ABSTRACT
A glasshouse study was conducted to evaluate the effects of high temperature growth conditions on leaf nitrate reductase activity (NaR) and crude protein content in maize (Zea mays L.) plants and how these effects are influenced by ascorbate and kinetin applications. Both leaf NaR and crude protein declined significantly with plant age (p>0.05). There were no definite similarities between the patterns of variation in NaR activity and crude protein content. However ascorbate applications (but not kinetin) increased the correlative effects between NaR and leaf protein contents, in plants that were grown under high temperature stress conditions. Both NaR activity and protein contents were positively correlated with temperature. These correlations were higher between NaR and temperature than they were between crude protein content and temperature.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2006.363.367
URL: https://scialert.net/abstract/?doi=ajps.2006.363.367
INTRODUCTION
Metabolism in plant leaves is strongly affected by high temperatures. Among other effects on aspects of metabolism such as the widely reported effects on photosynthetic apparatus and systems (Davidson and Simon, 1983; Egorova and Bukhov, 2002; Egorova et al., 2003; Burke, 1990) and other enzymes of carbohydrate metabolism (Lenne and Douce, 1994), rapid temperature changes, particularly those toward the upper end of the adaptation range for a species, can produce significant changes in patterns of gene expression. These have been generally associated with the induction of thermo-tolerance, including the production of heat shock proteins (HSP’s) (Cooper and Ho, 1987; Li et al., 1999). In addition, prolonged heat shock in maize has been reported to result in the appearance of a novel set of proteins unrelated to HSP’s (Berna and Bernier, 1999), while Maevskaya et al. (2003), also reported the production germin-like proteins (GLP’s) in wheat plants subjected to various biotic and abiotic stress factors. Apart from the production of these classes of defensive/adaptive proteins, protein synthesis in plants under heat stress should increase as the plant strives to repair/replace damaged/denatured proteins (Pollock et al., 1993). On the other hand, protein synthesis is generally a very thermo-sensitive process and changes in temperature can affect both the amount and type of protein formed (Ajakaiye and Chukwuma, 1990). If the temperature is increased above a certain limit, the process declines (Lillo, 1991). Activity of the nitrate reductase enzyme is generally considered a rate-limiting step during protein synthesis (Hufton et al., 1996) and positive correlations between the activity of this enzyme and protein content of both vegetative parts and storage organs have been widely reported. However, several other workers have reported lack of direct relationships between NaR and protein content (Hufton et al., 1996). Maevskaya et al. (2003) have reported that there is a relationship between high temperatures and both in vitro and in vivo NaR activity in Barley (Hordeum vulgare L.) leaves and isolated intact spinach (Spinacea oleracea L.) chloroplasts.
Applications of growth regulating substances and anti-oxidants have been known to ameliorate the negative impacts of some abiotic stress factors in plants. This includes applications of cytokinins to alleviate heat stress in tobacco (Itai et al., 1978), Potato tubers (Mauk and Langille, 1978) Maize kernels (Cheikh and Jones, 1994). Ascorbate treatment has also been reported to counteract the deleterious effects of dehydration and heat stress in safflower (Gadallah, 2000), while a new class of plant hormones-the brassinosteroids, have been reported to reduce the impact of salt stress on growth, pigment loss and nitrate reductase activity in rice (Oryza sativa L.) (Anuradha and Seeta Ram Rao, 2003). Little information is available on how applications of these protective substances influence the relationships between physiological variables such as enzyme activity and contents of bio-molecules such as proteins, in plants grown under heat stress conditions.
This research was therefore undertaken to evaluate the effect of high temperature on the activity of the nitrate reductase enzyme in relation to crude protein content of maize (Zea mays L.) plants and how this is modified by exogenous applications of kinetin and ascorbate, under tropical conditions obtainable in the Nigerian savanna.
MATERIALS AND METHODS
Plant culture and treatments: The experiment was a glasshouse experiment conducted in the experimental gardens of the Department of Biological sciences, Ahmadu Bello University, Samaru, Zaria, between 1998-2000. Zaria is located on latitude 11° 11’ N; 7° 38’E in the Northern guinea savanna region of Nigeria and at an altitude of 686 m above sea level. Seeds of an open pollinated variety of Maize (Zea mays L.) were sown in earthenware pots of approximately 38 cm depth and 30 cm diameter. The pots were filled with about 155 cm3 garden soil to within 3-4 cm of maximum capacity. Fertilizer (NPK, 15:15:15) was applied at computed rate equivalents of NAERLS (1992) recommendations.
Plants subjected to heat stress were exposed to a mean air temperature of about 7.0° C higher than ambient and a mean soil temperature of about 5°C higher than ambient for 8-12 h, each day throughout the duration of the experiment. Maximum mean air temperature to which heat stressed plants were exposed during this period was 41°C in the air and 35.8°C in the soil. Mean day temperature was 37.3°C (air) and 32.2°C (soil). For plants grown under ambient conditions, maximum temperatures during this period were 32.0°C in the air and 29.0°C in the soil. Mean temperatures were 30.3°C (air) and 27.3°C (soil).
After 5 weeks of growth, sampling for NaR and protein contents started and this was maintained at weekly intervals for the next 5 weeks. After the first two samplings, freshly prepared solutions of ascorbate and kinetin (6-furfurylaminopurine) were applied as foliar sprays using a simple atomizer. Concentrations of 100, 200 and 300 ppm (parts per million) of each were applied to appropriately labeled pots, which were randomly assigned. Twenty to fifty (20-50) mL of each were applied till the leaf surfaces were properly wet and dripping. No wetting agents were used (Krishnayya and Date, 1996). Each treatment had two plants per pot and was replicated three times.
Sampling and analyses: Sampling was done in triplicates at weekly intervals, from each treatment. Leaf nitrate reductase activity (NaR) was estimated as the rate of nitrite formation using the in vivo assay method described by Kryult et al. (1996), Jain and Gadre (1998) and Tel and Hagarty (1984). Assays were made using samples collected from the uppermost fully expanded leaf and sections from apical, median and basal portions of the leaves were bulked. For each assay, 0.3 g of fresh leaf material was used. Colorimetric readings were done with a Corning (253) colorimeter set at 540 nm. Readings were converted to μmoles NO2 ¯ g-1 fr. wt. h-1, using readings obtained from a serial dilution curve of pure nitrite. Crude protein content of samples was estimated from leaf samples, which had been dried in an oven set at 60°C for three days and then ground to fine powder. Ten mg samples were used for each determination. Each of these was done in duplicate and figures obtained represented the Total Kjehldal Nitrogen (TKN). These figures were converted to Crude Protein (CP), as outlined by Tel and Hagarty (1984) as follows:
Where:
TKN = Total Kjehldal Nitrogen
6.60 = Nitrogen: Protein conversion factor for plant leaf protein.
Total Kjehldal Nitrogen, was estimated by the micro-kjehldal method (AOAC, 1975) and included nitrogen found in non-protein nitrogen containing compounds such as amino acids, vitamins, purines, etc. Results obtained were subjected to analysis of variance (ANOVA) (Parker, 1980) and correlation coefficients were computed.
RESULTS
Both NaR and crude protein contents of plant leaves declined significantly (p = 0.05) with plant age in all categories of plants (Table 1-4). Generally, NaR and crude protein contents tended to be higher in plants grown under high temperature conditions than in those grown under ambient conditions, except in plants to which kinetin was applied, where those grown under ambient temperatures had NaR levels that tended to be higher than in those grown under high temperature conditions. However, these differences were not statistically significant (Table 1-4).
Between treatment levels, 100ppm of both ascorbate and kinetin increased NaR significantly (LSD (0.05) = 2.29; 2.56, respectively) over other treatments and untreated plants, under high temperature growth conditions. At ambient temperatures however, ascorbate at this concentration increased leaf NaR significantly (LSD (0.05) = 2.07) only over other concentrations, but not he untreated plants (Table 1), while kinetin at 200 ppm increased NaR significantly (LSD(0.05) = 3.17) over other concentrations as well as untreated plants (Table 2).
For leaf crude protein contents, while plants treated with 100 and 300 ppm of both ascorbate and kinetin had significantly (LSD(0.05) = 2.59; 2.11, respectively) higher crude protein contents when grown under high temperature conditions (Table 3), they had significantly lower LSD(0.05) = 2.13; 2.03, respectively), crude protein contents when grown under ambient temperature conditions (Table 4).
| Table 1: | Leaf nitrate reductase activity (NaR/μmoles NO2 ¯ g-1 fr. wt. h-1) in leaves of maize (Z. mays L.) plants grown under ambient (B) and high (A) temperature regimes as influenced by ascorbic acid treatments |
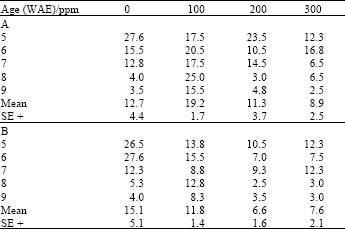 | |
| Table 2: | Nitrate reductase activity (NaR/μmoles NO2 ¯ g-1 fr. wt. h-1) in leaves of maize (Z. mays L.) plants grown under ambient (B) and high (A) temperature regimes as influenced by kinetin treatments |
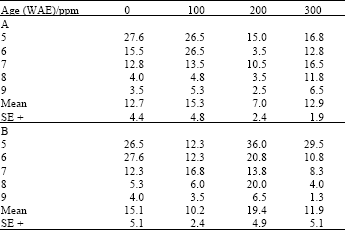 | |
| Table 3: | Crude protein content (%N) in leaves of maize (Z. mays L.) plants grown under ambient (B) and high (A) temperature regimes as influenced by ascorbic acid treatments |
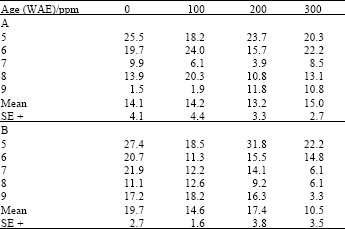 | |
| Table 4: | Crude protein content (%N) in leaves of maize (Z. mays L.) plants grown under ambient (B) and high (A) temperature regimes as influenced by kinetin treatments |
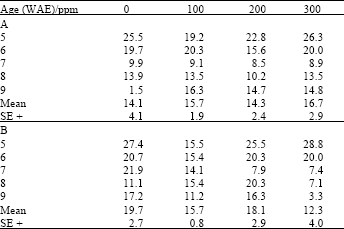 | |
| Table 5: | Correlation coefficients between leaves NaR, crude protein and air temperatures in plants grown under ambient and high temperature regimes as influenced by kinetin (KIN) and ascorbate (ASC) treatments |
 | |
| ns = not significant, * = significant, N = 10, p(0.05) = 0.63 | |
There were significant positive correlations between both leaf NaR and crude protein with temperature in all categories of plants. These correlations were generally higher between NaR and temperature than they were between leaf protein and temperature (Table 5).
DISCUSSION
That both NaR activity and protein synthesis showed sharp significant declines with plant age is a commonly observed trend in plants (Lillo, 1991). There is a tendency for higher NaR activity in younger tissues because of higher synthetic activities, which induce both structural and enzymatic proteins required for actively growing tissues.
Leaf NaR was not significantly increased by exposing the plants to high temperature growth conditions, although, ascorbate treatments tended to raise leaf NaR levels slightly, under high temperature while kinetin treatments had the same effects under ambient temperature. On the other hand, slightly higher crude protein contents occurred at higher temperatures as compared to ambient temperatures for both ascorbate and kinetin treatments. Lack of correlation between enzyme activity and crude protein observed in this study, seems to conflict with several observations which have been made in higher plants, Many studies have shown that there are correlations between increase in enzyme activity and protein content of vegetative as well as storage organs. In several species, the content of NaR associated proteins show fluctuations similar to enzyme activity (Hufton et al., 1996), leading to speculations that fresh enzyme synthesis and degradation are key factors in regulation of enzyme activity. However, many other workers have reported that this may not always be the case, as there are instances where NaR activity and protein contents were not correlated (Itai et al., 1978; Hufton et al., 1996). Furthermore, activity of the enzyme itself may represent very little proportion of leaf protein (Calza et al., 1987), although this enzyme plays a pivotal role in supply of nitrogen, growth and reproduction in plants especially cereals (Srivastava, 1995). The results of this study showed that ascorbate applications enhanced the correlative effects of NaR activity and leaf protein content and this was particularly so under high temperature stress conditions. The enhancement effect of ascorbate could be through the release of NADPH2 to the enzyme. The enzyme is NADPH2-dependent (Guerrero et al., 1981; Lea et al., 1994). NADPH2 is a common product of detoxification mechanisms involved in the quenching of free radicals generated under stress conditions (including water deficit) in plants (Zhang and Kirkham, 1996). On the other hand, that kinetin treatments did not show similar enhancement effects on NaR activity seemed to contradict reports by Lu et al. (1992) and Guerrero et al. (1981), that growth regulators such as cytokinins enhance NaR activity. This apparent contradiction is resolved when it is considered that cytokinins enhance stomatal opening and increase transpiration rates (Mansfield and Atkinson, 1990) and enhanced transpiration may impose water stress conditions, which will affect NaR negatively (Kaiser and Brendle-Behnisch, 1991). The role of ascorbate in enhancement of NaR activity in heat stressed plants however, seemed to be concentration-dependent, as lower ascorbate concentrations gave higher NaR activities than higher concentrations, suggesting that high ascorbate levels may be inhibitory to NaR. Similar results have been reported on the enhancement of NaR in salt-stressed rice plants treated with brassinosteroids (Anuradha and Seeta Ram Rao, 2003).
Correlations between NaR, protein and temperature emphasize the fact that activity of the enzyme is a measure of the habitat-dependent utilization of nitrate in plants (Larcher, 1995). Activity of NaR is known to fluctuate in response to changes in environmental factors such as light, temperature, pH, CO2 and O2 tension, water potential, nitrogen source and other factors, which affect cell permeability such as carbohydrates and hormones (Guerrero et al., 1981). Results of this study suggest the possibility that, the water relations of maize plants grown under heat stress conditions did not allow for full expression of NaR activity and subsequent protein synthesis. Further, Onwueme et al. (1971) observed in barley plants that exposure to supra optimal, but non-lethal temperatures reduced NaR activity progressively, suggesting that NaR is one of the first enzymes to be inactivated by high temperature. On the other hand, Maevskaya et al. (2003) reported that exposing both barley and spinach leaves and chloroplasts to sub-lethal high temperatures increased in vivo and in vitro NaR, but temperatures above 40°C decreased NaR.
REFERENCES
- Anuradha, S. and S.R. Rao, 2003. Application of brassinosteroids to rice seeds (Oryza sativa L.) reduced the impact of salt stress on growth, prevented photosynthetic pigment loss and increased nitrate reductase activity. Plant Growth Regul., 40: 29-32.
Direct Link - Berna, A. and F. Bernier, 1999. Regulation by biotic and abiotic stress of a wheat Germin gene encoding oxalate oxidase, a H2O2-producing enzyme. Plant Mol. Biol., 39: 539-549.
Direct Link - Calza, R., E. Huttner, M. Vincent, P. Ranze, H. Vaucheret and L. Cherel, 1987. Complimentary to tobacco nitrate reductase m-RNA and encoding epitopes common to the nitrate reductase from higher plants. Mol. Gen. Genet., 209: 552-562.
PubMedDirect Link - Cooper, P. and T.H.D. Ho, 1987. Intracellular localization of heat shock proteins in maize. Plant Physiol., 84: 1197-1203.
Direct Link - Egorova, E.A., N.G. Bukhar, U. Heber, G. Samson and R. Carpenter, 2003. Effect of pool size of stromal reductants on the alternative pathway of electron transfer to photosystem I in chloroplasts of intact leaves. Fiziol. Rast.i/Russian J. Plant Physiol., 50: 485-495.
Direct Link - Gadallah, M.A.A., 2000. Effect of acid mist and ascorbic acid treatment on the growth, stability of leaf membranes, chlorophyll content and some mineral elements of Carthamus tinctorius, the safflower. Water, Air Soil Pollen, 118: 311-327.
Direct Link - Guerrero, M.G., J.M. Vega and M. Losada, 1981. The assimilatory Nitrate-reducing system and its regulation. Ann. Rev. Plant Physiol., 32: 169-204.
CrossRef - Hufton, C.A., R.T. Besford and A.R. Wellburn, 1996. Effects of NO (+NO2) pollution on growth, nitrate reductase activities and associated proteins contents in glasshouse lettuce grown hydroponically in winter with CO2-enrichment. New Phytol., 133: 495-501.
Direct Link - Jain, M. and R. Gadre, 1998. Inhibition of chlorophyll synthesis and enzymes of nitrogen assimilation by selenite in excised maize leaf segments during greening. Water Air Soil Pollut., 104: 161-166.
CrossRefDirect Link - Kaiser, W.N. and E. Brendle-Behnisch, 1991. Rapid modulation of Spinach leaf nitrate reductase activity by photosynthesis I: Modulation of in vivo by CO2 availability. Plant Physiol. 96: 303-367.
Direct Link - Kryult, M., A. Karolak and A. Bytrnerowicz, 1996. Nitrate reductase activity as an indicator of Ponderosa pine response to atmospheric nitrogen deposition in the San Bernardino mountains. Environ. Pollen, 93: 141-146.
CrossRef - Larcher, W., 1995. Physiological Plant Ecology Ecophysiology and Stress Physiology of Functional groups. 3rd Edn., Springer-Verlag, Berlin/Heidelberg, Germany, ISBN: 9780387581163, Pages: 506.
Direct Link - Lenne, C. and R. Douce, 1994. A low molecular weight heat-shock protein is localized to higher plant mitochondria. Plant Physiol., 105: 1255-1261.
PubMedDirect Link - Li, Q., D.W. Haskell and G.L. Guy, 1999. Co-ordinate and non-coordinate expression of heat stress 70 family and other molecular chaperones at high and low temperatures in spinach and tomato. Plant Mol. Biol., 39: 21-34.
CrossRefDirect Link - Lillo, C., 1991. Diurnal variations of corn leaf nitrate reductase: An experimental distinction between transcriptional and post-transcriptional control. Plant Sci., 73: 149-154.
Direct Link - Lu, J.L., J.R. Ertl and C.M. Chen, 1992. Transcriptional regulation of nitrate reductase mRNA levels of cytokinin-abscissic acid interactions in etiolated barley leaves. Plant Physiol., 98: 1255-1260.
PubMedDirect Link - Maevskaya, S.N., E.A. Egorova and N.G. Bukhav, 2003. Effect of elevated temperature on nitrate and nitrate reduction in leaves and intact chloroplasts. Fiziol. Rast./Russian J. Plant Physiol., 50: 675-679.
Direct Link - Mauk, C.S. and A.R. Langille, 1978. Physiology of tuberization in Solanum tuberosum L. Plant Physiol., 62: 438-442.
Direct Link - Onwueme, I.C., H.M. Lande and R.C. Huffaker, 1971. Nitrate reductase activity in relation to heat stress in barley seedlings. Crop Sci., 11: 195-200.
Direct Link - Zhang, J. and M.B. Kirkham, 1996. Antioxidant responses to drought in sunflower and sorghum seedlings. New Phytol., 132: 361-373.
Direct Link








