ABSTRACT
Floral and fruit tissues of two cultivars of plantain (Musa paradisiaca L.), P100-F (susceptible) and P200-I (resistant to cigar-end rot disease), grown side by side in a field plot in Benin City, Nigeria, were analysed for eight mineral elements-N, P, K, Ca, Mg, Fe, Na and Zn. Calcium, sodium and zinc concentrations were higher in bracts (0.151, 0.265 and 0.490%) and immature peel (0.520, 0.629 and 0.063%) of the P100-F than in samples of the P200-I cultivar (0.077, 0.210 and 0.032%) and (0.482, 0.490 and 0.032%), respectively. Concentrations of P and K were significantly lower in all tissues analysed of the susceptible (P100-F) than the resistant (P200-I) cultivar. The relevance of these results in relation to the management of cigar-end rot disease of plantain is discussed.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2006.822.826
URL: https://scialert.net/abstract/?doi=ajps.2006.822.826
INTRODUCTION
Mineral imbalance is one of several factors that pre-dispose plants to infection by micro-organisms (McNew, 1953). Plants with varied mineral status react differently to pathogen. While some diseases are severe on weakened and under-nourished plants, others are most destructive when plants are growing vigorously (Walker, 1957). The elements necessary for normal growth and development of many plants have been identified by several authors (Arnon and Hoagland, 1940; Delvin, 1967). Amongst these are N, P, K, Ca, Mg, Fe, Zn, Mn, Bo, etc. Each of these elements plays a significant role in the physiology and metabolism of the plant. Deficiency or excess of any may be reflected as disease disorder or influence disease development (Delvin, 1967).
Augmented concentrations of certain elements have been observed to aggravate the disease conditions of some plants (Ayanru and Sharma, 1986; Mohan and Rao, 1986). McNew (1953) reported that severity of potato scab and clubrot of cabbage, mustard and turnip increased with enhanced supply of either Ca, K or N. Similarly, Ayanru and Sharma (1986) reported enhanced concentrations of N, Mg and Na in roots of cassava plants infested by the green spider mite (CGM) as compared to those of non-infected. Mohan and Rao (1986) observed that corms of Robusta banana with bunchy top disease had higher P accumulation than those of healthy plants.
Augmented concentrations of some elements are required for the healthy state of some plants. Potassium is effective in improving resistance of wheat and barley against powdery mildew (McNew, 1953). Potassium and N are decisive factors in influencing infection of leaves by obligate parasites. Ayanru and Sharma (1986) also reported enhanced concentrations of Ca in non-infected cassava leaves as compared with those infected by the CGM, while concentrations of P and K in all the tissues and of Mg in leaves were significantly higher in healthy than infected cassava plants. The balance of nutrient elements is often of more importance than the absolute concentrations, while some elements are known to be toxic to plants when present in enhanced concentrations. Zinc and Mg are among such elements (Ayanru and Sharma, 1986).
Information on the effect of mineral status on the susceptibility or resistance of plantain to the cigar-end rot disease is lacking. This needs to be investigated as mineral nutrients are said to play a role in disease resistance through their effect on phytoalexin formation (Cruickshank et al., 1979). This study focuses attention on the role of nutritional status on the susceptibility/resistance of plantain fruits to cigar-end rot disease caused by Verticillium theobromae.
MATERIALS AND METHODS
Concentrations of mineral elements in two test plantain (Musa paradisaca) cultivars with regards to Cigar-end Rot Disease (CERD) incidence was studied. Fresh tissues of the two test cultivars, P100-F (susceptible) and P200-I (resistant) to CERD, were analysed for eight mineral elements, namely, calcium, iron, magnesium, nitrogen, phosphorus, potassium, sodium and zinc at the Biochemistry Department of the University of Benin, Benin City, Nigeria, in 2004. Tissues investigated were mature lamina (fully expanded), bracts (flowers) immature fruit peel and pulp (4-7 day old), mature (2 month old) fruit peel and pulp and ripe peel and pulp. The peel was excised from the pulp using a scalpel. The samples were pre-treated according to the methods of Jones and Steyn (1973). They were washed in Teepol, a demineralised water containing a 0.2% sodium hexametaphosphate solution, to remove surface dirt and rinsed thrice with distilled water. The samples were wrapped with Whatman No. 1 filter paper, put in clean muslin bags and dried for 48 h at 65°C in a forced draught Gallenkamp (Great Britain) oven. The samples were powdered using a Gallenkamp Laboratory micro hammer mill and stored in labeled glass bottles at 4°C in a refrigerator for chemical analysis.
Digestion of samples for determination of phosphorus, magnesium and calcium was carried out according to the method of Baker and Grewelling (1967). About 0.5 g of each of finely ground samples (using a micro hammer mill) was weighed into 25 mL Pyrex test tubes in triplicates. Then 5 mL concentrated nitric acid (HNO3), 2 mL concentrated hydrochlorous acid (HCLO4) and 2 mL concentrated sulphuric acid (H2SO4)were added to each test tube. The test tubes were heated in a digestion rack until dense white fumes of H2SO4 evolved and the digest was free from carbon particles. The digests were cooled, each diluted to 100 mL with distilled water and filtered through whatman No. 542 filter paper and the filtrate was saved for phosphorus, magnesium and calcium determination.
Phosphorus was determined by the phosphovanadomolybdate complex (Kitson and Mellon, 1944) method. A 5.0 mL aliquot of each sample digest was pipetted into each of 100 mL volumetric flasks in triplicate and 20 mL of vanadomolybdate solution was added to each flask. The mixture was made up to 100 mL with distilled water and the colour intensity was determined using Bausch and Lamb Spectronic 20 Spectrophotometer at 400 μm wavelength. A calibration curve was prepared using potassium dihydrogen phosphate (KH2PO4) solution. The phosphate content in the digest was read off from the calibration curve and expressed as a percentage of dry weight of sample.
Determination of calcium and magnesium was carried out by an adaptation of the method of Isaac and Kerber (1977) on the diluted solutions of extracts of the digested plant samples, using a Perkin Elmar Model 406 Atomic Absorption Spectrophotometer (AAS). To prevent phosphate interference, strontium nitrate solution was added to the digest before aspiration into the AAS. Calcium was determined at 4226 μm wavelength, while Mg was determined at 2852 μm in the infrared region, using appropriate lamps. The Ca and Mg contents of the blank digest solutions were read along with the plant digest solutions and expressed as a percentage of dry weight of sample.
For the determination of iron, 1.0 mL of a 1.0% sample solution was dispensed into each of six test tubes and 6.0 mL of distilled water added to each tube. This was followed by the addition of 1.0 mL of 30% potassium thiocyanide (KSCN) solution and 0.5 mL of 2N hydrochloric acid (HCl). Each mixture was thoroughly stirred, allowed to stand for 10 min and read in a Spectrophotometer (Junior Coleman, Model 6/20 Spec II) at 480 μm wavelength. A blank tube was prepared as described for the digests, but with water replacing the sample solution. The blank was read along with the test samples at the same wavelength. A calibration curve was prepared using a standard iron (Fe2+, 100 mg mL-1) solution. The iron content in the digest was read off from the calibration curve and expressed as a percentage of dry weight of sample.
Determination of zinc was carried out by an adaptation of the method of Belcher and Nutten (1960). The reagents used were as follows: 0.1 M EDTA (37.2 g L-1), 0.2 g Erichrome Black T indicator, ammonium chloride salt (17.5 g NH4 CI in 250 mL distilled water) and 1.0 M ammonium hydroxide (NH4OH). One hundred milliliter of each sample solution was dispensed into a 250 mL conical flask, followed by the addition of 2 mL NH4Cl buffer solution and few drops of the Erichrome black T indicator. The mixture was titrated with 0.1 M EDTA solution standardized with a standard zinc solution until the colour changed from wine red to blue. The zinc content of each sample was determined by the following guide;
One milliliter of 0.1 M EDTA solution is equivalent to 6.538 mg of Zn (Belcher and Nutton, 1960).
RESULTS
There were variations in the concentrations of mineral elements in the tissues of the two test plantain cultivars studied. Concentrations of Ca, Na and Zn in bracts and immature peel and concentrations of Mg and Fe2+ in bracts alone of the susceptible cultivar (P100-F) were significantly higher (p≤0.01) than those of the resistant cultivar, P200-I (Table 1 and Fig. 1).
| Table 1: | Comparative concentrations of Ca, Na and Zn in tissues of the two test plantain cultivars |
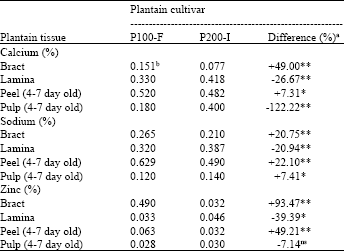 | |
| aDifference (%) = P100-F - P200-Ix100;/P100-F differences with * or ** are, significant at the 5 or 1% probability levels respectively; ns Not significant, bMeans of 3 replications | |
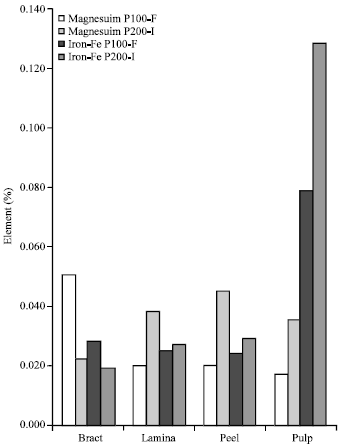 | |
| Fig. 1: | Comparative concentrations of magnesium and iron in tissues of the two test plantain cultivars |
The highest Ca content were in peels, similar to Na, while bracts had the highest Zn content. Zinc content in bracts of P200-I were highly significantly lower (p≤0.01) by 93.47%, compared to values in P100-F. Concentrations of P and K were significantly lower (p≤0.01) in all analysed tissues of susceptible (P100-F) as compared with those of the resistant cultivar (Fig. 2), using the t-test statistical analysis.
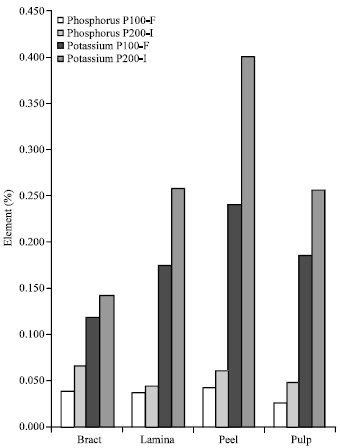 | |
| Fig. 2: | Comparative concentrations of phosphorus and potassium in tissues of the two test plantain cultivars |
| Table 2: | Comparative element concentrations in tissues of the susceptible plantain cultivar (P100-F) of different maturity categories (immature and mature) |
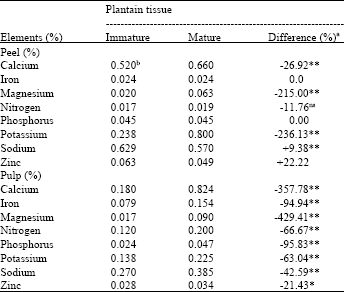 | |
| a Difference (%) = Immature - Mature/Immature x 100 differences with * or ** are, significant at the 5 or 1% probability levels, respectively; ns Not significant, b Means of 3 replications | |
| Table 3: | Comparative elemental concentration in tissues of the susceptible plantain cultivar (P100-F) of different maturity categories (mature green and ripe) |
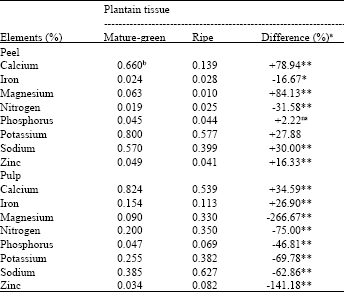 | |
| a Difference (%) = Mature green - Ripe/Mature x 100 green differences with * or ** are, significant at the 5 or 1% probability levels, respectively; ns Not significant. b Means of 3 replications | |
At different maturity categories (immature and mature, mature and ripe), Na and Zn were significantly higher, while Ca, Mg and K were significantly lower (p≤0.01) in the peel of the immature as compared with the mature (Table 2). Concentrations of all eight elements were significantly lower (p≤0.05 or 0.01) in the immature pulp as compared with the mature pulp (Table 2).
Calcium was consistently higher in the peel and pulp of the mature green as compared with the ripe. Mg, P, K, Na and Zn were significantly higher in peel, while they were comparatively lower in pulp of the mature green as compared with the ripe (Table 3). Iron was lower in the peel but higher in the pulp of the mature green as compared with the ripe. The t-test statistical analysis was used to compare the means.
DISCUSSION
Augmented concentrations of Ca, Na and Zn in bract and immature peel and Mg and Fe2+ in the bract of the susceptible (P100-F) as compared with the resistant cultivar (P200-I) observed in this study is of significance. Mineral elements play important roles in the physiology and metabolism of plants (McNew, 1953; Walker, 1957). Deficiency or excess of any may be reflected as disease disorder or influence disease development (Delvin, 1967).
Verticillium theobromae conidia infect dying bract parts (Slabaugh, 1994), finding their way into fruit tissues via wounds of perianth of young fingers (El-Helaly, 1954). Augmented concentrations of Ca, Na and Mg in diseased than healthy plant tissues have been reported by some other investigators. Such reports include those of McNew (1953), Marlatt and Orth (1970) and Ayanru and Sharma (1986) on plants, such as cabbage, mustard, turnip, Ficus elastica and cassava, respectively.
Concentrations of P and K diminished in all the tissues of the susceptible (P100-F) cultivar analysed as compared with the resistant (P200-I). Adequate concentrations of Ca in plantain have not been verified. However, concentration relative to that of K may be critical and have a role in plantain fruit susceptibility to cigar-end rot infection. McNew (1953) observed that the ratio of Ca to K is important in the mobilization of cationic nutrients. Such inbalance in the ratio of Ca and K may be responsible for the susceptibility of the P100-F cultivar. This proposition is not without precedent. McNew (1953) observed that potato scab infection increased as supplies of Ca were increased in the presence of inadequate supply of K.
Potassium is essential as an activator for enzymes involved in the synthesis of certain peptide bonds, regulating chemical reactions in plant cells. Phosphorus, on the other hand, is a constituent of nucleic acids, phospholipids, the coenzyme NAD and NADP and most importantly, of ATP (Delvin, 1967; McNew, 1953). The highest concentrations of these two elements are known to occur in the meristematic regions of actively growing plants (Nason and McElroy, 1963), where they are involved in the synthesis of nucleo-proteins. Deficiency of these elements, therefore, may affect such varied and important processes as respiration, transpiration, photosynthesis and chlorophyll development, thereby increasing the severity of many plant diseases (McNew, 1953; Nason and McElroy, 1963, Delvin, 1967). Deficiency, under most circumstances, implies thinner cell walls in epidermal tissues, reduced production of amino acids and slower growth of meristematic tissues that could retard replacement of diseased tissues (Delvin, 1967). As such, diminished P concentrations in the tissues of P100-F (susceptible) cultivar, as compared with those of P200-I (resistant) reported in this study (Fig. 2) is similar to the reports of Marlatt and Orth (1970), Ayanru and Sharma (1986), Mohan and Rao (1986) and may partly explain the differential susceptibility or resistance between the two test cultivars. Changes in plant tissues implied as a result of the deficiency of K and P concentrations may facilitate penetration of plantain fruit finger tip epidermis by pathogens. Destruction of plant tissues may also be promoted as a result of non development of new cells to replace those ravaged by the pathogen. The discernable patterns of elemental concentrations in relation to differences in plant age and cultivar types reported here may however, be species specific and merits further investigation.
REFERENCES
- Baker, J.K. and T. Grewelling, 1967. Extraction procedure for quantitative determination of six elements in plant tissues. J. Agric. Food Chem., 15: 340-344.
CrossRefDirect Link








