Research Article
Factors Influencing the Efficiency of Extraction of Polyphenols from Young Tea Leaves
Not Available
Deepu Vijayan
Not Available
R. Raj Kumar
Not Available
R. Premkumar
Not Available
Tea is mainly grown in Assam, Kerala, Tamil Nadu and Himachal Pradesh in India. Tea plantation in south India is going through a crisis, due to the import of tea from other tea growing countries where the production cost is comparatively low. To overcome these difficulties, novel methods have to be adopted other than the production of black tea. Product diversification is an alternative which can be adopted in the present tea manufacturing methods.
Tea plants contain various biochemical constituents, among which polyphenols and catechins are in higher amount. Polyphenols, particularly catechins gained much attention due to its therapeutic values such as anti cancerous, anti allergic and anti bacterial properties[1-3]. Nowadays polyphenols are having a high market value. Extraction of polyphenols from tea plants in higher quantity can be an additional source of revenue for the tea industry. Even though many procedures are available to extract the polyphenols from plant materials, application oriented studies are not adequate. Type and percentage of solvent used for extraction, nature of the leaves, leaf conditions, time of harvesting, acidification percentage and its type, drying temperature and storage temperature are the major factors affecting extraction of polyphenols[4].
Our earlier studies showed that methanol is the best solvent to extract polyphenols, since the processing cost is low with high yield[5]. Young leaf bits gives high yield when compared to other plant parts. In the present study we have developed a novel extraction method having advantage over other conventional methods.
Fresh tea shoots comprising of two leaves and a bud were collected from selected tea bushes of clone UPASI 9 from UPASI Experimental Farm. This work was conducted from the month of October to November 2004 at UPASI Tea Research Institute. Three different leaf conditions were selected for this study. In the first treatment, the leaf as such is processed for extraction. In the second treatment, the shoots were placed in an oven for a period of 24 h at 70°C and in the final treatment the leaf samples were frozen at -40°C for a period of 12 h.
Three types of treatments were given; 500 mL of methanol was added to the frozen cut leaves and placed in the shaker for 1h at 200 rpm. The liquid was collected separately and again 500 mL of fresh methanol was added and processed the same and the extract was pooled together. For the second experiment, the frozen leaves were macerated by using a mortar and pestle which are kept in ice cold conditions. With the macerated samples the methanol 500 mL (50°C) was added twice. The final experiment was done by refluxing the sample leaf with 500 mL methanol at the temperature of 50°C[6].
Percentage of the extracting solvent plays an important role in the percentage of polyphenol yield. Hence we used different percentage of the extraction solvent methanol (60, 70, 80, 90 and 100%) and processed for the extraction of the phenolic compounds. Acidification is another important factor for the complete extraction of the phenolic constituents from tea leaves. The extracting solvent was acidified with different percentage (1, 2 and 3) of hydrochloric acid and acetic acid and then the extraction was carried out[7].
From the plant sources the complete extraction of phenolic compounds required number of steps with fresh solvents. In order to confirm this, the percentage of extracted compounds was quantified after every round of extraction. We tried with fresh methanol for five times to analyze the level of phenolic compounds at each step. On the basis of this the amount of extracted polyphenols was calculated. Further purification was done with ethyl acetate with varying percentage of hydrochloric acid and trichloro acetic acid (1, 2 and 3 percentage)[7].
For the evaporation of the solvent rotary evaporator was used. Different temperature treatments were given for the study (35, 45, 55, 65 and 75°C).
Quantification of polyphenol and catechins: Total catechins were analyzed using the modified spectrophotometric method of Swain and Hillis[6] and the total polyphenols were estimated by using the procedure of Dev Choudhary and Goswami[8].
Catechin fraction analysis by HPLC: The catechin molecule EGCG was extracted and analyzed using HPLC (Hewlett Packard series 1100, USA) fitted with phenomenex column by following the ISO method ISO/CD 14502-2[9]. An authentic standard of epigallo catechin gallate (Sigma-Aldrich, USA) was used in this study for comparison and for spiking test. Relative distribution of these constituents was expressed in percent individual component (w/w) according to ISO method.
Percentage of the extraction was analyzed by using the formulae ES = (100/native level of phenolic compounds)*(native level of phenolic compounds-total extraction)[10].
Young cut leaves yielded higher amount of polyphenols than mature leaves. This is due to the presence of high phenolic content in young tissues. When the leaves are cut, the contact of the cell surface and the solvent is maximum, also it submerges into the medium completely[11]. The native level of phenolic compounds revealed that the total polyphenol constitutes two third of catechins (Table 1). Yield of the extract was high in the frozen leaves, followed by drying the leaves in hot air oven (Table 2).
| Table 1: | Native level of secondary metabolites in tea clone UPASI 9 (%) |
| Table 2: | Leaf conditions for the complete extraction (%) |
 | |
| Table 3: | Different types of solvent processing for extraction |
 | |
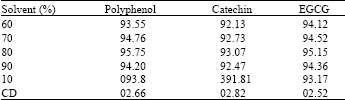
| Table 4: | Percentage of methanol for extraction |
 | |
Lower amount of extract was recorded in the leaves processed under room temperature. This is due to the action of oxidizing enzymes like polyphenol oxidase and peroxidase, which oxidize catechins into theaflavins and thearubigins which reflected in the lower amount of phenolic compounds. Enzymes were active in certain period of time and also some of the phenolic compounds were thermally degradable, so the extraction from dried samples was found to be low[12,13].
Hence samples were frozen and used for further studies. Refluxing of the extract followed by maceration gave good yield (Table 3). As per the broken cell wall theory the heat energy will increase the temperature inside the cell and the wall will rupture thus releasing the phenolic compounds[11]. The solvent mixture deactivated enzymes during maceration hence the thermal degradation method of extraction is not used[14].
Extractability of the solvent with the refluxing method was high when compared to maceration (Table 4). In the refluxing technique thermal degradation occurs but to a lesser extend. Still it has an edge over other methods like complete extractability, less amount of solvent, short duration and with high yield. To avoid the thermal degradation the temperature was maintained between 45°C to 55°C. The temperature was maintained by mixing hot water with the solvents[15].
The percentage of methanol played a vital role in the extraction. It was observed that 80 to 90% of the methanol gave high yield when compared to other mixtures.
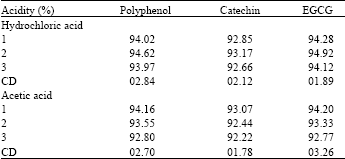
| Table 5: | Acidification of the extracting solvent |
 | |
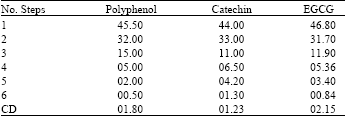
| Table 6: | Number of extraction with the same solvent (individual extracted amount) |
 | |
| Table 7: | Different purification tried using ethyl acetate |
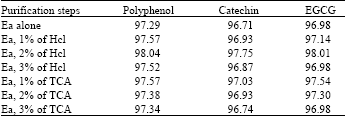 | |
| Ea: Ethyl acetate, Hcl: Hydrochloric acid, TCA: Trichloro acetic acid | |
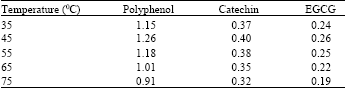
| Table 8: | Different temperatures tried to dry the samples under rotary evaporator |
 | |
Phenolic compounds dissolves in water by using the solvent in water and the extractability was also high[12]. Warm solvent mixture gave high yield when compared to others[4]. Moreover it has been reported that when the solvent polarity was modified by the addition of water, yield also increases[16]. Solvent with small quantity of acid gives high yield and normally acetic acid and hydrochloric acids were used for the present study. Among the different concentrations of acids tried, 2% Hcl in the solvent was superior to acetic acid (Table 5).
Number of extraction steps is an important part in the extraction of phenols. Chopped leaves were extracted with 80 percentage of solvent by using refluxer and it was collected separately. New solvent was added to the residue for further extraction. This has been continued for repeated extractions. There are several reports quoting that the efficiency of the extraction increases along with the number of extraction steps[17]. In the first extract the amount of the polyphenols was high followed by second and vice versa. Quantitative yield of polyphenols were obtained when 5-6 sequential extractions of the original leaf material was carried out. Almost 42% of the phenols were extracted in the first step and in the second it was 30%. After sixth extraction almost all the polyphenols were extracted (Table 6). The maximum amount of the extract was collected in the first three steps and the content was very low in the final step (Table 6). All the collected supernatants were mixed and filtered through Whatman No. 1 filter paper.
For purification of the extract numerou steps were tried. Centrifugation is the best way to collect pure supernatant of the solvents containing the polyphenols. Polyphenol yield was high in high-speed centrifugation. Earlier reports also support the same[12]. After centrifugation the extract contained numerous non-phenolic compounds like fats, terpenes, pigments etc. Hexane was used for the removal of these compounds. Addition of hexane dissolved the liposoluble compounds[18]. In order to remove the other pigments in the extract, it was first treated with chloroform for dissolving chlorophyll and caffeine. Further washings were carried out with ethyl acetate, hydrochloric acid and trichloro acetic acid[19]. For efficient purification 2% hydrochloric acid was added to the ethyl acetate. Comparatively acidified ethyl acetate was highly suitable for the efficient purification when compared to ethyl acetate alone or with trichloro acetic acid and ethyl acetate mixture (Table 7). In all the extraction methods the final extract was concentrated by vacuum evaporation at low temperature (45°C), is recommended[10]. Although it is not advisable to dry the extract completely, further dissolution of compounds from the residue may be much more difficult and some degradation may occur. There are reports regarding the degradation of polyphenolic compounds when excessive temperatures were attempted[15]. The degradation of the compounds was high in the samples dried above 65°C and is comparatively low in the temperature below 45°C (Table 8). The evaporation time of the extract to concentrate was high in the case of low temperature treatments and it was low in the case of high temperature evaporation. The solvents used for extraction can be collected and reused for the same extraction. Normally the loss of solvents was high in the method using vacuum evaporator. By using hot water (<55°C) to the solvent mixture will reduce the loss of solvent[17].
The authors are thankful to Dr. N. Muraleedharan, Director, UPASI Tea Research Foundation, Tea Research Institute, Valparai for his constant encouragement and suggestions. We are also grateful to NMITLI of CSIR funded project, "Functional genomics in plants: Niche pathway engineering in tea". We are thankful for the timely help rendered by Mr. Rahul and Mr. M. Saravanan.