ABSTRACT
Comparison of the leaf epidermis of three species (A. canescens Welw. ex Bak., A. precatorius L. and A. pulchellus Wall. ex Thw.) and a new collection of Abrus adanson (simply referred to as Abrus sp.) found in tropical West Africa was undertaken by simple microscopy. The epidermal cells are irregular with wavy, sinuous or arcuate anticlinal walls, while the coastal cells are parallel, elongated and rectangular, terminating obliquely. Six types of stomata: paracytic (laterocyclic), diacytic, anomocytic, anisocytic, staurocytic and contiguous, were observed in the species. Anomocytic stomata are absent in A. canescens. The new collection Abrus sp., is distinguished from the other three species by its amphistomatic nature and possession of stomata in-groups of two or three bordered by six continuous subsidiary cells on its abaxial surface. It also has a Stomatal Index (SI) of 18.19, the highest in the genus. The other species are hypostomatic. Simple unbranched trichomes (long and short category) occur on both leaf surfaces of the species except in the new collection Abrus sp. where trichomes are confined to the abaxial surface. Trichome Index (TI) of 3.79 on the abaxial surface of A. canescens was observed to be the highest in the genus. Venation is of the camptodromous-brochidodromous type. Crystals are of common occurrence in the species. The study was aimed at improving systematic information on the tropical West African species of Abrus, which is hitherto lacking and with the collection of a new species, stimulate interest on Abrus taxonomy and conservation in the region.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2006.41.49
URL: https://scialert.net/abstract/?doi=ajps.2006.41.49
INTRODUCTION
Abrus Adanson is of the family Papilionaceae. In tropical West Africa, Dalziel[1], Hutchinson and Dalziel[2] and Burkill[3] briefly described three members of this genus. These are A. canescens Welw. ex Bak., A. pulchellus Wall. ex Thw. and the widespread A. precatorius L. The species are among the lesser known and utilized legumes although great potentials for exploitation abound for the genus[4]. Several medicinal, pharmacological and toxicological uses are ascribed to the species especially A. precatorius[1,3], including the preparation of liquorice from leaves and leafy twigs[5,6]. Despite the enormous uses of Abrus species in tropical West Africa as reported by Burkill[3], information on their taxonomy is almost lacking. Efforts to make fresh collections of A. canescens in the course of this study within Nigeria where it was reported to occur[2,3] were not successful; hence only herbarium samples were used. This may imply gradual extinction of the species without proper taxonomic documentation. There is also the problem of variability of the vegetative features and the conflicting reports of the exact number of species in the genus. The need therefore arises to commence detailed systematic studies on the species in tropical West Africa to provide factual taxonomic information based on fresh field investigations complemented by herbarium data to resolve some of these taxonomic controversies. A good taxonomic documentation of members of this genus will unarguably help in conserving their biodiversity.
The usefulness of epidermal characteristics in comparative anatomy and taxonomy in some members of the Leguminosae has been highlighted[7-11]. These reports utilized information from the structure, development and types of stomata, trichome and trichome types and various other quantitative and qualitative leaf epidermal characters to achieve taxa elucidation. Cutler[12] reported that apart from the usefulness of these characters in taxonomy, they are also effectively used in the identification of fossil leaf impressions in paleobotany and authentication of foliar drugs in pharmacognosy. Though Abrus species have found enormous use in ethnomedical practices in Tropical West Africa[1,3], information on the systematics of the genus is scanty amidst extinction treat to some species. Even where taxonomic information exists, there is need to resolves controversies associated with variability and overlaps in vegetative features, which creates difficulty in species identification. The detailed descriptive qualitative and quantitative epidermal data amassed herein together with a diagnostic key based on the epidermal features provides good taxonomic guide to the species. Also described are the epidermal characteristics of a new collection in this genus whose identity is undergoing confirmation. We hope that findings will be invaluable for conservation and biotechnological exploitation of this plant.
MATERIALS AND METHODS
Fresh and herbarium specimens were used in this study (Table 1). Herbarium specimens were obtained from Forestry Herbarium Ibadan (FHI) and University of Ibadan Herbarium (UIH). Voucher specimens of all the living materials studied were collected between 1998 and 2000 and are deposited in the University of Port Harcourt Herbarium (UPH).
The leaf epidermis of Abrus species were studied from either fresh samples (A. precatorius, A. pulchellus and the new collection Abrus sp.), fixed directly into the fixative formol acetic alcohol i.e., FAA (1 part formaldehyde, 1 part glacial acetic acid, 18 parts 70% ethanol v/v), or from herbarium materials (A. precatorius, A. pulchellus and A. canescens) using the scraping method[12], with modifications[13]. Initial soaking in water for 8-12 h first revived the herbarium samples. The surface to be examined was placed on a glass slide or tile while the other surface was carefully cleared by flooding with 5% sodium hypochlorite (domestic bleach) for about 2 min. Scraping was achieved with razor blade. The clear epidermal layers obtained were subsequently washed in several changes of distilled water, stained in 1% safranin for 1 min and temporarily mounted in glycerin. The preparations were observed and studied with a LEITZ microscope at objective lens 10 and 40 microscope magnification. Twenty microscope field views chosen at random were used to enumerate and study the number and types of stomata, trichomes and other epidermal cells.
| Table 1: | Examined sources of plant materials |
 | |
 | |
| UPH*: Fresh specimens deposited at the University of Port Harcourt Herbarium | |
Stomatal indices were computed according to Stace[14], stomatal type according to Metcalfe and Chalk[7], Stace[14] and Dilcher[15], trichome morphology and venation description according to Metcalfe and Chalk[7], Sasikala and Narayanan[16], trichome density and distribution according to Olowokudejo[17]. Photomicrographs were taken with a LEITZ DIAPLAN (Photomicroscope) fitted with LEICA WILD MPS 52 camera at objective lens of 10 and 40 magnification.
RESULTS
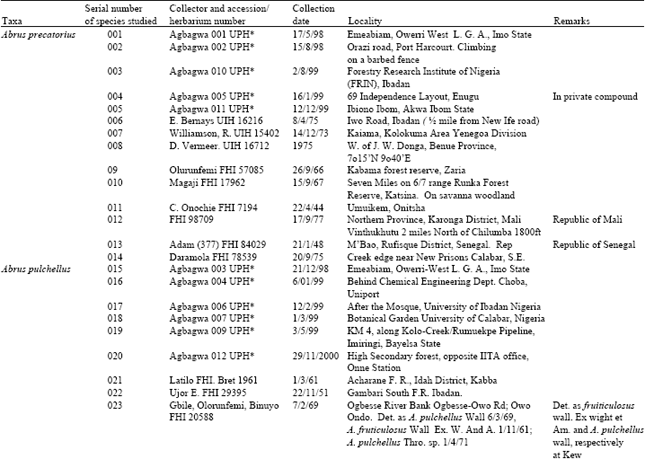
Results showed that the epidermis on both leaf surfaces of the species have one type of cell shape, i.e. irregular. The anticlinal wall of the leaf surfaces may be wavy, sinous or arcuate (Fig. 1-4 and Table 2). The adaxial anticlinal wall in A. precatorius and A. pulchellus is wavy and in Abrus sp. and A. canescens, they are sinuate and arcuate, respectively. Except in A. precatorius where the abaxial anticlinal wall is wavy, the other species show sinuate to arcuate anticlinal wall patterns. On the adaxial surface epidermal cell size varied from 42.20±17.31 x 15.06±5.06 μm in A. canescens to 73.80±47.68 x 12.04±3.44 μm in A. pulchellus. On the abaxial surface, the average size of cells of A. pulchellus and A. precatorius are 76.18±40.31 x 13.50±3.60 μm and 51.30±8.25 x 15.37±2.55 μm, respectively, while 31.86±16.83 x 16.61±6.11 μm for A. canescens. For the new collection Abrus sp., the epidermal cell sizes on both surfaces are the same (Table 2). A. pulchellus had the highest cell length while the new collection Abrus sp. scored the highest cell width. Values for epidermal cell size in A. canescens are more on the adaxial surface.
 | |
| Fig. 1: | a: (x 560)-showing adaxial leaf epidermis of A. precatorius, b: (x 875)- showing abaxial leaf epidermis of A. precatorius |
 | |
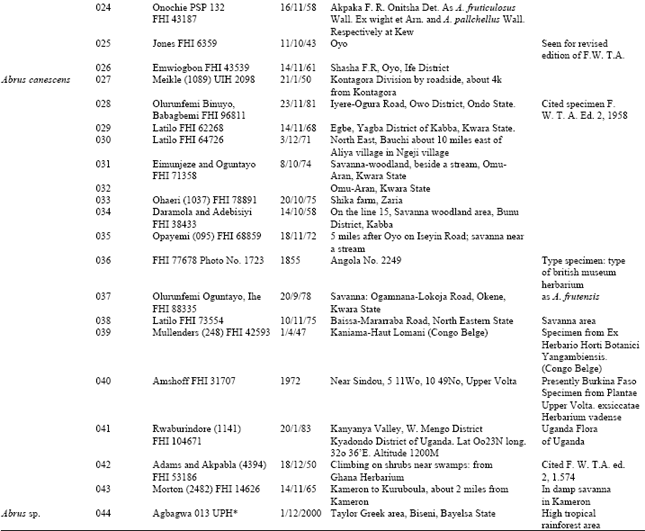
| Fig. 2: | a: (x 560)-adaxial leaf epidermis of A. pulchellus, b: (x 875)-abaxial leaf epidermis of A. pulchellus. Arrow shows contiguous stomata |
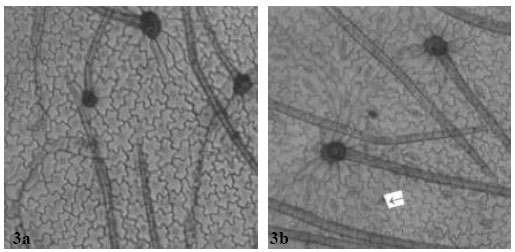 | |
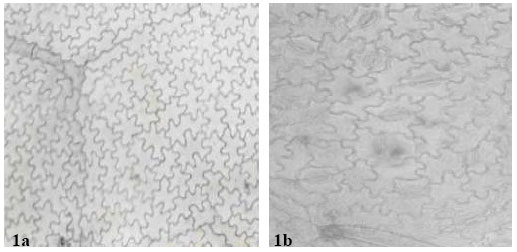
| Fig. 3: | a: (x 560) Showing adaxial leaf epidermis of A. canescens, b: (x 560)-showing abaxial leaf epidermis of A. canescens with several paracytic stomata. Arrow shows staurocytic stomata with four subsidiary cells |
 | |
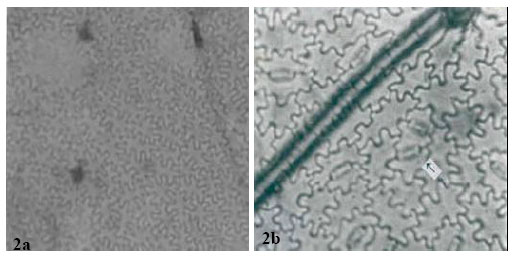
| Fig. 4: | a: (x 560)-Adaxial leaf epidermis of Abrus sp. Arrow points to a single stoma in the field view. Species is hypostomatic, b: (x 875)-Abaxial epidermis of Abrus sp. Arrow shows contiguous stomata. c (x 875)-Arrow shows three stomata joined by 6 paracytic cells. Double arrow shows semi-contiguous stoma |
| Table 2: | Epidermal cell characteristics of the leaf of Abrus species studied |
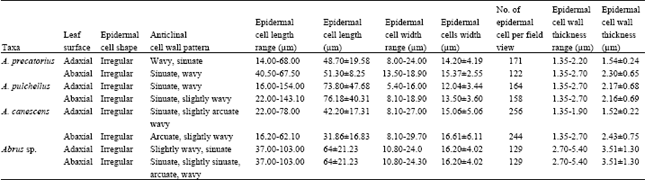 | |
The cell wall thickness varies generally from 1.35 to 5.40 μm on the adaxial and abaxial leaf surfaces of the species. A. precatorius, A. pulchellus and A. canescens have closely related cell wall thickness on their leaf. The new collection Abrus sp. has thicker epidermal cell wall. The costal cells are parallel, elongated and rectangular shaped, with long arms straight, arcuate or wavy and having straight to oblique end walls. The average epidermal cell number per field view varied from 129 in Abrus sp. to 256 in A. canescens on the adaxial surface and from 122 in A. precatorius to 244 in A. canescens on the abaxial surface.
| Table 3: | Stomatal and Cuticular features of the leaf of Abrus species studied |
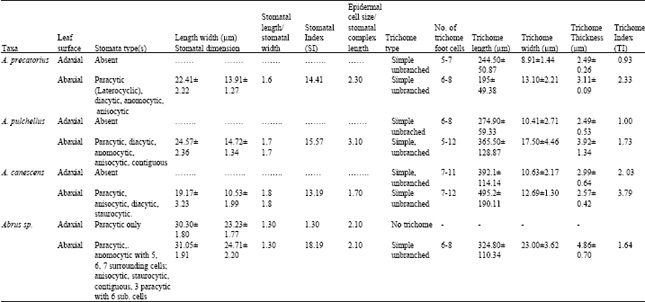 | |
All the species except the new collection of Abrus sp. have stomata on the abaxial epidermis (hypostomatic) only; Abrus sp. has stomata on both surfaces (amphistomatic) with stomata index of 1.30 on the adaxial surface. Six types of stomata were observed on the leaf surfaces: paracytic (laterocyclic), diacytic, anomocytic, anisocytic, staurocytic and contiguous (Table 3). Staurocytic stomata occurred only in A. canescens and Abrus sp. and anomocytic was absent in A. canescens. Contiguous stomata were observed in A. pulchellus and Abrus sp. Stomata in-groups of 3 bordered by 6 continuous subsidiary cells (Fig. 4c) were observed in the new collection of Abrus sp. Abrus sp. had the highest SI of 18.19. The stomatal sizes of the species ranged from 19.17±3.23 x 10.53±1.20 μm in A. canescens to 31.05±1.91 x 24.71±2.20 μm in the new collection Abrus sp. On the average, the stomatal sizes and indices of Abrus sp. are the highest followed by those of A. pulchellus (Table 3). Prismatic crystals were observed in the leaf surfaces of the species. They are sparingly distributed in few cells of A. precatorius, A. pulchellus and A. canescens, but occur ranging from cluster of four to several chains linked together in Abrus sp.
One type of trichome, the simple unbranched (long and short categories) was observed on the leaf surfaces of the four species. The adaxial surface of Abrus sp. has no trichome. The trichome or hair-base cells are polygonal, isodiametric or wavy on the leaf surfaces. The number of cells at the foot of the trichomes (trichome-base cells) varied from species to species (Table 3). These cells are highest in A. canescens with 7-11 and 7-12 cells on the adaxial and abaxial surfaces, respectively. The longest trichomes were observed on the abaxial surface of A. canescens i.e., 495.20±190.11 μm, while the least was recorded on the abaxial surface of A. precatorius i.e., 195±49.38 μm. Apart from the situation in A. precatorius where the average trichome length was higher on the adaxial surface, all other species had longer trichomes on their abaxial surfaces. Trichome index was highest on the abaxial surface of A. canescens (3.79) and least on the adaxial surface of A. precatorius (0.93).
In all the species, the venation is pinnate, camptodromous-brochidodromous. Primary vein is of moderate size, straight and unbranched with secondary veins diverging at 45°-60°; divergent angle almost uniform. Secondary veins branched, loop joining superadjacent secondaries at obtuse angle, enclosed by 2° or 3° arches. Intersecondary veins are composite, traversing the intercostal areas. Tertiary veins are percurrent, forked and more or less at right angles to the primary vein; a ratio of 1:1 alternate to opposite. Veinlet endings range from zero through linear to branched; branching up to twice. Areole well developed and oriented; mostly pentagonal with few guadangular.
DISCUSSION
The observed epidermal characters are of taxonomic relevance in the study of Abrus species. While the cell shape on the leaf surfaces of these species are generally irregular, the anticlinal wall pattern may be wavy, sinus or arcuate (Table 2), with wavy and sinuate pattern dominating. These similarities, which were also observed in the ranges and mean values of the epidermal cell wall thickness on the adaxial and abaxial surfaces of A. precatorius, A. pulchellus and A. canescens (Table 2 and 3), indicate relatedness and generic affinity. Similar findings have been made on other leguminous genera and used in taxa elucidation[9-11]. Epidermal cell numbers were more on the abaxial surfaces with species to species differences. Apart from the systematic importance of the occurrence of more epidermal cells on the upper surface of these species, a situation previously reported in another leguminous genus[11], this condition could be related to their hypostomatic nature. Thus, the stomata and its subsidiary or adjoining cells may have taken over positions initially occupied by epidermal cells on the lower surface.
Micromorphological studies of another leguminous genus revealed high variability in the dimensions of epidermal cells of the species[11]. Thus it was difficult to make clear distinction between the species using such character. This study shows that though variability in dimensions in epidermal cells of Abrus species occur, the final values over a range of samples are diagnostic at the species level (Table 2). Abrus pulchellus, a polyploid[18], is close in epidermal size with the new collection, Abrus sp. Since increased or large somatic cells has been reported as providing preliminary lead to the detection or confirmation of polyploids[19-22], it is likely that the new collection, Abrus sp. is a polyploid. The values of the epidermal size seem to be fairly but constantly high on the abaxial as against the adaxial surface. This may be connected with light related stimuli, as the species may have to guide against excessive loss of water.
The incidence of hypostomatic stomatal distribution in three species, A. precatorius, A. pulchellus and A. canescens shows the relatedness of these species despite other differences. However, the occurrence in the new Abrus sp. collection of amphistomatic condition is species diagnostic and further distinguishes it from the other ones. A similar but opposite situation was observed by Abubakar and Yunusa[10] who reported among four species of another legume Acacia, one hypostamotic and three hypoamphistomatic species. They concluded that the hypostomatic species, Acacia macrostachya, could be distinguished from the other three based on stomatal distribution. Some of the stomatal types observed in this genus have been previously reported in some other leguminous genera[9- 11,23]. The similarities in stomata types among these species as in Table 3 support their inclusion in the same genus. However, the observed differences like the occurrence of contiguous stomata in A. pulchellus and Abrus sp., the discovery of staurocytic stomata and lack of anomocytic type in A. canescens all serve diagnostic purposes at the species level. It is important to note that the occurrence of contiguous stomata is a feature associated with polyploidy. A special type of stomatal complex was observed in the new collection Abrus sp. (Fig. 4c and d) in which 3 stomata occurs in group and is bordered continuously by 6 subsidiary cells. The occurrence of stomata in groups is a useful generic diagnostic character in some families, which in the Papilionaceae has only been reported in Euchestra[7]. This advanced contiguous situation is thus important in the taxonomy of this new collection and the genus. Significant taxonomic variation on stomatal size and index useful in distinguishing species were observed on the leaf surfaces (Table 3). Carlquist[24] emphasized the contributions of the variation in stomatal size and index in delimiting species within a genus. Gill and Nyawuame[25] used similar characters in the systematics and phylogeny of the members of Bicarpillatae. It is noteworthy that the reduced stomatal index and dimensions in A. canescens could be more of an adaptation to functional since evidence from distribution proved that it exists in the xerophytic savanna areas. Thus the species has to device mechanisms to check water loss.
Ogundipe and Akinrinlade[11] have recently reported the occurrence of crystal sand and prismatic crystals in foliar epidermis of a leguminous genus Albizia. These were of common occurrence on the leaf surfaces of the Abrus species studied. Of particular note is the distribution of this crystal in the new collection Abrus sp. where they occur ranging from four to several chains linked together. The role of these crystals is not yet known.
The taxonomic value of trichomes has long been documented[7]. The presence of one type of trichome (Table 4), the simple unbranched type on the leaf surfaces of the species except the adaxial surface of the new collection Abrus sp. is an indication of affinity.
| Table 4: | Morphology, density and distribution of trichomes in Abrus species studied |
 | |
The trichome index and mean length observed on the abaxial surface of A. canescens, which is the highest in the genus, is an indication of high pubescence. This is probably an adaptation to savanna/xerophytic conditions. Foliar venation pattern as a diagnostic tool in taxonomy has been highlighted[7,26,27]. In the four Abrus sp. the venation pattern is generally pinnate, comptodromous- brochidodromous. Seetharam and Kotresha[28] recently employed foliar venation patterns to separate 12 species of the genus Bauhinia. into three groups. They further stated that this character could be used to identify the litter and fossil impressions of leaves of Bauhinia. Thus an insight into the leaf venation pattern as provided through this study not only supports the taxonomy of these species but also provides a lead to paleobotanical studies of the genus. A diagnostic key based on foliar epidermal characteristics of the Abrus species is presented below:
| 1. | Abrus species with contiguous stomata …………………………………. 2 |
| 11. | Abrus species without contiguous stomata ……………………………. 3 |
| 2. | Species amphistomatic, with stomata in-groups and cluster of crystals ……….. Abrus sp. |
| 21. | Species not amphistomatic and without stomata in-groups ……….. A. pulchellus |
| 3. | Stomata staurocytic, anomocytic type completely absent.. ….. A. canescens |
| 31. | Stomata not staurocytic, predominantly anomocytic, paracytic (Laterocyclic) and diacytic… A. precatorius |
REFERENCES
- Olowokudejo, J.O., 1990. Comparative morphology of leaf epidermis in the genus Annona (Annonaceae) in West Africa. Phytomorphology, 40: 407-422.
Direct Link - Agbagwa, I.O. and B.E. Okoli, 2005. Chromosome number and cytomorphological characterization of a polyploid Abrus. Afr. J. Biotechnol., 4: 607-610.
Direct Link - Allard, R.W., 1960. Principles of Plant Breeding. 1st Edn., John Wiley and Sons Inc., New York pp: 372-372.
CrossRefDirect Link - Carlquist, S.J., 1961. Comparative Plant Anatomy: A Guide to Taxonomic and Evolutionary Application of Anatomical Data in Angiosperms. Holt, Rinehart and Winston, New York, USA., Pages: 146.
Direct Link - Hickey, L.J., 1973. Classification of the architecture of dicotyledonous leaves. Am. J. Bot., 60: 17-33.
CrossRefDirect Link








