ABSTRACT
Stevia rebaudiana Bert. is an economically important medicinal plants act as a sugar substitute for diabetic and obese people. In the present investigation a novel protocol was developed to accelerate the production of embryogenic calli so that large number of in vitro plant regeneration can be feasible. Two different types of amino acids (Trypthphan and Glutamine) in different concentration and three different types of additives (yeast extract, casein hydrolysate and Potato extract) were utilized along with MS medium supplemented with 2,4-D (2.0 mg L-1), BAP (0.5 mg L-1), sucrose (30 g L-1) and agar with leaf as explant to elucidate their role in developing and maintaining embryogenic calli. It was found that glutamine (50 mg L-1) and casein hydrolysate (100 mg L-1) produced greenish, healthy nodular calli with more embryogenic potential and less necrotic lesion in comparison with only PGR supplemented basal medium which further performed better in regeneration medium for producing large number of regenerated shoots.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/biotech.2010.368.372
URL: https://scialert.net/abstract/?doi=biotech.2010.368.372
INTRODUCTION
Stevia (Stevia rebaudiana Bert., Asteraceae) a sweet herb of Paraguay is a non-caloric natural sugar alternative to artificially produced sugar substitutes. The stevia leaf is 30 times sweeter than sugar and sweetness is due to glycosides of which the most abundant is stevioside (Kinghorn, 1992). The sweet compounds pass through the digestive process without chemically breaking down, making stevia safe for diabetic and obese people (Yodyingyuad and Bunyawong, 1991). Stevia finds its use as a natural sweetener, replacing the chemical sweeteners and even table sugar (Kumar and Kaul, 2005). In stevia propagation through seed and cutting is problematic. So, in vitro propagation is the sound technique for large scale production of this plant because the species is becoming rare in natural habitat condition due to habitat destruction and over exploitation (Carneiro, 1990).
The mass propagation of plants through multiplication of embryogenic propagules is the most commercially attractive application of somatic embryogenesis (Jiminez and Bangerth, 2001). Various biotechnological applications in plant breeding, such as gene transformation, mutation and in vitro selection, rely on the availability of efficient plant tissue culture systems (Livingstone and Birch, 1999). Somatic embryos can be distinguished from adventitious shoots, because they are bipolar, having both a root and shoot pole and they do not have any vascular connections with the underlying parental tissue (Haccius, 1978). It can be initiated by two mechanisms directly on explanted tissues (clonation), where plants are genetically identical and indirectly from unorganized tissue (callus) (Williams and Maheswaran, 1986). Amino acids are important for growth regulation and as modulators of growth and cell differentiation, which may be affecting general metabolism and consequently morphogenesis (Basu et al., 1989). Supplementation of amino acids in culture media has been reported to enhance somatic embryogenesis in many plant species (Wang et al., 2002; Kopertekh and Stribnaya, 2003; Abdullah et al., 2005). Use of complex organic extract as supplements in basal media containing inorganic nutrients have been used by various workers to enhance the production of embryogenic callus (Ichihashi and Islam, 1999; Ogita et al., 2001; Vasudevan et al., 2004; Coruzzi and Last, 2000). The optimal composition of phytohormones and requirements of other complex organic extracts varies with plant species and massive investigations are required to determine these compositions. Developing methods of supporting the actions of amino acids and additives as a supplement of growth regulators will be useful in the establishment of a reliable operation to induce somatic embryos from various plants.
So, in this background the aim of this study presented here was to enhance the efficiency of somatic embryogenesis manipulated by addition of various organic extracts and amino acids in different concentration.
MATERIALS AND METHODS
Collection of explants and surface sterilization: To initiate the present work on the callus culture of stevia, young, actively growing leaves were collected from two months old stevia plants being aseptically maintained in the greenhouse of Bidhan Chandra Krishi Viswavidyalaya, Mohanpur, India from 2005 to 2009. The collected explants were surface sterilized by treating with mercuric chloride solution (0.1% w/v dissolved in 50% v/v alcohol) for 2 min, rinsed 5-6 times with sterile water. Excess water adhering to the explants was soaked on sterilized blotting paper.
Culture conditions: For callus induction and maintenance, explant was cultured in MS (Murashige and Skoog, 1962) basal medium with 30 g L-1 sucrose and after solidifying with 7 g L-1 agar. Different Plant Growth Regulators (PGRs) like N6-benzylamino purine (BAP), 2, 4-dichloro phenoxy acetic acid (2,4-D) were added at various concentrations to MS before the pH of the medium was adjusted to 5.6. Media were autoclaved at 1.06 kg cm-2 and 121°C for 20 min. Cultures at all growth stages were incubated under artificial conditions: 24±1°C, 60% RH, under diffuse light (500 lux) and a 16 h photoperiod under a photosynthetic photon flux density of 20 μmol/m2/sec.
In vitro propagation: For callus induction and maintenance leaf explants were inoculated in the media and incubated following the above mentioned artificial growth conditions, until greenish callus appeared. Two amino acids namely glutamine and tryptophan were used at various concentrations (50 and 100 mg L-1) and as growth additives required amounts of yeast extract, casein hydrolysate and potato extract were prepared freshly and added for supplementing the nutrient medium and also for exerting their effect on callus induction, maintenance and regeneration potential. In all experiments 20 tubes for each treatment combinations were inoculated and maintained up to 45th day.
Data analysis: Complete Randomized Design (CRD) was followed for the in vitro culture experiments. Each single explant was considered as an experimental unit. The experiments were carried out in six replications including 20 explants in each and standard deviation was calculated. Angular transformations of the percent response values were done before the collected data were subject to analysis of variance (ANOVA). Significant differences among the treatments were tested by Duncan’s multiple range test (Duncan, 1955) at 5% level using WINDOWSTAT 7.5 (Indostat services, Hyderabad, India) software package.
RESULTS AND DISCUSSION
Leaf explant was inoculated on the MS basal medium with 2, 4-D (2 mg L-1), BAP (0.5 mg L-1), sucrose (3%), agar (0.7%) and two different types of amino acids (Trypthphan and Glutamine). So far as induction of callus and their further growth and regeneration in question, best response was obtained on medium supplemented with glutamine at 50 mg L-1 (Table 1). Glutamine at this concentration induced 88.09% callus, with 35.77 and 102.24 mg callus weight on 20th and 45th day, respectively. Callus induction percentage by using glutamine at 50 mg L-1 as growth adjuvant was 1.43% increased than the medium with only optimum growth regulators combination. In this combination nodular calli with more embryogenic region was produced (Fig. 1). Surprisingly utilization of glutamine at 100 mg L-1 could not reveal any significant result. The effect of tryptophan at 50 and 100 mg L-1 were not satisfactory at all. Glutamine act as a source of nitrogen, increases explant responsiveness to callus induction medium and also indirectly increasing endogenous auxin and cytokinin levels during the induction period (Coruzzi and Last, 2000).
| Table 1: | Effect of amino acids on callus induction and growth with leaf as explant. MS: basal medium + 2, 4-D (2.0 mg L-1) + BAP (0.5 mg L-1) + amino acids + sucrose (3%) + agar (0.7%), pH 5.8 |
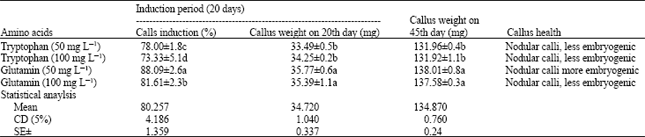 | |
| Data represent Mean± SD of 20 replicants per treatment in five repeated experiments. Different letter(s) indicate significant difference at p<0.05 (or means within columns separated by Duncan’s Multiple Range Test p = 0.05; Duncan, 1955) | |
| Table 2: | Effect of additives on callus induction with leaf as explant. MS: basal medium+ 2, 4-D (2.0 mg L-1) + BAP (0.5 mg L-1) + sucrose (3%) + agar (0.7%) + Glutamine (50 mg L-1) +additive, pH 5.8 |
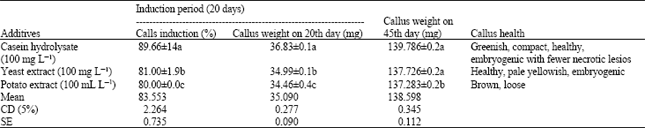 | |
| Data represent Mean±SD of 20 replicants per treatment in seven repeated experiments. Different letter(s) indicate significant difference at p<0.05 (or means within columns separated by Duncan’s Multiple Range Test p = 0.05; Duncan, 1955) | |
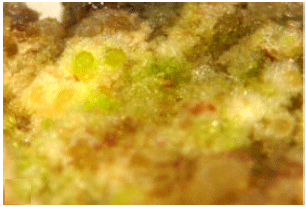 | |
| Fig. 1: | Nature of 45 days old leaf derived calli grown in MS medium supplemented with BAP (0.5 mg L-1), 2,4-D (2.0 mg L-1) and glutamine (50 mg L-1) |
Addition of glutamine which is relatively non toxic would enable the cells to maintain a high growth rate for a longer period (Gamborg and Miller, 1968). In the present investigation it was clearly established that glutamine played vital role in the induction and maintenance of lession and necrosis free embryogenic calli which can ultimately produce maximum number of regenerated shoots. Glutamine was metabolized extensively to glutamate in the supplied explant and recovery of L-glutamate as the main component suggested transport in this form. L-glutamine or other amino acids, which can be readily transferred to other amino acids and incorporated into proteins (Li et al., 1993; Higashi et al., 1996). On the other hand no promotive effect of tryptophan in the present study can be explained in this way because tryptophan acts as a precursor of IAA in plants and exogenous application has been demonstrated to increase IAA synthesis in plant tissue (Dunstan et al., 1979). But in the present study IAA did not have any satisfactory role in this contest so indirectly tryptophan has no promotive role in case of callus induction and maintenance of stevia.
 | |
| Fig. 2: | Nature of 45 days old leaf derived calli grown in MS medium supplemented with BAP (0.5 mg L-1), 2,4-D (2.0 mg L-1), glutamine (50 mg L-1) and casein hydrolysate (100 mg L-1) |
Responses of three growth additives indicated that casein hydrolysate (100 mg L-1) performed better than the other two growth additives (Table 2). Callus induction percentage in case of casein hydrolysate (100 mg L-1) was 89.66%, which was even greater than the medium supplemented with growth regulators and glutamine at 50 mg L-1. Callus growth was very good and on 20th day of induction period callus weight was 36.83 mg. Gain of callus weight on 45th day of maintenance period was 102.95 mg. Callus in this case was greenish, compact, healthy and embryogenic with fewer necrotic lesions (Fig. 2). On the other hand though yeast extracts (100 mg L-1) supplemented medium was able to produce embryogenic callus but in case of potato extract (100 ml L-1) supplementation callus was brown in color and loose in nature. Several early workers have indicated that casein hydrolysate contain carbohydrates, protein, fat, several vitamins, phenolic compounds and low levels of some amino acids and fatty acids. Any of these or other substances, yet unknown, single or in combination might be factor(s) that enhance(s) callus growth (Islam et al., 1997).
 | |
| Fig. 3: | Greenish, compact, healthy and embryogenic calli prior to regeneration |
 | |
| Fig. 4: | Regeneration of multiple shootlets from callus maintained in MS medium supplemented with BAP (0.5 mg L-1), 2,4-D (2.0 mg L-1), glutamine (50 mg L-1) and casein hydrolysate (100 mg L-1) |
According to Parabia et al. (2007) casein hydrolysate without growth regulators in nutrient medium reduces the time period of shoot initiation up to 3-4 days as well as new shoot proliferation from axillary buds and also increased the percentage of explant response.
So, from the studies it was revealed that the MS medium supplemented with 2,4-D (2.0 mg L-1), BAP (0.5 mg L-1), sucrose (30 g L-1), agar (0.7%), glutamine (50 mg L-1) and casein hydrolysate (100 mg L-1) produced the best result regarding callus induction and maintenance for regeneration in comparison with only growth regulator supplemented media (Fig. 3). This combination can only able to produce greenish, healthy nodular calli with more embryogenic potential and less necrotic lesion which further performed better in regeneration medium for producing large number of regenerated shoots (Fig. 4).
REFERENCES
- Abdullah, R., Z. Alizah, W.H. Wee, C.L. Leaw and C.B. Yeap et al., 2005. Immature embryo: A useful tool for oil palm (Elaies guineensis Jacq.) genetic transformation studies. Elect. J. Biotechnol., 8: 25-34.
Direct Link - Dunstan, D.I., K.C. Short, E. Dhaliwal and E. Thomas, 1979. Further studies on plantlet production from cultured tissue of Sorghum bicolor. Protoplasma, 101: 355-361.
CrossRef - Ichihashi, S. and M.O. Islam, 1999. Effects of complex organic additives on callus growth in three orchid genera, Phalaenopsis, Doritaenopsis and Neofinetia. J. Jap. Soc. Hort. Sci., 68: 269-274.
Direct Link - Jiminez, V.M. and F. Bangerth, 2001. Endogenous hormone levels in explants and in embryogenic and non embryogenic cultures of carrot. Physiol. Plant, 111: 389-395.
Direct Link - Kopertekh, L.G. and L.A. Stribnaya, 2003. Plant regeneration from wheat leaf explants. Russ. J. Plant Physiol., 50: 365-368.
CrossRef - Murashige, T. and F. Skoog, 1962. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant., 15: 473-497.
CrossRefDirect Link - Ogita, S., H. Sasamoto, E.C. Yenug and T.A. Thorpe, 2001. The effects of glutamine on the maintenance of embryogenic culture of Cryptomeria japonica. In vitro Cell. Dev. Plant, 37: 268-273.
CrossRef - Vasudevan, A., N. Selvaraj, A. Ganapathi, S. Kasthurirengan, V.R. Anbazhagan and M. Manickavasagam, 2004. Glutamine: A suitable nitrogen source for enhanced shoot multiplication in Cucumis sativus L. Biol. Plant., 48: 125-128.
CrossRefDirect Link - Wang, Y., B.A. Ruemmela, J.M. Chandlee, W.M. Sullivan, J.E. Knapp and A.P. Kausch, 2002. Embryogenic callus induction and plant regeneratio media for bentgrasses and annual blue grass. In Vitro Cell. Dev. Biol. Plant, 38: 460-467.
CrossRef - Williams, E.G. and G. Maheswaran, 1986. Somatic embryogenesis: Factors influencing coordinated behaviour of cells as an embryogenic group. Ann. Bot., 57: 443-462.
Direct Link - Yodyingyuad, V. and S. Bunyawong, 1991. Effects of stevioside on growth and reproduction. Hum. Reprod., 6: 158-165.
PubMed








