ABSTRACT
Objective: The aim of this study was to prepare and characterize activated carbon from local oak sawdust in order to relate the preparation conditions (carbonization and chemical activation) to the carbon’s essential physical, chemical and adsorptive properties. Methodology: Activated carbons were prepared from raw sawdust carbon (SDC) and by first activation with 3.07 N H3PO4 (PASC). The carbon yield for SDC was 33.8% while, phosphoric acid activated sawdust carbon (PASC) showed 29.4% of carbon yield. The pH values for the SDC and PASC solutions were found to be 7.4 and 6.0, respectively. The electrical conductivities for the SDC and PASC solutions were found to be 144 and 327 μS cm–1. Moisture contents for SDC and PASC were 5.4 and 4.6% and ash contents for SDC and PASC were 6.8 and 6.6% respectively. The surface concentration of the functional groups on SDC titrated by the equilibrium Boehm method is 1.508 mmol g–1, of which 1.062 mmol g–1 have acidic and 0.446 mmol g–1 have basic character. While, the surface concentration of the functional groups on PASC titrated by the equilibrium Boehm method is 2.615 mmol g–1, of which 2.467 mmol g–1 have acidic and 0.148 mmol g–1 have basic character. Results: The values of iodine number obtained for PASC (872.4 mg g–1) is greater than the value obtained for SDC (554.6 mg g–1). The methylene blue number obtained for PASC (306.0 mg g–1) is greater than the value obtained for SDC (180.3 mg g–1). The Langmuir maximum adsorption capacity of phenol on the resultant PASC and SDC was equal 99.0 and 72.4 mg g–1 respectively. Conclusion: The results show that agricultural waste oak sawdust can be used as a promising precursor material for the production of low cost activated carbon.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajsr.2016.45.56
URL: https://scialert.net/abstract/?doi=ajsr.2016.45.56
INTRODUCTION
Activated carbons are produced with a wide range of properties and physical forms, which leads to their use in numerous applications. Activated carbon also called as activated charcoal or activated coal, is a form of carbon that has been processed to make it extremely porous and thus to have a very large surface area available for adsorption or chemical reactions. Activated carbons are extremely versatile adsorbents of great industrial significance in the removal of gas and purification of chemicals1. They are also used as catalysts and catalyst supports in the catalytic processes2.
Most organic materials rich in carbon that do not fuse upon carbonization can be used as raw material for the manufacture of activated carbon. Raw materials together with the production method determine the final characteristics of the activated carbon product as well as the overall economic viability. Polyacrylonitrile is considered to be best precursor for making performance carbon fibers. Activated carbons can be prepared from large number of precursor materials with a high carbon content and low level ash including coal, woods and bones2. In addition, some agricultural by-products residues from agriculture and agro industries can be also used as raw materials for preparing activated carbons. These includes olive stones3, almond shells4, apricot and peach stones5, maize cob6, linseed straw7, sawdust8, rice hulls9, cashew nuts10, etc. Besides these other sources are, coconut shells, eucalyptus bark, linseed cake, tamarind seeds and tea waste ash suffocated coal, baggase, ground nut husk, activated bauxite, palm seed coat, de-oiled soya and cement kiln dust.
Activated carbon can be prepared either by physical (or dry) activation or chemical activation. Physical activation involves two steps; in first step precursor material will undergo carbonization process (400-900°C) in the absence of air and second step involves activation/oxidation by gases like carbon dioxide, oxygen and steam, etc.11-15, whereas, chemical activation is one stage process, carbonization and activation of a precursor occur synchronously in the presence of oxidative chemicals. Chemical activation has more advantages over physical activation with respect to higher yield, more surface area and better development of porous structure in carbon. It also helps to develop oxygenated surface complexes on the surface of activated carbon16. The surface oxygen functional groups can be easily introduced to the carbon by different activation methods like the reaction between the carbon surface and solutions of oxidizing agents such as phosphoric acid, nitric acid, hydrogen peroxide, zinc chloride, potassium permanganate, potassium hydroxide, etc. From the above oxidizing agents, phosphoric acid and zinc chloride are usually used for the activation of lignocellusic materials. However, phosphoric acid activation is widely preferred over zinc chloride because ZnCl2 has had environmental impact and the activated carbon produced when using it can not be used in the food and pharmaceutical industries17.
High surface area activated carbon has always fascinated researchers for its application as adsorbent. Chemical activation using phosphoric acid can easily open up pores and cavity of the prepared activated carbon to enhance their adsorption capability. Phosphoric acid activation has been applied to coconut shell, coals, hardwoods, shells of nuts like almond, pecan, black walnut, macadamia nut, cotton stalks and palm date pits18. Mariet19 showed that the reaction of a lignocellulosic precursor with a strong chemical reagent, such as phosphoric acid (H3PO4) at temperatures up to 350°C or above can lead to the formation of a porous carbon. Depending on the reagent, its concentration and the carbon precursor material, the carbon will possess a highly developed internal pore structure and be an effective adsorbent for molecules in the gas or liquid phase. Gonzalez-Serrano et al.20, showed that activated carbons with a high BET surface area and a well-developed porosity have been prepared from pyrolysis of H3PO4 impregnated lignin precipitated from Kraft black liquors. Impregnation ratios within the range of 1-3 and activation temperatures of 623-873 K have been used, giving rise to carbons with different porous and surface chemical structure. Increasing the activation temperature and the impregnation ratio leads to a widening of the porous structure with a higher relative contribution of mesoporosity. Armagan et al.21. In his study, investigated the activation features of phosphoric acid using waste peach stones as the raw material in the production of granular activated carbon. The activated carbon produced was found to be as good as that of the commercial ones. Physical properties of activated carbon, such as ash content and moisture content can affect the use of a granular AC and render them either suitable or unsuitable for specific applications. While the specific surface area of activated carbon and surface chemistry is classified as chemical properties.
In developing countries, the costs associated with manufacturing or more often importation of water-treatment chemicals tend to push the costs of processed water to levels that are not affordable to many people. In a largely agrarian country such as Jordan, there are so many agricultural by-products lying waste, littering and polluting the environment. The need to research on these wastes to convert them into agents of environmental control in order to reduce waste and conserve cost is the key to the present study. The objective of this study was to prepare and characterize activated carbon from local oak sawdust in order to relate the preparation conditions (carbonization and chemical activation) to the carbon’s essential physical, chemical and adsorptive properties. The chemical species used here was phosphoric acid (H3PO4). Surface chemistry was carried out by FTIR and Boehm titration method.
MATERIALS AND METHODS
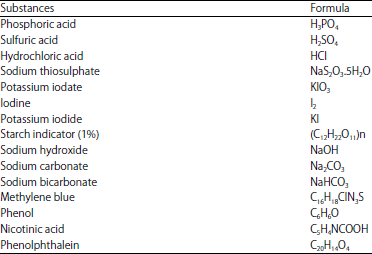
Materials: All the chemicals and reagents used were of analytical grade and procured from Merck, Fluka and Aldrich-Sigma. Following are the chemicals used in this study are listed in Table 1.
Preparation of activated carbons: Oak sawdust (SD) was collected from local sawmill. The SD was washed with hot distilled water to remove dust like impurities. Then it was dried in sunlight until all the moisture evaporated. This SD was used as a precursor for the preparation of two brands of carbons carbon. Firstly, 15 g of grounded raw SD was weighed into clean and weighed crucibles. They were introduce into an electric furnace at 500°C for 5 min after which they were poured from the crucible into a bath of ice water. The excess water was drained off. The carbonized sample was washed suing 0.1 M HCl to remove surface ash, followed by hot water wash and further washing with distilled water to remove residual acid. The resulting sawdust carbon (SDC) was then sun dried and further dried in the oven at 100°C for 1 h. The SD sample (30 g) was impregnated with 150 g of 3.07 N phosphoric acid (H3PO4) solution for 140 min under agitation at 160 rpm. The impregnated SD was charred in electrical furnace (Carbolite Philip Harris limited, UK) at 500°C for 35 min to ensure complete charring22. The carbonization temperature 500°C was chosen because higher temperature leads to excess carbonization, excess carbonization leads to the formation more ash there by produces larger diameter pores by pore widening and pore collapse, this also result in a decrease of surface area. The average weight loss of 37% was obtained during pyrolysis experiment. The charred carbonaceous material thus prepared is then cooled, washed with dilute ammonia solution and distilled water successively to make it completely acid free. The absence of phosphate ion in solution was also tested by addition of barium nitrate (Ba(NO3)2), the formation of precipitate (Ag3PO4) was not observed. The resulting phosphoric acid activated sawdust carbon (PASC) was oven dried at 110°C overnight and next kept in a tightly closed container for further characterization.
| Table 1: | Chemicals used in presented study |
 | |
Characterization of the activated carbons
Physical characteristics
Carbon yield: The total yields were determined after sample processing in terms of raw material mass. The dried weight, Wo of each pre-treated sample was determined using Metler balance and the carbon yield calculated as:
| (1) |
where, Ych is carbon yield (%), W is dried weight of AC prepared and Wo is pre-treated sample used in the carbonization and activation processes.
pH and conductivity: The standard test method for determination of activated carbon pH ASTMD3838-80 was used23. The 1.0 g of SDC and PASC was weighed and transferred into a beaker. One Hundred milliliters of distilled water was measured and added and stirred for 1 h. The samples were allowed to stabilize before the pH was measured. The pH was determined using a pH meter (metrohm 744, USA) and the conductivity was taken using a conductivity meter (Mettler Toledo, USA) at room temperature. Samples were run in duplicates.
Solubility in water and HCI: For water solubility (S), 0.5 g AC samples were added to 100 mL of distilled water in 250 mL flasks and shaken at 200 rpm in a shaker (Labortechnik mbH, Germany) for 2 h at ambient temperature. The mixtures were filtered through pre-weighed Whatman No. 1 size 15 filter papers. The filter papers containing the residual carbons were dried in the oven (Thermo lab industries) for 12 h at 105°C. After cooling to ambient temperature, in a desiccator, the filter papers together with residual carbons were weighed. The percentage solubility in water was calculated as the weight ratio of unrecovered carbon to the original sample. For solubility in HCl, the same method was followed but 0.2 M HCI was used instead of water24:
| (2) |
Moisture content: Thermal drying method was used in the determination of moisture content of the samples according to the method of Rengaraj et al.25. About 1 g of the powdered air dried activated carbon powder was taken in a previously weighed crucible. The crucible was placed in an electric hot air oven maintained at about 110°C. After one hour the crucible is taken out, cooled in a desiccator and weighed again. The loss in weight of the powder reported on percentage basis gives moisture content in the sample as:
| (3) |
Ash content: The ash content of the carbons was determined using the standards ASTM procedure26 D2866-94. This method involve heating the sample at 650°C for several hours until constant weight has been achieved. The ash is then weighed and the ash content of the AC is calculated as:
| (4) |
where, B is the weight of the crucible (g), C is the weight of crucible plus original carbon sample (g) and D is weight of crucible plus ash containing sample (g).
Determination of apparent density: An empty dry graduated cylinder was weighed. A samples of dry activated carbon SAC and PASC were packed into the cylinder and reweighed27 as:
| (5) |
Chemical characteristics
Acidic and basic groups: Surface oxygen groups on carbon materials are usually determined by titrations in aqueous solutions. The amount of acidity/basicity of functional groups was determined by Boehm titration method28. Boehm titration is an acid-base titration method which is used to determine the amount of surface oxygen groups (acidic or basic) present on carbon surfaces (activated carbon, carbon black, graphene, carbon nanotubes, etc). The NaOH solution was standardized by standard KHC8H4O4 and the HCl solution was standardized by Na2CO3 solution. Both Na2CO3 and NaHCO3 were dried in an oven at 110°C for 3 h before the standardizations were carried out. Similarly, all the produced chars were dried in the oven at 110°C for 3 h before the Boehm titration was carried out. A known amount of adsorbent was added into 50 mL of the solutions of NaHCO3 (0.1 N), Na2CO3 (0.1 N), NaOH (0.1 N) and HCl (0.1 N). The conical flasks were sealed and shaken at room temperature for 44 h. The solutions were then filtered and 10 mL of each filtrate was titrated with HCl (0.1 N) and NaOH (0.1 N). The acidic strength was calculated by measuring the volume of HCl consumed due to the titration with the solutions of NaOH, Na2CO3 and NaHCO3. Accordingly, the basic strength was calculated by measuring the volume of NaOH consumed in order to neutralize HCl. For the titration of HCl and NaOH solution, phenolphthalein was used as an indicator while for Na2CO3 and NaHCO3, the pH meter was used; the end points were 8.3 and 4.5 respectively. The basic group content on the char was calculated from the amount of HCl that reacted in the bottle. The acidic groups were calculated using the facts that:
| • | NaOH neutralizes carboxylic, phenolic and lactonic groups |
| • | Na2CO3 neutralizes carboxylic and lactonic groups |
| • | NaHCO3 neutralizes only carboxylic group |
Surface functional groups: The IR spectroscopy was employed to determine the presence of functional groups in the adsorbent at room temperature. The IR spectra of SDC and PASC were recorded using a MATTSON 5000 infrared spectrophotometer. A weight of sample (2 mg) was mixed with 100 mg of IR-grade KBr. The resulting mixture was grind into powder and pressed into KBr discs. The samples were scanned 20 times at 4 cm–1 resolution in the 4000-500 cm–1 range and then averaged.
Determination of the iodine number: For the iodine number test of Gimba and Musa29 was adopted. A stock solution was prepared containing 2.7 g of iodine crystals and 4.1 g of potassium iodide per litre. The prepared stock solution was standardized using a standard solution of sodium thiosulphate. Into a 100 cm3 volumetric flask, 0.5 g of the activated carbon and 10 cm3 of 5% v/v hydrochloric acid was introduced. The flask was swirled until the carbon was wetted. Then 100 cm3 of the stock iodine solution was added and agitated at a fast speed using an electric shaker for a period of 60 min. The mixtures were filtered through a sintered glass crucible. An aliquot portion (20 mL) was titrated with 0.1 M sodium thiosulphate using starch as indicator. The concentration of iodine adsorbed by the activated carbon at room temperature was calculated as amount of iodine adsorbed in milligrams from the following relationship:
| (6) |
where, Ms is molarity of thiosulphate solution (mol dm–3), Vs is volume of thiosulphate (cm3) for titration of carbon aliquot, Vb is volume of thiosulphate (cm3) for blank titration and Ma is mass of carbon sample (g).
Determination of the specific surface areas: The specific surface areas of SDC and PASC were determined using the methylene blue method described elsewhere30. The 2.0 g of SDC and PASC powder was dissolved in 200 mL of deionized water and 100 mg L–1 methylene blue solution was added to the adsorbent suspension and agitated in a shaker for 2 h. Thereafter, it was kept for 24 h to reach equilibrium, after which 10 mL aliquot was taken and centrifuged. The aliquot was then analyzed in a UV/visible spectrophotometer at maximum wavelength of 660 nm to determine the amount of methylene blue absorbed. Also 200, 300, 400, 600 and 700 mg L–1 solutions of methylene blue were then added sequentially and the previous steps repeated. A graph of concentration of MB added versus the absorbed MB was used to identify the point of complete cation replacement. The Specific Surface Area (SSA) was calculated as:
| (7) |
where, MMB is amount of MB absorbed at the point of complete cation replacement, Ms is mass of adsorbent, Av is Avogadro’s number and AMB is the area covered by MB molecule (assumed to be 130 A°2).
Determination of the phenol number: The 100 cm3 of 300 ppm solution of phenol was taken in a 250 cm3 leak proof reaction flask. The pH of the solution was adjusted to neutral by adding 0.1 N NaOH or 0.1 N H2SO4. A known weight of the activated carbon powder was added to it. The solution was equilibrated for 48 h at room temperature followed by filtration and subsequent analysis of the filtrate for phenol concentration. The residual concentration of phenol was determined by spectrophotometric analysis at a wave length of 269 nm31.
Adsorption of nicotinic acid: The study of nicotinic acid adsorption took place in a constant temperature bath (20°C) under continuous shaking. Ultra pure nicotinic acid was obtained from Merck. A stock nicotinic acid solution was prepared initially at concentration of 500 mg L–1, using distilled and deionised water. Equilibrium experiments were carried out by the placement of various amounts of activated carbons in 250 mL Erlenmeyer flasks and addition of the nicotinic acid solution. The mixtures were then shaken for 3 days, this time was considered adequate for nicotinic acid adsorption to reach equilibrium. At the end of the reaction time, the content of each flask was filtered and the filtrate was analyzed by a UV-spectrophotometer (Spectronic 20D) at 272 nm for the determination of nicotinic acid. The Removal Efficiency (RE) of nicotinic acid by the activated carbon at specific time and concentration was calculated according to Eq. 1:
| (8) |
where, Co and Ce (mg L–1) are the initial and equilibrium concentrations respectively.
RESULTS AND DISCUSSION
Carbons characterization
Physical characteristics: Characterization of the carbons is important in the formulation of a consistent quality carbon that can be used in water treatment plants or any other industrial applications. The physical characteristics of the prepared carbons are listed in Table 2. The carbon yield for SDC was 33.8% while, PASC showed 29.4% of carbon yield.
| Table 2: | Physical characteristics of SDC and PASC |
 | |
| SDC: Sawdust carbon and PASC: Phosphoric acid activated sawdust carbon | |
These values are typical range for most AC prepared from agro waste. The measured values for the pH, electrical conductivities, solubility in water and HCI, moisture content, ash content and densities of the SAC and PASC are shown in Table 2. The pH values for the SDC and PASC solutions were found to be 7.4 and 6.0 respectively. The pH Value of activated carbon is a measure of whether it is acidic or basic. The pH is defined as the minus logarithm of the hydrogen ion (H+) concentration. The dissociation constant, kW of water has a value of 10–14 at 25°C and is equal to [H+][OH¯]. The proton is hydrated according to Cπ+2H2O→Cπ·H3O++OH¯. Thus, for water in equilibrium with a carbon, if the water has a pH value <7, then the water is acidic from the dissociation of H+ from the surface oxygen groups, such as a carboxylic group, COOH32,33. The high pH values are indicative of soluble inorganic material, which may need to be removed from the AC for specific uses such as solvent recovery processes, adsorption gas chromatography and catalyst supports34. The surface of PASC was more acidic (pH = 6.0) than that of SAC (pH = 7.4). Too high pH indicates much contamination while too low pH indicates that acid wash was incomplete. The acidic properties of synthetic phosphoric acid activated carbons may be related to phosphorus-containing compounds formed during carbonization. It should be stressed that the acidic character of synthetic chemically activated carbons cannot be ascribed to an excess of phosphoric acid remaining in pores after carbonization, because the carbons were extensively washed with water until a neutral pH was attained. The chemical treatment of surface results in a large increase in number of acidic sites, mainly carboxylic groups, compared with basic sites. Moreover, the presence of acid functional group such as carboxyl, phenolic and others on the surface of carbon may cause the acidic property of activated carbon. Moreover, The reason for this may be due to the residual acid in the interior pores of activated carbon.
The electrical conductivities for the SDC and PASC solutions were found to be 144 and 327 μS cm–1, respectively as shown in Table 2. The low conductivity of SDC shows that this activated carbon has very little amount of charged and exchangeable ions. Residual ionic compounds on the surface and pores are completely washed out, which results in poor conductivity. The higher value of electrical conductivity for the PASC compared with that value for SDC can be attributed to the release of additional amount of proton [H+] having high ionic mobility and hence conductivity from the surface of PASC. Water solubility of the SAC and PASC were determined to be 6.7 and 8.4% respectively. While, the HCl solubility of the SAC and PASC were determined to be 7.8 and 9.3% respectively. Any degree of solubility in either water or acid is an indication of presence of impurities. Pure carbon does not dissolve in either water or acid since carbon is very unreactive due to lack of electron donating or accepting species in the structure such as lone pair electrons35. Moisture contents for SDC and PASC were 5.4 and 4.6% respectively as shown in Table 2. Generally, the recommended AC storage moisture content is <3%36. If the moisture content of AC is high, fungi and other micro-organisms degrade the carbon utilizing it in their metabolic processes. The micro-organisms can also multiply within the AC macro and micro-pores, blocking the pore structure, thereby reducing the adsorptive capacity of the carbon24. It should be noted that when exposed to air the AC are capable of adsorbing moisture from atmosphere. This normally could lead to high moisture content. Ash contents for SDC and PASC were 6.8 and 6.6% respectively as shown in Table 2. Ash content indicates the quality of an activated carbon. It is the residue that remains when carbonaceous portion is burned off. High-ash content raw materials contain high levels of impurities that lead to blockage of pores during the activation process reducing the surface area of the AC. Low ash content of PASC should impact positively on surface area parameter. Ash content can also affect activated carbon i.e., it reduces the overall activity of activated carbon. It also reduces the efficiency of reactivation, the lower the ash value therefore, the better the activated carbon for use as adsorbent29. The apparent density of 0.48 g cm–3 was got for SDC while 0.59 g cm–3 was got for PASC. The higher the density the better the filterability of activated carbons.
Chemical characteristics
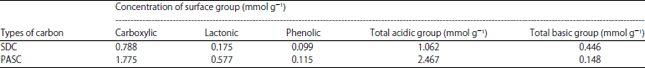
Acidic and basic groups: Boehm titration method was used to characterize the surface chemistry of the adsorbents. Carboxylic groups were therefore quantified by direct titration with NaHCO3. The difference between the groups titrated with Na2CO3 and those titrated with NaHCO3 was assumed to be lactones and the difference between the groups titrated with NaOH and those titrated with Na2CO3 was assumed to be phenol. Basic sites were determined by titration with HCl. Results of Boehm titration are showed in Table 3. Table 3 shows the amount of acidic and basic functional groups of the produced carbons.
The PASC sample shows the concentration of acidic groups (2.467 mmol g–1) which is greater than that of the SDC (1.062 mmol g–1). The H3PO4 treatment showed an increase in phenolics, carboxyl and lactone groups. The total acidity is considered to be the amount of NaOH neutralized and will consist of any organic acids with an acid dissociation constant (pKa) less than 12 and those acids formed when lactones hydrolyze.
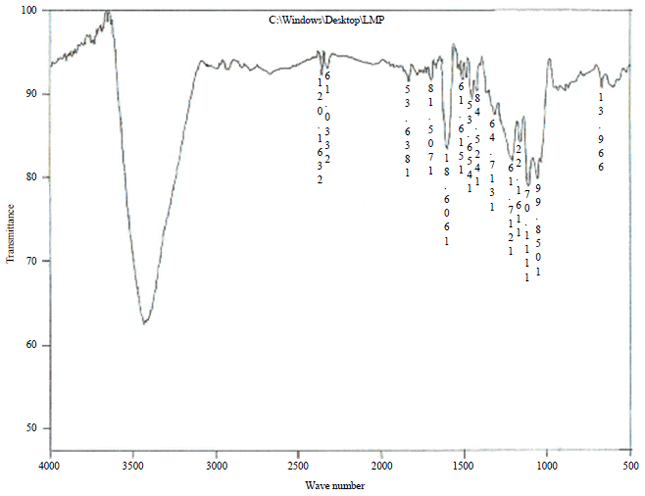 | |
| Fig. 1: | FTIR spectrum of SDC |
| Table 3: | Results of Boehm titration for SDC and PASC |
 | |
| SDC: Sawdust carbon and PASC: Phosphoric acid activated sawdust carbon | |
The amount of NaHCO3 neutralized is considered to be due to the strongest of the organic acids and is assumed to be primarily carboxylic acids, referred to as strong acid fraction. The amount of Na2CO3 neutralized is considered to be due to the strong organic acid fraction and the moderate strength organic acids (low pKa phenols) and lactones fraction. When lactones hydrolyze into carboxylic acids and phenols, the carboxylic acids and some or all of the phenols are expected to be neutralized37 by the Na2CO3. The results of the acid-base neutralization from the respective aqueous solutions given in Table 3 PASC shows more number of total acidic groups compared to basic groups which will also account for efficient uptake of different adsorbates.
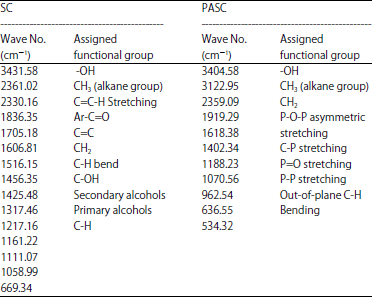
FTIR analysis: The infrared spectroscopy that provides information on the chemical structure of carbons is shown in Fig. 1 and 2 and the observed absorption bands in FTIR spectra of SDC and PASC are summarized in Table 4. The activated carbons showed a wide band at about 3404-3431 cm–1 that can be assigned to the O-H stretching mode of hexagonal groups and adsorbed water. The band at about 2330-2359 cm–1 is assigned for CH3 of alkane while that band at about 1836 cm–1 is assigned for C=C-H stretching. The FTIR bands can be assigned to the presence of carboxyls, lactones and phenols groups as reported by the surface chemistry analysis using Boehm method. The carboxyl groups show two main absorption features corresponding to a carbonyl (C=O) at 1705 cm–1 stretching vibrations in ketones, aldehydes, lactones and carboxylic groups and C-OH vibrations in the range of 1200-1300 cm–1. Peak appears38 at 1456 cm–1 shows the C-H bend in CH3.
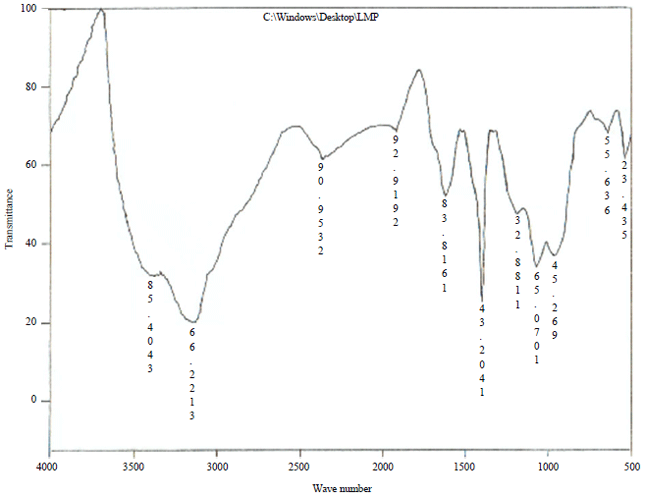 | |
| Fig. 2: | FTIR spectrum of PASC |
| Table 4: | FTIR data of SDC and PASC |
 | |
| SDC: Sawdust carbon and PASC: Phosphoric acid activated sawdust carbon | |
The peaks at 1058, 1217 and 1317 cm–1, attributable to C-O absorption, probably phenol, aromatic ether and OH deformation structure. The C-O stretching band at 1217 cm–1 confirms the lignin structure of the carbons. The peaks at 1606 cm–1, the well-known carbonaceous hand which is variously attributed to aromatic structure and to unconjugated, chelated carbonyl group and the peaks at 1618 cm–1 attributable to C-O stretch, probably carbonyl (conjugated C = 0) and/or carboxyl structure. The bands between 1161 and 1111 cm–1 are assigned to C-O stretching vibration in phenol and alcohol groups. The vibration of band 669 cm–1 was attributed to aromatic C-H bending. The band observed at 1402 cm–1 is ascribed to C-O-H stretching vibration in carboxylic group which shows stronger intensities at higher temperature. Peaks at 534 and 636 cm–1 are assigned to the out-of-plane C-H bending mode39. Some fundamental IR absorption frequencies of phosphorized activated carbon are given in Table 4. The IR spectrum shows absorption bands due to aliphatic (2359 2361 cm–1) and aromatic structures (2730 cm–1). A strong new band at 1618 cm–1 on the phosphoric acid treatment is assigned to C=O stretch in carboxylate, ketones, lactones and aldehydes. The absorption peak at (1170-1188 cm–1) reported the appearance of C-P bonds in carbon obtained from activation by phosphoric acid, this peak was assigned to phosphates. P-OH bending, P-O asymmetric stretching and P=O stretching were shown in phosphate-carbon complexes40.
 | |
| Fig. 3: | Adsorption isotherms of MB onto SDC and PASC |
| Table 5: | Adsorptive characteristics of SDC and PASC |
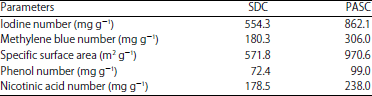 | |
| SDC: Sawdust carbon and PASC: Phosphoric acid activated sawdust carbon | |
It can be notified that increasing the intensity of the FT-IR spectra for C=O (carboxylic group), O-H (hydroxyl group) and C-O (phenol group) stretching vibrations indicate the presence more oxygen functional groups on the phosphorized activated carbon40. The processes occurring during the reaction of phosphoric acid at low temperatures are probably analogous to acid-catalyzed cracking reactions in coal liquefaction using Lewis acids. Acid catalysts can promote rupture of methylenic, etheric and thioetheric linkages between cyclic structures in carbon by ionic mechanisms which involve protonation followed by bond scission41. Proton donation from phosphoric acid to aromatic entities, -Ar- can be represented as:
| (9) |
Adsorptive characteristics
Iodine number: Adsorptive properties are directly linked with the porosity of activated carbon as the highly porous carbon can adsorb relatively large quantities of adsorbate. Iodine number can be correlated with ability to adsorb low molecular weight substances as the iodine molecule is relatively small and so provides a measure of surface area or capacity available to small adsorbates. Iodine numbers for commercial adsorbents range from 300-1200 mg g–1. Iodine number is a fundamental parameter used to characterise activated carbon performance. It is a measure of the micropore content of the activated carbon and is obtained by the adsorption of iodine from solution by the activated carbon sample. The micropores are responsible for the large surface area of activated carbon particles and are created during the activation process. It is in the micropores that adsorption largely takes place. Table 5 suggested that the values of iodine number obtained for PASC (872.4 mg g–1) is greater than the value obtained for SDC (554.6 mg g–1). The iodine number is used to measure the porosity of the activated carbon by adsorption of iodine from solution. Each 1.0 mg of iodine adsorbed is ideally considered to represent 1.0 m2 of activated carbon internal surface area. Hence, the internal surface areas of SDC and PASC can be estimated to be 554.6 and 872.4 m2 g–1 respectively.
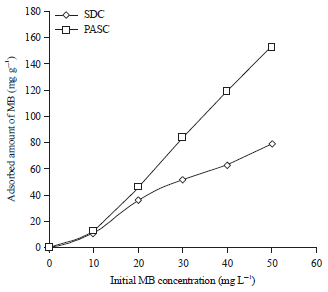
Methylene blue number and surface area: Methylene blue number indicates ability of adsorbing high molecular weight substances and colour. The adsorption isotherms of methylene blue onto PASC and SDC are shown in Fig. 3. By using Langmuir isotherm equation the methylene blue number obtained for PASC (306.0 mg g–1) was greater than the value obtained for SDC (180.3 mg g–1). Any carbon showing methylene blue number above 200, indicates the carbon with good activity for adsorption. So results of adsorptive properties indicate that sawdust is a very good source of raw material for the production of activated carbon of high efficiency. The surface areas calculated from methylene blue monolayer adsorption for PASC and SDC were found to be 970.6 and 571.8 m2 g–1 respectively.
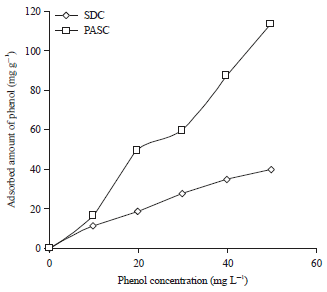
The adsorption isotherms of phenol onto PASC and SDC are shown in Fig. 4. The Langmuir maximum adsorption capacity of phenol on the resultant PASC and SDC was equal 99.0 and 72.4 mg g–1 respectively. The adsorption isotherms of nicotinic acid onto PASC and SDC are shown in Fig. 5. The Langmuir maximum adsorption capacity of nicotinic acid on the resultant PASC and SDC was equal 99.0 mg g–1 and 72.4 mg g–1 respectively. The results obtained show that the prepared activated carbons can serve as a good adsorbents.
Cost estimation: The economic viability of the adsorption process depends on the cost effectiveness as well as the availability of adsorbent. Because of their great capacity to adsorb pollutants ACs are the most effective adsorbents. However, commercial AC is quite expensive and the higher the quality, the greater the cost. The precursor use in the present study, sawdust, was collected free of cost from the nearby located sawmill.
 | |
| Fig. 4: | Adsorption isotherms of phenol onto SDC and PASC |
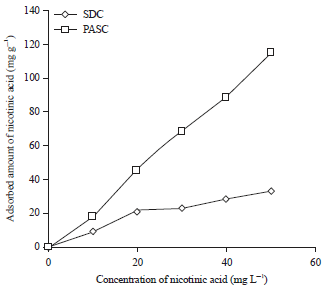 | |
| Fig. 5: | Adsorption isotherms of nicotinic acid onto SDC and PASC |
After consideration the expenses for transport ($20 t–1), chemicals ($40 t–1), electrical energy ($20 t–1) and man power ($20 t–1), the final developed carbon would cost approximately $100 t–1. The cheapest variety of commercial grade ACs available in Jordan cost approximately $900-$1100 t–1. This big difference in cost confirms the viable application of sawdust AC as a low cost adsorbent.
Results of this study could provide activated carbon consumers with cost effective and environmentally friendly alternative sources. The study aims to promote innovative use of local raw materials and relatively low-cost techniques in production of activated carbon. In the present studies related to the production of powdered activated carbon, by using oak sawdust, as a precursor material and phosphoric acid (H3PO4) as an activating agent are presented. Phosphoric acid activation has been applied to coconut shell, coals, hardwoods, shells of nuts like almond, pecan, black walnut, macadamia nut, cotton stalks and palm date pits18. Mariet19 showed that the reaction of a lignocellulosic precursor with a strong chemical reagent, such as phosphoric acid (H3PO4), at temperatures up to 350°C or above can lead to the formation of a porous carbon. The activated carbons thus obtained were characterized based on the following parameters viz., pH, conductivity, solubility, moisture content, ash content and apparent density. Chemical characterization of the resultant activated carbons was studied by Fourier transform infrared spectroscopy and Boehm titration which revealed the presence of oxygen containing surface functional groups like carboxylic, lactonic, phenolic in the carbons. The results of FTIR (Fourier-transform infrared spectroscopy) strongly support significant chemical and spectral changes occurring with the activation of the sawdust for phosphoric acid treatment. Phosphoric acid treatment resulted in the introduction of more acidic groups (carbonylic, phenolic, carboxylic and lactonic). The final developed carbon would cost approximately $100 t–1.
CONCLUSION
The results show that agricultural waste oak sawdust can be used as a promising precursor material for the production of low cost activated carbon. The result obtained also shows that these activated carbons can serve as a good adsorbents.
ACKNOWLEDGMENTS
The author acknowledges Mr. Sayel Alnezami for providing help in carbonization of sawdust samples and UV-VIS measurements, Mr Hmood Alhamaideh for help in FTIR analysis.
REFERENCES
- Linares-Solano, A., J.D.D. Lopez‐Gonzalez, M. Molina‐Sabio and F. Rodriguez‐Reinoso, 1980. Active carbons from almond shells as adsorbents in gas and liquid phases. J. Chem. Technol. Biotechnol., 30: 65-72.
CrossRefDirect Link - Nassar, M.M. and M.S. El-Geundi, 1991. Comparative cost of colour removal from textile effluents using natural adsorbents. J. Chem. Technol. Biotechnol., 50: 257-264.
CrossRefDirect Link - Bousher, A., X. Shen and R.G.J. Edyvean, 1997. Removal of coloured organic matter by adsorption onto low-cost waste materials. Water Res., 31: 2084-2092.
CrossRefDirect Link - Kadirvelu, K., M. Palanival, R. Kalpana and S. Rajeswari, 2000. Activated carbon from an agricultural by-product, for the treatment of dyeing industry wastewater. Bioresour. Technol., 74: 263-265.
CrossRefDirect Link - Rengaraj, S., B. Arabindoo and V. Murugesan, 1999. Preparation and characterisation of activated carbon from agricultural wastes. Indian J. Chem. Technol., 6: 1-4.
Direct Link - Carrasco-Marin, F., M.A. Alvarez-Merino and C. Moreno-Castilla, 1996. Microporous activated carbons from a bituminous coal. Fuel, 75: 966-970.
CrossRefDirect Link - Molina-Sabio, M., M.T. Gonzalez, F. Rodriguez-Reinoso and A. Sepulveda-Escribano, 1996. Effect of steam and carbon dioxide activation in the micropore size distribution of activated carbon. Carbon, 34: 505-509.
CrossRefDirect Link - Delannay, F., W.T. Tysoe, H. Heinemann and G.A. Somorjai, 1984. The role of KOH in the steam gasification of graphite: Identification of the reaction steps. Carbon, 22: 401-407.
CrossRefDirect Link - Lillo-Rodenas, M.A., D. Cazorla-Amoros and A. Linares-Solano, 2003. Understanding chemical reactions between carbons and NaOH and KOH: an insight into the chemical activation mechanism. Carbon, 41: 267-275.
CrossRefDirect Link - Gonzalez-Serrano, E., T. Cordero, J. Rodriguez-Mirasol, L. Cotoruelo and J.J. Rodriguez, 2004. Removal of water pollutants with activated carbons prepared from H3PO4 activation of lignin from kraft black liquors. Water Res., 38: 3043-3050.
CrossRefDirect Link - Armagan, B., M. Turan, O. Ozdemir and M.S. Celik, 2004. Color removal of reactive dyes from water by clinoptilolite. J. Environ. Sci. Health Part A: Toxic/Hazard. Subst. Environ. Eng., 39: 1251-1261.
CrossRefDirect Link - Kadirvelu, K., M. Kavipriya, C. Karthika, M. Radhika, N. Vennilamani and S. Pattabhi, 2003. Utilization of various agricultural wastes for activated carbon preparation and application for the removal of dyes and metal ions from aqueous solutions. Bioresour. Technol., 87: 129-132.
CrossRefDirect Link - Rengaraj, S., S.H. Moon, R. Sivabalan, B. Arabindoo and V. Murugesan, 2002. Agricultural solid waste for the removal of organics: Adsorption of phenol from water and wastewater by palm seed coat activated carbon. Waste Manage., 22: 543-548.
CrossRefDirect Link - Kalderis, D., D. Koutoulakis, P. Paraskeva, E. Diamadopoulos, E. Otal, J.O. del Valle and C. Fernandez-Pereira, 2008. Adsorption of polluting substances on activated carbons prepared from rice husk and sugarcane bagasse. Chem. Eng. J., 144: 42-50.
CrossRefDirect Link - Ioannidou, O. and A. Zabaniotou, 2007. Agricultural residues as precursors for activated carbon production-a review. Renew. Sustain. Energy Rev., 11: 1966-2005.
CrossRefDirect Link - Goertzen, S.L., K.D. Thariault, A.M. Oickle, A.C. Tarasuk and H.A. Andreas, 2010. Standardization of the Boehm titration. Part I. CO2 expulsion and endpoint determination. Carbon, 48: 1252-1261.
CrossRefDirect Link - Santamarina, J.C., K.A. Klein, Y.H. Wang and E. Prencke, 2002. Specific surface: Determination and relevance. Can. Geotech. J., 39: 233-241.
CrossRefDirect Link - Laszlo, K. and A. Szucs, 2001. Surface characterization of polyethyleneterephthalate (PET) based activated carbon and the effect of pH on its adsorption capacity from aqueous phenol and 2,3,4-trichlorophenol solutions. Carbon, 39: 1945-1953.
CrossRefDirect Link - Rodriguez-Reinoso, F., 1998. The role of carbon materials in heterogeneous catalysis. Carbon, 36: 159-175.
CrossRefDirect Link - Rodrıguez-Reinoso, F. and M. Molina-Sabio, 1998. Textural and chemical characterization of microporous carbons. Adv. Colloid Interf. Sci., 76-77: 271-294.
CrossRefDirect Link - Aloko, D.F. and G.A. Adebayo, 2007. Production and characterization of activated carbon from agricultural waste (Rice-husk and Corn-cob). J. Eng. Applied Sci., 2: 440-444.
Direct Link - Minkova, V., M. Razvigorova, E. Bjornbom, R. Zanzi, T. Budinova and N. Petrov, 2001. Effect of water vapour and biomass nature on the yield and quality of the pyrolysis products from biomass. Fuel Process. Technol., 70: 53-61.
CrossRefDirect Link - Helleur, R., N. Popovic, M. Ikura, M. Stanciulescu and D. Liu, 2001. Characterization and potential applications of pyrolytic char from ablative pyrolysis of used tires. J. Anal. Applied Pyrolysis, 58-59: 813-824.
CrossRefDirect Link - Valix, M., W.H. Cheung and G. McKay, 2004. Preparation of activated carbon using low temperature carbonisation and physical activation of high ash raw bagasse for acid dye adsorption. Chemosphere, 56: 493-501.
CrossRefDirect Link - Puziy, A.M., O.I. Poddubnaya, A. Martınez-Alonso, F. Suarez-Garcia and J.M.D. Tascon, 2002. Characterization of synthetic carbons activated with phosphoric acid. Applied Surf. Sci., 200: 196-202.
CrossRefDirect Link








