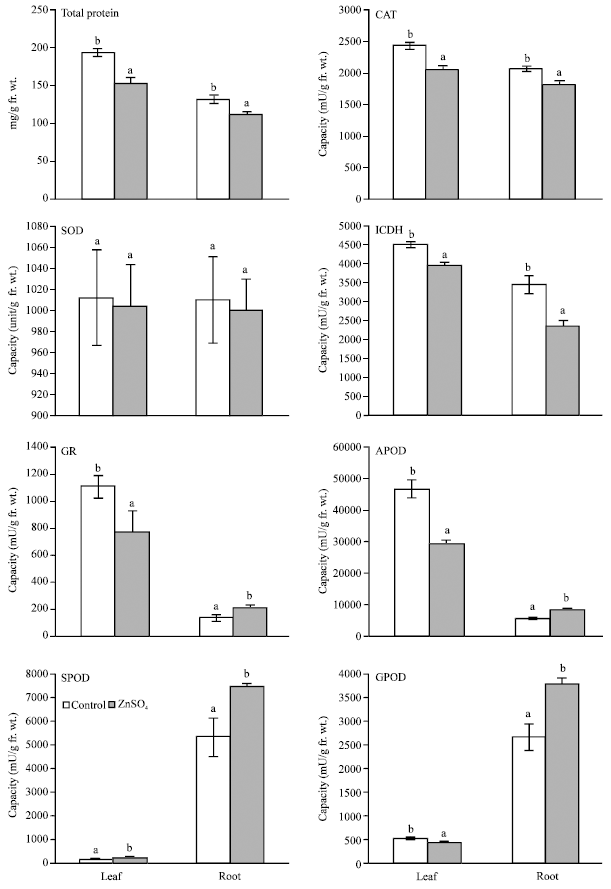 | |
| Fig. 1: | Effect of ZnSO4 (600 mg kg–1 peat moss soil) on total protein content and capacity of catalase (CAT), superoxide dismutase (SOD), isocitrate dehydrogenase (ICDH), glutathione reductase (GR), ascorbate peroxidase (APOD), syringaldazine peroxidase (SPOD) and guaiacol peroxidase (GPOD) in primary leaves and roots of 14-day-old Phaseolus vulgaris seedlings. Value of each bar represents mean±SD. Similar letter(s) on tops of corresponding bars indicate statistically non-significant differences at p≤0.01, based on F-test and LSD (n = 3) |
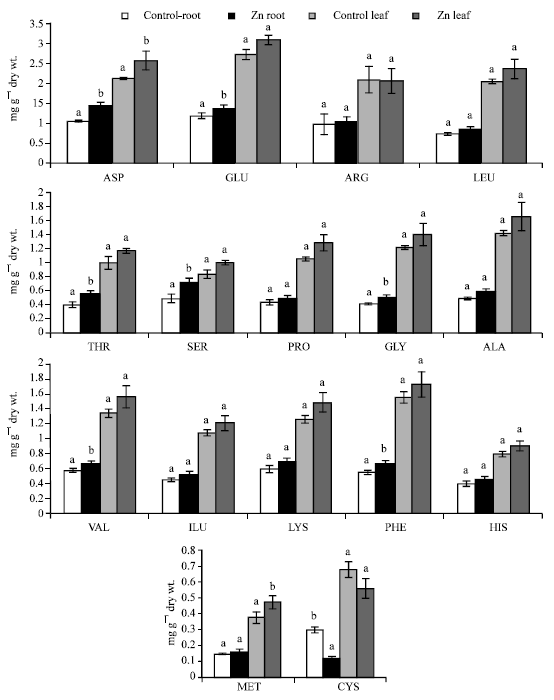 | |
| Fig. 2: | Effect of ZnSO4 (600 mg kg–1 peat moss soil) on the contents of 16 amino acids in primary leaves and roots of 14-day-old Phaseolus vulgaris seedlings. Value of each bar represents mean±SD. Similar letters on tops of corresponding bars indicate statistically non-significant differences at p≤0.05, based on F-test and LSD (n = 3) |
Zinc tended to enhance the accumulation of 15 individual amino acids in primary leaves and roots, but caused significant depletion of cysteine in the two organs.
Protein pattern: Only the protein bands with different occurrence in the control and Zn-treated primary leaves and roots are presented in Table 2; the 14 bands consistently found in both organs (MM 242.37-226.4, 136.12-120.43, 45.69, 41.29-38.81, 33.65, 32.77-32.12, 31.11, 28.08, 26.05, 17.55, 15.69, 14.77, 13.77, 8.51 kDa) were omitted.
Leaves had a much wider variety of protein bands than roots (Table 2). Each of the two organs possessed its distinctive set of proteins. Bands with MM 169.18, 140.18, 103.45, 43.51, 25.11, 22.75, 12.94, 12.64 and 9.41-5.63 kDa were uniquely found in primary leaves. Four of these bands (MM 169.18, 140.18, 103.45 and 22.75 kDa) disappeared in the Zn-treated leaves, while the band with MM 20.29 kDa was the only protein induced in response to Zn.
| Table 1: | Capacity ratios of seven enzymes in primary leaves and roots of 14-day-old Phaseolus vulgaris seedlings treated with 600 mg ZnSO4 kg–1 peat moss soil |
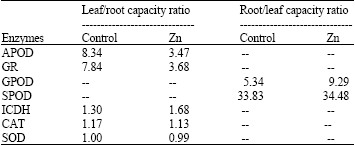 | |
| Ratios are obtained from the mean values shown in Fig. 1 | |
| Table 2: | Occurrence of protein bands and their molecular masses (MM) as revealed by SDS-PAGE of total protein in primary leaves and roots of 14-day old Phaseolus vulgaris seedlings treated with 600 mg ZnSO4 kg–1 peat moss soil |
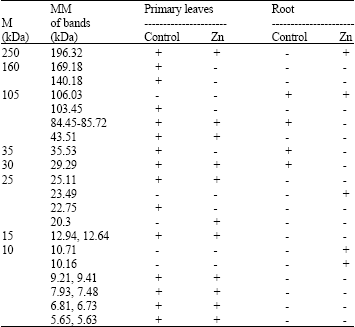 | |
| M = Marker, + = Present; - = Absent | |
Similarly, the band with MM 106.03 was found exclusively in Zn-treated and untreated roots, while those with MM 23.49, 10.71 and 10.16 kDa were induced only in response to the Zn treatment. Zn caused the repression of two protein bands (MM 84.45-85.72 and 29.29 kDa) in roots and protein band (MM 35.53 kDa) in primary leaves and roots.
DISCUSSION
In a previous work (Kasim, 2006), it was observed that shoots of Phaseolus vulgaris cv. Limburgs vroege seedlings (14-day-old) were sensitive to Zn-stress, whereas roots were not significantly affected. In the present work, shoots and roots of these seedlings seemed to have different enzymatic defense mechanisms against Zn stress. In this respect, significant reductions were shown in the capacities of the two main defensive enzymes GR and APOD, which act as scavengers of reactive oxygen species in the primary leaves (Fig. 1). This might allow Zn-induced peroxidation of membrane lipids and consequently rupture of chloroplast membranes and disorganization of thylakoids as shown previously. The Zn-induced reduction in GPOD capacity in leaves not only shared in the peroxidation of membrane lipids but might have also contributed to the reduction in lignin biosynthesis (Schreiber et al., 1999; Cuypers et al., 2002). On the other hand, Zn treatment induced enhancement of two main antioxidant peroxidases of the root SPOD and GPOD. These antioxidant enzymes might keep maintenance of the membrane integrity and thus interpret the root tolerance to Zn stress.
On the other hand, the activities of SOD, CAT and ICDH were reduced by Zn stress in both leaves and roots at a more or less similar magnitude. Therefore, these enzymes might be assumed to play only a marginal role in the differential resistance of shoots and roots of the seedlings to Zn stress. Suppressed activities of SOD and CAT (as O2¯ and H2O2 scavengers, respectively; Shah et al., 2001) by Zn treatment were also recorded in Mentha pulegium (Candan and Tarhan, 2003).
In addition, differences in the behaviour of GR, APOD, GPOD and SPOD in shoots and roots of P. vulgaris, in response to Zn stress, were also recorded by Cuypers et al. (2001, 2002). However, discrepancies between their findings and results of the present study might be attributed to basic differences in experimental conditions especially Zn concentration, duration of exposure to Zn stress, seedling age (10 vs. 14 days) and the growth medium (hydroponics vs. peat moss). Clearly, the response of P. vulgaris seedlings to Zn stress is dependent upon the metal dose, exposure time as well as the developmental stage of the plant as indicated by Van Assche and Clijsters (1990) and Cuypers et al. (1999).
In the present research, the Zn-induced increase in amino acid content of primary leaves and roots was associated with a decrease in total protein, thus indicating that Zn retarded the assembly of amino acids into proteins in roots and in above-ground organs of P. vulgaris seedlings.
The above mentioned results were concomitant with de novo synthesis of three proteins having molecular masses (MM) 23.49, 10.71 and 10.16 kDa only in roots of the Zn-treated seedlings (Table 2). None of these proteins could be matched with similar proteins in the data base of P. vulgaris (http://www.ebi.uniprot.org). However, according to Prasad (1995), the protein having MM 23.49 kDa might be assumed to represent a phytochelatin (PC) synthase, whereas, those having MM 10.71 and 10.16 kDa might represent Zn-binding metallothionine-like proteins. In this respect, PC (derived from glutathione) is generally responsible for the binding of heavy metals in plants in both normal constitutive tolerance and adaptive hypertolerance (Schat et al., 2002). It is therefore tempting to suggest that these proteins might be the phytochelatin synthase (MM 23.49 kDa) and the two Zn-binding metallothionine-like proteins (MM 10.71 and 10.16 kDa) responsible for the clear amelioration of the impact of Zn on the growth criteria of root, despite the greater accumulation of Zn recorded by Kasim (2006) in root than in shoot.
In contrast, a 20.30 kDa protein was the only one induced in the primary leaves of P. vulgaris seedlings in response to Zn treatment (Table 2). This protein might be tentatively assumed to resemble the 19.96 and 20 kDa proteins shown by Keller et al. (1988) and Agrawal and Sharma (2006), respectively as precursors of the glycine-rich structural protein responsible for the stiffening of cell walls. Thus, on these bases, the protein with MM 20.30 kDa induced in the present work in the primary leaves, in response to Zn treatment, might participate in the obvious reduction of cell enlargement in both parenchymatous and xylem vessels of leaves (Kasim, 2006).
Integration of the results of the present study and those obtained previously (Kasim, 2006) seems to suggest that Zn accumulation in intercellular spaces and impregnation of middle lamellae between cells of the shoot obstructed internal gas diffusion (short distance transport). This would result in metabolic disturbances in the shoot which might include: (i) inhibition of antioxidant enzymes of the ascorbate-glutathione pathway (mainly GR and APOD), (ii) reduction in the capacity of other antioxidants enzymes (GPOD and SPOD) involved in lignin biosynthesis and (iii) the induction of a 20 kDa protein with a role in the stiffening of cell walls. These disturbances might lead to: (i) oxidative damage of cellular membranes, (ii) reduced lignin productivity and (iii) premature cessation of cell enlargement, respectively. The reduction of lignin productivity and cell enlargement would interpret the observed reductions in stem length and surface area of primary leaves caused by Zn stress.
It might be concluded that tolerance of P. vulgaris root to high Zn accumulation includes: (i) the synthesis of metallothionine-like proteins, (ii) the absence of cell wall stiffening proteins and (iii) the enhanced activity of certain peroxidases (GPOD and SPOD) with the dual role of protecting cell membranes through scavenging reactive oxygen species and participating in the development of lignified xylem elements.
ACKNOWLEDGMENTS
I wish to thank Prof. J. Vangronsveld and Dr. W. Geebelen (Hasselt University, Belgium) for supply of seeds and laboratory facilities for preliminary experiments on enzymes of ascorbate-glutathione pathway and Prof. Seham M. Mostafa (Ain Shams University, Cairo, Egypt) for critically reading the manuscript.
REFERENCES
- Agrawal, V. and K. Sharma, 2006. Phytotoxic effects of Cu, Zn, Cd and Pb on in vitro regeneration and concomitant protein changes in Holarrhena antidysenterica. Biol. Plant., 50: 307-310.
Direct Link - Bradford, M.M., 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem., 72: 248-254.
CrossRefPubMedDirect Link - Candan, N. and L. Tarhan, 2003. The correlation between antioxidant enzyme activities and lipid peroxidation levels in Mentha pulegium organs grown in Ca2+, Mg2+, Cu2+, Zn2+ and Mn2+ stress conditions. Plant Sci., 163: 769-779.
CrossRefDirect Link - Clijsters, H., A. Cuypers and J. Vangronveld, 1999. Physiological responses to heavy metals in higher plants: Defense against oxidative stress. Z. Naturforsch., 54c: 730-734.
Direct Link - Cobbett, C.S., M.J. May, R. Howden and B. Rolls, 1998. The glutathione-deficient, cadmium-sensitive mutant, cad 2-1, of Arabidopsis thaliana is deficient in γ-glutamylcysteine synthase. Plant J., 16: 73-78.
Direct Link - Cuypers, A., J. Vangronsveld and H. Clijsters, 1999. The chemical behaviour of heavy metals plays a prominent role in the induction of oxidative stress. Free Radic. Res., 31: S39-S43.
CrossRefDirect Link - Cuypers, A., J. Vangronsveld and H. Clijsters, 2001. The redox status of plant cells (AsA and GSH) is sensitive to zinc imposed oxidative stress in roots and primary leaves of Phaseolus vulgaris. Plant Physiol. Biochem., 39: 657-664.
Direct Link - Cuypers, A., J. Vangronsveld and H. Clijsters, 2002. Peroxidases in roots and primary leaves of Phaseolus vulgaris. Copper and zinc phytotoxicity: A comparison. J. Plant Physiol., 159: 869-876.
Direct Link - Kasim, W.A., 2006. Physiological consequences of Zn-induced structural and ultra-structural changes in Phaseolus vulgaris I. Growth and the photosynthetic apparatus. Int. J. Bot., 3: 15-22.
CrossRefDirect Link - Laemmli, U.K., 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature, 227: 680-685.
CrossRefDirect Link - Noctor, G. and C.H. Foyer, 1998. Ascorbate and glutathione: Keeping active oxygen under control. Annu. Rev. Plant Physiol. Mol. Biol., 49: 249-279.
CrossRefPubMedDirect Link - Prasad, M.N.V., 1995. Cadmium toxicity and tolerance in vascular plants. Environ. Exp. Bot., 35: 525-545.
CrossRefDirect Link - Schat, H., M. Liugany, R. Vooijs, J. Hartley-Whitaker and P.M. Bleeker, 2002. The role of phytochelatins and adaptive heavy metal tolerances in hyperaccumulator and non-hyperaccumulator metallophytes. J. Exp. Bot., 53: 2381-2392.
Direct Link - Schreiber, L., K. Hartman, M. Skrabs and J. Zeier, 1999. Apoplastic barrier in roots: Chemical composition of endodermal and hypodermal cell walls. J. Exp. Bot., 50: 1267-1280.
Direct Link - Shah, K., R.G. Kumar, S. Verma and R.S. Dubey, 2001. Effect of cadmium on lipid peroxidation, superoxide anion generation and activities of antioxidant enzymes in growing rice seedlings. Plant Sci., 161: 1135-1144.
CrossRefDirect Link - Van Assche, F. and H. Clijsters, 1990. Effects of metals on enzyme activity in plants. Plant Cell Environ., 13: 195-206.
CrossRefDirect Link








