ABSTRACT
Pollen tube formation is a good and simple model of growth and development. During pollination, a pollen tube emerges from the hydrated pollen grain, penetrates the stigma surface, traverses the transmitting tract and finally reaches the ovule for fertilization. This complicated and tightly controlled process presumably involves signaling between the pollen tube and the pistil. In this review, several subjects about pollen hydration, pollen germination (in vivo and in vitro), pollen tube wall structure and growth, incompatibility and pollen tube growth, the role of Ca+2 in pollen tube growth, pollen tube and pistil interactions will be discussed briefly.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2005.702.710
URL: https://scialert.net/abstract/?doi=ajps.2005.702.710
INTRODUCTION
Pollen grains (from the greek palynos for dust or pollen) contain the male gametophyte (microgametophyte) phase of the plant. Pollen grains are produced by meiosis of microspore mother cells that are located along the inner edge of the anther sacs (microsporangia)[1]. The primary functions of male plant gametophyte are the production of two sperm cells and their transport within the pollen tube through the tissues of the style and ovary into the embryo sac in the ovule, where they participate in double fertilization. When the pollen grain released from the anther, it then begins another phase of its life. If the pistil is a compatible one, the pollen grain germinates and extrudes a tube[2]. The factors that control pollen germination and tube growth are of considerable interest for the comprehension of the metabolic processes involved in fertility and reproduction in plants and also for the development of molecular tools aimed at manipulating pollen tube growth for practical purposes. Pollen tubes are an excellent system for the study of polarized tip growth, cell movement, cell to cell communication[3], cell to cell recognition and signaling in plants.
Pollen tubes are formed by the vegetative cells and grow uni-directionally. Pollen tube elongation is a dynamic process in which pollen tubes navigate and respond to female tissues to accomplish their mission of delivering the sperm cells for fertilization. Pollen tubes extend exclusively at the cell apex via an extreme form of polar growth, known as tip growth, producing uniformly shaped cylindrical cells. Pollen tube elongation can be extremely rapid, for example, reaching 1 cm h-1 in maize pollen tubes and growth is usually oscillatory[4]. Chemical gradients and structural features within the pistil have been previously proposed as factors determining the directionality of pollen tube growth[5].
Coupled with the advent of live-cell imaging techniques, investigations into the factors that affect pollen tube growth have reached an impressive level of detail. Although many of these recent investigations have focused on establishing the properties of normally growing pollen tubes, studies on the signals involved in the reorientation and inhibition of pollen tube growth have also received much interest. Other studies have focused on the specific inhibition of pollen tube growth during the Self-Incompatibility (SI) response[6].
Our knowledge of pollen tube growth, in particular the signals that regulate pollen tube growth and guidance, has advanced over the past 10 years or so. In this review, several topics will be discussed that are important for an understanding of the major functions of the male gametophyte in the production of the pollen tube and the conduction of the sperm cells within the tube to the embryo sac. These topics include pollen hydration, pollen germination (in vivo and in vitro), pollen tube wall structure and growth, incompatibility and pollen tube growth, the role of Ca+2 in pollen tube growth, pollen tube and pistil interactions.
POLLEN HYDRATION
Pollen hydration is usually tightly regulated and in vivo occurs only when desiccated pollen grains acquire water from the female, thus enabling pollen tube growth. In fact, in species exhibiting sporophytic self-incompatibility, the control of water uptake provides a mechanism for preventing self-pollinations. Despite this normally tight regulation of hydration (in vivo), it is nonetheless true that pollen will also hydrate and grow a pollen tube if placed in a simple medium containing sucrose, boric acid and calcium (in vitro)[7-10].
Several detailed analyses by Heslop-Harrison document the alterations that occur in the actin cytoskeleton of pollen as it hydrates and germinates[11], some of which may underlie developmental changes. For example, once it is hydrated, the pollen grain attains a distinct polarity and germinates to produce a pollen tube, which grows by tip extension. It is thought that pollen coat proteins, waxes and lipids initiate signals required for adhesion and germination. Indeed, mutations that provoke alterations in the waxy cuticle can result in defective pollen and impaired fertility. For example, the cer and POP-1 mutants, in which the mutant pollen fails to hydrate on wild-type stigmas[12,13], lack long-chain lipids that are present in the tryphine layer of wild-type pollen grains. These data suggest that long-chain lipids act as signals to stimulate hydration, a contention supported by evidence indicating that specific lipids play a role in providing a directional supply of water that is important for pollen tube penetration of the stigma[14]. In addition to lipids, there are also data implying that flavonols play an important role in pollen germination[15] .
In Arabidopsis thaliana, a self-compatible plant, once a pollen grain contacts a stigma papillar cell, the pollen coat, composed of lipids and proteins, softens into a gelatinous mixture and flows onto the papillar surface. This bridge between the pollen grain and papillar cell, sometimes termed a foot, establishes the route of water flow into the dessicated pollen grain. Pollen hydration is complete when the oblong shape of the dessicated pollen grain becomes round. As the pollen grain hydrates, Ca+2 flows into the pollen grain; this inflow, together with the shape change of the pollen grain, triggers cytoplasmic reorganization and the pollen grain is then activated for germination. This reorganization results in the formation of a cytoplasmic gradient of Ca+2 beneath the site of germination. When fully hydrated, the pollen grain deposits callose, a β 1,3-glucan, at one of the three pores, where the pollen tube will emerge[16].
POLLEN GERMINATION
Pollens normally germinate in stigmata of plants. In some plant groups they germinate in anther loculus (in situ). In situ pollen germination takes place as a result of genetic, morphologic and chemical factors[8]. Scientists observed that some of the species of Linum and Vicia, pollens germinate in anther loculus[17-19]. Pollen grains of many species germinate and grow in vitro. The required environment for pollen germination in vitro is related to genetic composition and also the quality and quantity of nutriment reserves of pollen[2,20]. Altough several attempts have been made to improve pollen tube growth on artificial media, no method is yet known which gives pollen germination and tube growth as good as that obtained in nature[10]. Pollen tubes growing in vitro do not achieve the high speed and length of in vivo pollen tube growth[21]. Pollen tubes growing in vitro lack the biochemical, physiological and physical environment of the pistil and the possible interactions with adjacently growing pollen tubes. During compatible pollen tube growth in vivo, considerable change in pollen tube growth rates and cytology may be observed[22], whereas the process in a corresponding in vitro pollen culture is essentially constitutive and constant. In vitro pollen tube growth studies are usefull for unraveling the mechanism of in vivo tip growth.
During the past few years pollen tubes grown in vitro became a popular model system for cell biology studies of signal transduction in plant cells[23]. Pollen grains are morphologically simple and the process of tube formation is a relatively uncomplicated example of growth and development. In vitro germination of pollen has been used as a powerful tool for genetical, physiological, biochemical and cytochemical studies for a wide range of plant species belonging to different families[11]. It might also be possible to select resistant pollen grains in vitro and after it to transfer germinated grains on the style of mother plant for artificial pollination. The possibilities of such an approach are experimentally shown for a number of plants[10]. Most studies on pollen tube growth were performed with in vitro or semi-in vivo cultures. Composition of the media and the pollen densities used for in vitro pollen tube culture vary highly among species.
In vitro germination assays showed that petunia (Petunia hybrida) pollen can germinate and grow not only in medium containing Sucrose (Suc) as a carbon source, but also in medium containing the monosaccharides Glucose (Glc) or Fructose (Fru). Furthermore, high-performance liquid chromatography analysis demonstrated a rapid and complete conversion of Suc into equimolar amounts of Glc and Fru when pollen was cultured in a medium containing 2% Suc. This indicates the presence of wall-bound invertase activity and uptake of sugars in the form of monosaccharides by the growing pollen tube. A cDNA designated pmt1 (petunia monosaccharide transporter 1), which is highly homologous to plant monosaccharide transporters, was isolated from petunia. Pmt1 belongs to a small gene family and is expressed specifically in the male gametophyte, but not in any other vegetative or floral tissues. Pmt1 is activated after the first pollen mitosis and high levels of mRNA accumulate in mature and germinating pollen. A model describing the transport of sugars to the style, the conversion of Suc into Glc and Fru and the active uptake by a monosaccharide transporter into the pollen tube is presented [24].
In vitro pollen germination and tube length studies are valuable in elucidating mechanisms (germination capacity and rate, tube growth rate) possibly associated with genetic differences in male transmission[25]. Many of the pollen-specific genes that have been identified[26] are expressed in mature pollen and mature pollen grains of many species contain all the mRNAs that are required for their germination and for pollen tube growth[1]. This has led to the suggestion that some pollen proteins may play an active role during germination and growth. Characterization of these late pollen-specific genes should help elucidate some of the processes required for pollen tube growth.
It has been documented that a lot of proteins and mRNAs were pre-synthesized during pollen maturation. Protein synthesis is initiated very rapidly during pollen germination. Within 2 min of transfer of ungerminated Tradescantia pollen to a growth medium, there is a large increase in polysomes and a decrease in single ribosomes. Biochemical analysis showed, moreover, that the mRNAs, ribosomes and tRNAs required for germination are synthesized during pollen maturation and persist in the pollen grain until they are utilized for translation during the germination process[2]. It was recently reported that pollen-specific sequences encoding proteins with regulatory or signaling functions are transcribed during the earliest moments of pollen germination in petunia[27].
The ubiquitin/proteasome system is a major pathway of proteolysis in eukaryotic cells and may contribute to controlling the intracellular levels of a variety of short-lived regulatory proteins. Data on levels of free and conjugated ubiquitin in developing pollen from different plant species are contradictory. Callis and Bedinger found a developmentally related loss of free ubiquitin and ubiquitinated proteins in maize inbred line Ky21, so that the ubiquitin monomer was not detectable at pollen maturity[28]. A reduction in the free ubiquitin level also takes place in the mature pollen stage in olive. On the contrary, high levels of free ubiquitin and ubiquitin conjugates have been found in mature pollen from several plant species, including some maize inbred lines[29]. In vitro, GABA (Gamma-amino Butyric Acid) stimulates pollen tube growth, although vast excesses are inhibitory. The Arabidopsis POP-2 gene encodes a transaminase that degrades GABA and contributes to the formation of a gradient leading up to the micropyle. POP-2 flowers accumulate GABA and the growth of many POP-2 pollen tubes is arrested, consistent with their in vitro GABA hypersensitivity[30].
POLLEN TUBE WALL STRUCTURE AND GROWTH
The pollen tube wall is an extension of the inner, or intine, pollen wall and is composed largely of callose. The pollen tube extends by tip growth through the pistil, in order to deliver the sperm to the embryo sac[16]. There are three parts related to the pollen tube wall: the primary wall secreted in the growing tip, the callosic wall deposited behind the tip (secondary wall) and local callose depositions known as callose plugs at more or less regular distances behind the tip[31]. Pollen tube growth is restricted to the tip region. The fine structure of the pollen tube as it relates to wall synthesis has been reviewed several times. Figure 1 provides a diagrammatic picture of the important structural features near the tip region of a growing pollen tube (Fig. 1). The cytoplasm of the pollen tube in the non-growing region behind the tip contains an abundance of mitochondria, dictyosomes, endoplasmic reticulum and vesicles. The growing tip region, by contrast, contains large number of secretory vesicles and absence of other organelles[2].
The primary wall of the growing pollen tube is composed chiefly of pectic substances that are readily removed by pectinases. Pectin is present in the wall of the growing tips of various species, including tabocco and lily. Pectin was shown to carry mainly esterified epitopes, whereas acidic epitopes were less abundant[32]. After deposition, the esterified pectin polimers become de-esterified and polimers of acidic pectin are presumed to become rigid by dimerization via ionic interactions with Ca+2. Though (1,4)-β-D glucans always occur, the presence of pure cellulose microfibrils is disputed[33]. In addition to pectin and cellulose, Arabino Galactan Proteins (AGPs) were detected in the primary walls of tobacco and lily pollen tubes cultured in vitro[34]. AGPs are widely distributed and highly abundant extracellular proteins that are hypothesized to be involved in plant morphogenesis.
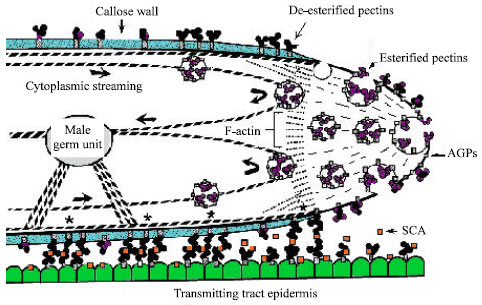 | |
| Fig. 1: | Diagrammatic picture of growing pollen tube tip[3] |
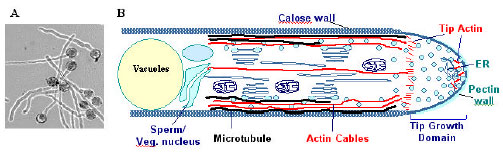 | |
| Fig. 2: | The pollen tube as a model system for cell polarity studies. A. In vitro-cultured pollen tubes show uniformly cylindrically-shaped cells. B. Schematics showing polar distribution of the cytoplasm in pollen tubes. Note the apex contains dynamic F-actin and Golgi vesicles[36] |
AGPs have been shown to accumulate spatially and temporally at different stages of plant development and to play a role during early tissue differentiation, in cell-cell communication and in cell-matrix interactions[31].
The tube growth process itself is driven by regulated intracellular conditions that maintain the appropriate ionic environment, actin dynamics and a balance level of exocytosis and endocytosis to support growth at the tube apex[4]. The growth of pollen tubes is characterized by an intense cytoplasmic streaming, during which the movements of smaller organelles (like secretory vesicles) and larger ones (including the generative cell and vegetative nucleus) are precisely coordinated. A well characterized cytoskeletal apparatus is likely responsible for these intracellular movements (Fig. 2). In recent years both microfilament and microtubule based motor proteins have been identified and assumed to be the translocators of the several organelle categories[32].
Microtubule based motor proteins of both kinesin and dynein families have been identified in the pollen tube. Most of these microtubule motors have also been found in association with membrane bounded organelles, which suggest that these proteins could translocate organelles or vesicles along microtubules. The biochemical features of these proteins are typical of the motor protein class. Although a number of microtubule motors have been identified in the pollen tube, the role of these proteins during pollen tube germination and growth or organelle movement is not yet recognized, as tube elongation and organelle movement in the pollen tube depend mostly on actin filaments[35].
Tradescantia cells were microinjected and used as an experimental system to study the behavior of pollen actin in vivo. It was demonstrated that purified pollen actin ameliorated the effects of injecting excess profilin into live stamen hair cells A vast array of actin binding proteins, together with intracellular signaling molecules, modulates the spatiotemporal distribution of actin filaments in eukaryotic cells. Pollen actin was purified by one cycle of polymerization and depolymerization. The recovery of functional actin by this rapid and convenient procedure was substantial; the average yield was 6 mg of actin from 10 g of pollen. Under physiological conditions, pollen actin polymerized with kinetics similar in quality to those for vertebrate -actin and had a critical concentration for assembly of 0.6 μM. Moreover, pollen actin interacted specifically and in a characteristic fashion with several actin binding proteins[11].
In Nicotiana tabacum, the movement of the vegetative nucleus and generative cell close to the tube tip can be thought to divide the function of the tube roughly into two parts: a tip part that contains the secretion machinery for primary wall formation and growth, with highly efficient exo- and endocytotic pathways and a basal part behind the vegetative nucleus and generative cell, where secretion is more directed to secondary wall thickening, callose plug formation and vacuolar biogenesis[37].
Most importantly, a defined Plasma Membrane (PM) region of pollen tube must be established, to which post-Golgi vesicles are targeted to and fuse with a defined domain of the plasma membrane at the cell apex, leading to localized extension of the PM (from vesicle membrane) and cell walls (from contents of vesicles). The PM domain is referred as the ‘tip growth domain’. Because in vitro pollen tubes always extend by tip growth into uniformly shaped cells (e.g. pollen tubes from the same genetic background exhibit constant radial diameters), it is reasonable to assume that internal signals of some kind turn on tip growth mechanisms and define the radial diameter of the pollen tube by spatially regulating the tip growth domain. Similarly, it is anticipated that during in vivo growth, external guidance signals also spatially regulate the tip growth domain to redirect pollen tube growth[38].
INCOMPATIBILITY AND POLLEN TUBE GROWTH
Pollination precedes fertilization and refers to the landing and subsequent germination of the pollen on the stigma. Hence it involves an interaction between the gametophytic generation of the male (pollen) and the sporophytic generation of the female (stigmatic surface of the carpel). The arrival of a viable pollen grain on a receptive stigma does not guarantee fertilization. Interspecific incompatibility refers to the failure of pollen from one species to germinate and/or grow on the stigma of another species. Intraspecific incompatibility occurs within a species. Self-incompatibility is an example of intraspecific incompatibility; it blocks fertilization between two genetically similar gametes, increasing the probability of new gene combinations by promoting outcrossing (pollination by a different individual of the same species).
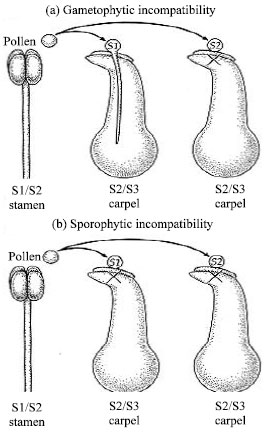 | |
| Fig. 3a: b: | Gametophytic incompatibility Sporophytic incompatibility in plant[46] |
Not all angiosperms are self-incompatible. Recognition of self depends on the multiallelic Self-incompatibility (S) gene[39,40].
S1, S2 and S3 are different alleles of the self-incompatibility locus. Plants with gametophytic self-incompatibility reject pollen only when the genotype of the pollen matches one of the carpel's alleles. In sporophytic self-incompatibility, the genotype of the pollen parent, not just the haploid pollen grain itself, can trigger an incompatibility response (Fig. 3).
Gametophytic self-incompatibility occurs when the S allele of the pollen grain matches either of the S alleles of the stigma (remember that the stigma is part of the diploid sporophyte generation, which has two S alleles). In this case, the pollen tube begins developing but stops before reaching the micropyle that leads into the female gametophyte. Sporophytic self-incompatibility occurs when one of the two S alleles of the pollen-producing sporophyte (not the gametophyte) matches one of the S alleles of the stigma. Most likely, sporophyte contributions to the exine are detected.
Style squashes and stylar grafts were used to examine the growth of Nicotiana alata pollen tubes in self-compatible and self-incompatible styles.
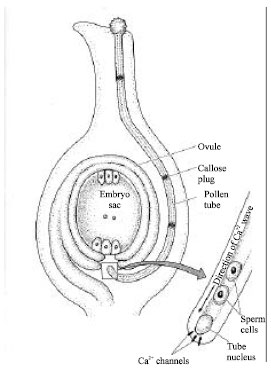 | |
| Fig. 4: | Growth of the pollen tube toward the micropyl[46] |
Compatible tubes typically showed a uniform layer of callose deposition in the walls and in small plugs spaced at regular intervals within the tube. Incompatible tubes were characterised by the variability of callose deposition in the walls and by larger, closer and more irregularly spaced plugs. Grafting compatibly pollinated styles onto incompatible styles showed that inhibition of pollen tube growth occurred in both the upper and lower parts of the transmitting tract[41].
Self-Incompatibility (SI) in Papaver rhoeas involves an allele-specific recognition between stigmatic S-proteins and pollen, resulting in inhibition of incompatible pollen. A picture of some of the signaling events and mechanisms involved in this specific inhibition of pollen tube growth is beginning to be built up. This highly specific response triggers a Ca2+ dependent signalling cascade in incompatible pollen when a stigmatic S-protein interacts with it. Rapid increases in cytosolic free Ca2+ concentration ([Ca2+]i) can now be attributed (at least in part) to Ca2+ influx. The rapid loss of the pollen apical Ca2+ gradient within ~1-2 min is accompanied by the inhibition of pollen tube tip growth[42].
THE ROLE OF Ca+2 IN POLLEN TUBE GROWTH
If the pollen and the stigma are compatible, the pollen tube grows down the style of the carpel towards the micropyle. The tube nucleus and the sperm cells are kept at the growing tip by bands of callose. Calcium has long been known to play an essential role in pollen tube growth[43]. Calcium accumulates in the tips of pollen tubes, where open calcium channels are concentrated[44, 45]. There is direct evidence that pollen tube growth in field poppy is regulated by a slow-moving calcium wave controlled by the phosphoinositide signaling pathway. After compatible pollen germinates, the pollen tube grows toward the micropyle. Calcium plays a key role in the growth of the tube (Fig. 4)[46].
Calcium is essential for pollen germination and pollen tube growth. A large body of information has established a link between elevation of cytosolic Ca2+ at the pollen tube tip and its growth. Since the action of Ca2+ is primarily mediated by Ca2+-binding proteins such as Calmodulin (CaM), identification of CaM-binding proteins in pollen should provide insights into the mechanisms by which Ca2+ regulates pollen germination and tube growth. A CaM-binding protein from maize pollen (maize pollen calmodulin-binding protein, MPCBP) was isolated in a protein-protein interaction-based screening using 35S-labeled CaM as a probe[47].
POLLEN TUBE AND PISTIL INTERACTIONS
During pollination, the pollen tube grows into the style and toward the ovary via extracellular matrices, collectively referred to as a Transmitting Tract (TT)[48]. The function of the TT during the progamic phase of pollen-pistil interactions is thought to be nutrition, attraction, guidance and signalling[31]. The transport of the pollen tube cell and the sperm cells involves a cell adhesion and migration event. Molecules involved in adhesion of the pollen tube to the TT cells in lily were studied and it was found that at least two molecules is required for this adhesion event. The first one was determined to be 9 kD, basic, cystein-rich protein with some sequence similarity to lipid transfer protein. The second one was found to be composed mainly of galacturonic acid (70-75 M%) with arabinosyl, galactosyl, rhamnosyl and glucuronosyl residues and reacts strongly with JIM 5 and slightly with JIM 7 (monoclonal antibodies to low esterified pectin, respectively)[48].
Several style-expressed genes, which may be involved in pistil development and/or pollen-pistil interactions, have been characterized at the DNA, RNA and/or protein level in different species. So far, based on sequence comparisons and on their (putative) function, these products can be divided roughly into three groups: proteins involved in SI (S-glycoproteins, i.e., S-Rnases), Pathogenesis-Related proteins (PR proteins) and a large group of proline-rich and hydroxyproline-rich glycoproteins. Enzyme activities and corresponding substrates in pollen and pistil probably play a role in pollen-pistil interactions and may determine germination, pollen tube growth and ultimately fertilization success[31].
The pollen tube enters the embryo sac through the micropyle and grows through one of the synergids. The two sperm cells are released and a double fertilization event occurs[49]. One cell fuses with the egg, producing the zygote that will develop into the sporophyte. (The zygote of the angiosperm produces only a single embryo; the zygote of the gymnosperm, on the other hand, produces two or more embryos after cell division begins, by a process known as cleavage embryony or polyembryony). The second angiosperm sperm cell fuses with the bi- or multinucleate central cell, giving rise to the endosperm that nourishes the developing embryo.
There has been controversy over whether a diffusible signal attracts the pollen tube or whether female tissues define its path. Emerging genetic and physiological data show that the female gametophyte produces at least two directional signals and that at least one of these signals is diffusible and derived from the two synergid cells[50].
The two synergid cells have been implicated in pollen tube guidance. The synergid cell is morphologically active in secretory functions and contains a relatively high concentration of calcium which can potentially control the direction of pollen tube growth. From the present observations, two synergid cells remained intact at the start of discharge by the pollen tube. It seems possible that both synergid cells actively secrete chemoattractants of pollen tubes until a tube reaches the synergid cells. Once the pollen tube begins to discharge, the receptive synergid cell might immolate itself for the succeeding transport of male gametes to their target female gametes. It has been proposed that the two sperm cells are transported along actin bundles in the embryo sac by actomyosin interactions[51].
Genetic approaches have been useful in investigating how pollen tube growth is guided toward unfertilized ovules. Arabidopsis, small weed in the mustard family, is used as a model system because of its very small genome. The pollen tube in Arabidopsis appears to be guided toward the ovule by a long-distance signal from the ovule[52]. Analysis of pollen tube growth in ovule mutants of Arabidopsis indicates that the haploid embryo sac is particularly important in the long-range guidance of pollen tube growth. Mutants with defective sporophyte tissue in the ovule but a normal haploid embryo sac appear to have normal pollen tube development.
While the evidence points primarily to the role of the gametophyte generation in pollen tube guidance, diploid cells may make some contribution. Two Arapidopsis genes, POP-2 and POP-3, have been identified that specifically guide pollen tubes to the ovule with no other apparent affect on the mutant plants[53]. These genes function in both the pollen and the pistil, thus implicating the sporophyte generation in the guidance system.
Pollen germination and growth are accompanied by a number of phenomena that appear to be rather complex and diverse among the many angiosperm species. During the past few years, there has been an almost explosive increase in the number of laboratories engaged in investigations of reproductive development in plants because of its relevance to plant biotechnology. Much of this interest has been focused on the male gametophyte. A large number of genes are expressed in pollen and appear to have functions in pollen tube growth. Thus far, no genes critical to the regulation of pollen development and function have been isolated. We hope that the present review will serve as a guidiance for further studies about the pollen germination. Recently, the approaches have become available that allow to more precisely determine the nature of this process that necessarily must occur at the borders between the male gametophyte and the sporophytic host tissues.
REFERENCES
- Franklin-Tong, V.E., 1999. Signaling and the modulation of pollen tube growth. Plant Cell, 11: 727-738.
Direct Link - Malhó, R., N.D. Read, M.S. Pais and A.J. Trewavas, 1994. Role of cytosolic free calcium in the reorientation of pollen tube growth. Plant J., 5: 331-341.
CrossRef - Park, S.Y., G.Y. Jauh, J.C. Mollet, K.J. Eckard, E.A. Nothnagel, L.L. Walling and E.M. Lord, 2000. A lipid transfer-like protein is necessary for lily pollen tube adhesion to an in vitro stylar matrix. Plant Cell, 12: 151-163.
PubMed








