ABSTRACT
Chloroindole auxins are useful tools in understanding the signal transduction pathways of the auxins. However, their presence is not acclaimed in majority of the plants. This gives an impression that these auxins could be having certain roles in plants enriched with them. Therefore, the seeds of Solanum melongena (L.) cv. Pusa were soaked in 0, 10-8 or 10-6 M aqueous solutions of IAA, 4,Cl-IAA, 5,Cl-IAA, 6,Cl-IAA, 7,Cl-IAA, 4,6,Cl2-IAA and 4,7,Cl2-IAA for 8 h and cultivated in clay pots. All the auxins improved the growth and NR activity compared with water soaked control. 4,Cl-IAA was most prominent in its effect, generating 29, 54, 30, 34 and 47% higher values for shoot and root length, fresh and dry mass and NR activity, respectively, in 15 day old plants. The order of response to various auxins was 4,Cl-IAA>7,Cl-IAA>6,Cl-IAA = 5,Cl-IAA = 4,6Cl2-IAA = 4,7Cl-IAA>Control.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2006.14.16
URL: https://scialert.net/abstract/?doi=ajps.2006.14.16
INTRODUCTION
The plant hormone, Indole-3-acetic Acid (IAA) is ubiquitous in plants (Davies, 1995). However, chloro-substituted auxins of rare occurrence in plants. 4-chlorindole –3-acetic acid (4,Cl-IAA) has been identified in the extracts of Vicieae (Gandar and Nitsch, 1967) and the shoot, root and the cotyledons of 3 day old etiolated seedlings of Pisum (Schneider et al., 1985). Both monochloroindole acetic acids (4,Cl-IAA, 5,Cl-IAA, 6,Cl-IAA and 7,Cl-IAA) and dichlorindole acetic acids (4,6Cl2-IAA and 4,7Cl2-IAA) are known to have many times more auxin like activity (Reinecke et al., 1995) in various bioassays. They stimulate the growth of excised tissue (Katekar and Geissler, 1982), rooting and ethylene production in pea cuttings (Ahmad et al., 1987) and the synthesis of specific enzymes in detached cotyledons (Hirasawa, 1989).
Chlorosubstituted auxins are also known to increase the activity of carbonic anhydrase, net photosynthetic rate (Ahmad et al., 2001a) in Brassica and α-amylase activity (Ahmad et al., 2001b) in wheat grains. The response of chloroindole acetic acids varied with the plant type (Reinecke et al., 1998). Therefore, in the present study the effect of chloroindole auxins on growth and nitrate reductase activity of Solanum melongena is being reported.
MATERIALS AND METHODS
The experiment was conducted in the year 2003-04, in the net house of the Department of Botany, Aligarh Muslim University, Aligarh, India. The authentic seeds of Solanum melongena (L.) cv. Pusa were surface sterilized with sodium hypochlorite solution followed by five washings with sterilized double distilled water and subsequently soaked in 0, 10-8 or 10-6 M aqueous solutions of IAA, 4,Cl-IAA, 5,Cl-IAA, 6,Cl-IAA, 7,Cl-IAA, 4,6-Cl2-IAA and 4,7-Cl2-IAA for 8 h. Each treatment was replicated five times. The soaked seeds were sown in 15 cm clay pots containing autoclaved loamy soil and compost in a ratio of 3:1. The fresh and dry mass of seedlings, the length of the shoot and root and leaf Nitrate Reductase (NR) activity (Jowarski, 1971) were studied at 15 day after sowing. All the data were subjected to statistical analysis by SPSS 7.5 and LSD was calculated at 0.05 level of probability.
RESULTS AND DISCUSSION
The seedlings, at 15 day stage, had values for most of the characteristics, significantly different from each other (Table 1). The growth and the activity of NR, in the leaves was favoured by seed treatment with auxins. However, among the auxins, best response was generated by 4,Cl-IAA followed by 7,Cl-IAA. The former, increased the shoot and root length, fresh and dry mass and NR activity by about 29, 54, 30, 34 and 47% over the water soaked control, respectively (Table 1). NR is responsible for the initiation of nitrate metabolism and consequently for the protein synthesis at various levels of plant body.
| Table1: | Effect of various auxins on shoot and root length (cm), fresh and dry mass (mg) and leaf NR activity (n mol NO2 g-1 h-1) in the seedlings of Solanum melongena at 15 days |
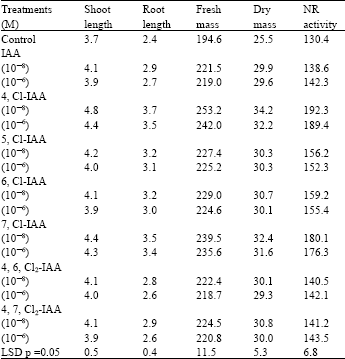 | |
However, the rate of nitrate reduction is possibly a repercussion of co-ordinated action of three main variable factors, by involving some form of signal transduction and/or sensors (Campbell, 1999). The substrate (nitrate) concentration which essentially induces functional NR (Saroop et al., 1998) produces a nitrate sensing protein of unknown nature that presumably binds with the regulatory regions of NR-genes and transcripts NR-mRNA and other regulator proteins involved in the metabolic response (Redinbaugh and Campbell, 1991; Crawford, 1995). The activity of NR is very much unsteady and depends on the presence of hormones such as gibberellic acid and/or cytokinin (Roth-Bejerano and Lips, 1970) auxin or its substituents (Ahmad and Hayat, 1999) and/or monochloroindole acetic acids (Ahmad et al., 2001a). Luxuriant growth of the seedlings, as evident from the characteristics observed (Table 1), may be an expression of the effect of the chloro-indole acetic acid at the level of the cell division, enlargement and/or making available more and more organic nitrogen because of the elevated level of NR which could have activated the process of nitrate reduction (Table 1). This effect of the auxins will automatically help the early establishment of plant, particularly in areas facing one or the other environmental stress.
The superiority of 4,Cl-IAA over IAA was found in elongation of Avena coleoptiles (Marumo et al., 1974), rooting, growth, ethylene bio-synthesis (Ahmad et al., 1987) and induction of α-amylase activity in cotyledons of pea (Hirasawa, 1989) and wheat seeds (Ahmad et al., 2001b). These differential responses to various auxins are presumably an expression of their varied degree of degradation and/or conjugation and other related properties in which they differ from each other and the IAA (Engvild, 1994). Moreover, the impact of 4, Cl-IAA on the receptor of signal transduction pathway was also suggested as another reason for its higher auxin like activities (Reinecke et al., 1998). The order of response to various auxins was 4,Cl-IAA>7,Cl-IAA>6,Cl-IAA = 5,Cl-IAA = 4,6Cl2-IAA = 4,7Cl2-IAA>control.
ACKNOWLEDGEMENTS
Authors thank Dr. K.C. Engvild, Riso National Laboratory, Roskilde, Denmark, for the generous supply of chloroindole acetic acid. Financial assistance rendered by Department of Science and Technology, New Delhi to S. Hayat is gratefully acknowledged.
REFERENCES
- Shankar, N.K., S.R. Khan and H.S. Srivastava, 2001. The response of nitrate reductase activity and nitrate assimilation in maize roots to growth regulators at acidic pH. Biol. Plant., 44: 599-601.
Direct Link - Jaworski, E.G., 1971. Nitrate reductase assay in intact plant tissues. Biochem. Biophys. Res. Commun., 43: 1274-1279.
CrossRefDirect Link - Redinbaugh, M.G. and H.W. Campbell, 1991. Higher plants responses to environmental nitrate. Physiol. Plant., 82: 640-650.
Direct Link - Saroop, S., V.S. Thaker, S.V. Chanda and Y.D.Singh, 1998. Light and nitrate induction of nitrate reduction in kinetin and gibberellic acid treated mustard cotyledons. Acta Physiol. Plant., 20: 359-362.
CrossRef - Schneider, E.A., C.W. Kazakoff and F. Wightman, 1985. Gas chromatography mass spectrometry evidence for several endogenous auxins in pea seedlings organs. Planta, 165: 232-241.
CrossRef








