ABSTRACT
Comparative epidermal studies using simple microscopy on herbarium and fresh fruit samples of three Abrus Adanson species (A. canescens Welw. ex Bak, A. precatorius L. and A. pulchellus Wall. ex Thw.) and a new collection of Abrus sp. occurring in parts of Tropical West Africa shows they are characterized by stomata, trichomes and other epidermal features. The epidermal cell shape in the species is irregular with straight anticlinal pattern in the new collection Abrus sp. but straight and arcuate in the other three. Observed differences in the dimensions of epidermal cell is such that a similarity of sort exists between A. pulchellus and the new collection Abrus sp. With ten stomata types observed in the species, stomata features are the most striking features. The restriction of sunken paracytic stomata in A. precatorius, brachy-para-hexacytic monopolar type in the new collection Abrus sp. and hexacytic and contiguous types in A. canescens, are reasonably diagnostic. Stomata in-groups of two occurring in clusters of between 10-13 were observed in A. pulchellus and A. canescens. Stomatal Index (SI) values are distinct for each species with values of 2.81 and 15.74 in Abrus sp. and A. pulchellus, respectively being the lowest and highest in the genus. The glandular multicellular and simple unbranched trichomes are present in all species as well as crystals encrusted in cells. The study aims at improving systematic information on the genus, which is hitherto scanty.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2005.652.659
URL: https://scialert.net/abstract/?doi=ajps.2005.652.659
INTRODUCTION
Abrus Adanson is a small pantropic genus belonging to the family Papilionaceae[1]. Its members are lesser known and utilized among the legumes[2] although great potentials for exploitation abound for the plant. Some members have been employed in ethnomedical, ethnopharmacological and toxicological uses[3-9]. The leaves and leafy twigs of some are used in the preparation of liquorice[10,11].
The re-emergence of medicinal plants as healthcare alternatives[12] calls for greater interest into poorly utilized plant genetic resources as these. In the face of increasing threat to these resources, mounting bioprospecting for new drugs of plant origin and general complacency on conservation, any effort at better understanding and proper documentation of plant genetic resources is worthwhile. Abrus species are potential genetic resources open to bioprospecting. The species are climbing, twining or scrambling slender sub-woody liana with pinnate leaves, rachis ending in a bristle, stamens connate in a sheath with racemose flowers[1]. Brettler[13] recorded four species in the genus while Verdcourt[14,15] reported thirteen. Labat[16] reported a new species A. longibracteatus Labat from Laos and Vietnam, while Thulin[17] published A. baladensis Thulin and A. gawwenensis from Somalia as species nova. In tropical West Africa, three species of Abrus, A. precatorius L., A. pulchellus Wall. and A. canescens Welw. ex Bak, have been reported by Hutchinson and Dolgiel[1]. At present A. canescens, which used to occur in the northern savanna area of Nigeria is almost extinct as efforts made to make collections for this study failed. The variability of the vegetative features and the conflicting reports of the exact number of species in this genus somewhat poses a bit of controversy on the taxonomy of the genus.
Epidermal characters as taxonomic tools have been extensively used amongst the Papilionaceae[18-21]. These studies, which utilized information from the structure, development and types of stomata, trichome and trichome types and various other quantitative and qualitative epidermal characters for taxa elucidation dwelt only on the leaf surface. Studies on the epidermal features of the fruit surface per se are generally uncommon. However, Olowokudejo[22] demonstrated the importance of epidermal characteristics of the fruit surface in the genus Biscutella where he reported remarkable topographic diversity in trichome and stomata features useful in taxa elucidation. Despite the enormous uses of Abrus species in Tropical West Africa[9], information on the systematics of the genus is scanty. Thus, the search for characters of taxonomic value has been extended to the fruit surfaces of the species of this genus as part of a multidisciplinary approach[23] to the systematics of the genus. The detailed descriptive qualitative and quantitative epidermal data amassed herein provides additional information on the taxonomy of the genus. Also included are the fruit epidermal characteristics of a new collection simply referred to as Abrus sp. collected from the Niger Delta area of Nigeria[23] whose identity is undergoing confirmation. We hope that findings will be invaluable to conservation and biotechnological exploitation of this plant.
MATERIALS AND METHODS
Fresh and herbarium specimens were used in this study (Table 1). Herbarium specimens were obtained from Forestry Herbarium Ibadan (FHI) and University of Ibadan Herbarium (UIH). Voucher specimens of all the living materials studied are deposited in the University of Port Harcourt Herbarium (UPH).
The fruit epidermis of Abrus species occurring in tropical West Africa were studied from fresh and herbarium samples (A. precatorius, A. pulchellus and the new collection Abrus sp.) fixed directly into the fixative formal acetic alcohol i.e. FAA (1 part formaldehyde, 1 part glacial acetic acid, 18 parts 70% ethanol v/v) and from herbarium materials only (A. canescens) (Table 1). Initial soaking in water for between 8-12 h revived the herbarium samples. The fresh samples were washed under tap water and together with the herbarium samples of A. canescens transferred into dishes containing 5% sodium hypochlorite (domestic bleach) for 12 to 24 h. Within this period, the sodium hypochlorite achieves the gradual digestion and disintegration of the hypodermal tissues of the fruit surfaces leaving a thin epidermal layer, which simply floats in the bleach solution or is carefully removed using a camel hair brush. The clear epidermal layers obtained were subsequently washed in several changes of distilled water, stained in 1% safranin for 1 min and temporarily mounted in glycerin. The preparations were observed and studied with a LEITZ microscope at objective lens 10 and 40 microscope magnification. Twenty microscope field views chosen at random from 5 fruit samples for each species were used to enumerate and study the number and types of stomata, trichomes and other epidermal cells. In each case, the median section of the fruit surface was used for the studies. Stomatal indices were computed according to Stace[24], stomatal type according to Dilcher[25], Metcalfe and Chalk[26], Stace[27], trichome morphology according to Metcalfe and Chalk[26], Sasikala and Narayanan[28], trichome density and distribution according to Olowokudejo[29]. The microscopic observations on these species were supplemented by ocular measurements of the length and width of epidermal cell, stomata and trichome at objective lens 40 on a LEITZ microscope. Standard deviation of the means in each case was computed as a means of comparison.
Photomicrographs were taken with a LEITZ DIAPLAN (Photomicroscope) fitted with LEICA WILD MPS 52 camera at objective lens 10 and 40 as indicated on the plates.
RESULTS
Cell shape: The epidermis of the fruits of the species show one type of cell shape, i.e. irregular (Fig. 1-4 and Table 2).
Anticlinal cell wall patterns: The anticlinal wall pattern in the species is straight to arcuate with only Abrus sp. possessing straight wall pattern (Table 2 and Fig. 1-4).
Cell size: This varied from 27.27±9.64x13.77±2.97 μm in A. precatorius to 61.40±43.92x13.77±5.61 μm in Abrus sp. (Table 2). A wide range of variation occurred in the epidermal cell length but epidermal cell width of the species were observed to exist within a somewhat close range of 10.80 – 29.70 μm.
Cell wall thickness: The cell wall thickness ranged generally from 1.35 to 8.10 μm. A. precatorius, A. pulchellus and A. canescens have closely related cell wall thickness. Mean epidermal cell wall thickness of 4.05±1.85 μm in Abrus sp. which is distant from the other species, was the highest.
Costal cells: The costal cells are parallel in arrangement, elongated, rectangular shaped, with long arms straight, arcuate or wavy and having straight to oblique end walls.
Epidermal cell number: The average epidermal cell number per field view varied from 175 in A. pulchellus to 304 in Abrus sp. It is 293 in A. canescens and 300 in A. precatorius.
Stomatal complex: Ten types of stomata were observed to adorn the fruit epidermis of the Abrus species.
| Table 1: | Sources of plant materials examined |
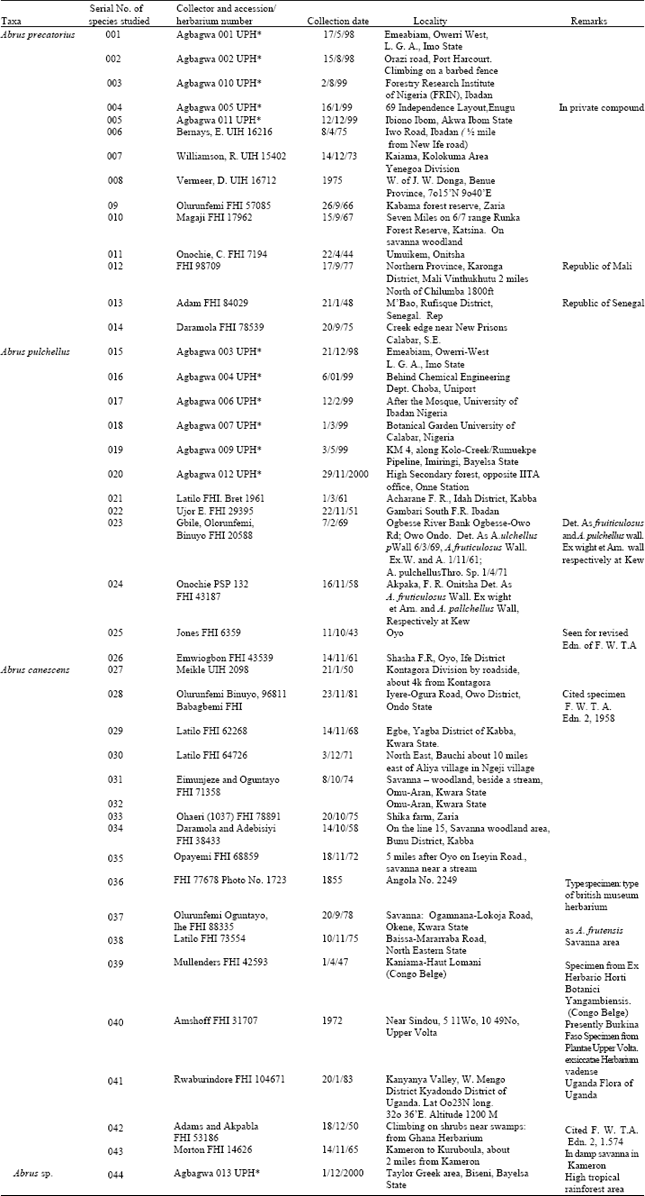 | |
| UPH*: Fresh specimens deposited at the University of Port Harcourt Herbarium | |
| Table 2: | Qualitative and quantitative epidermal cell characteristics of fruits of Abrus species studied |
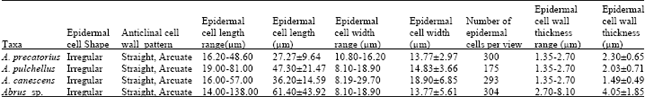 | |
| Table 3: | Qualitative and quantitative stomatal and trichome features of fruits of Abrus species |
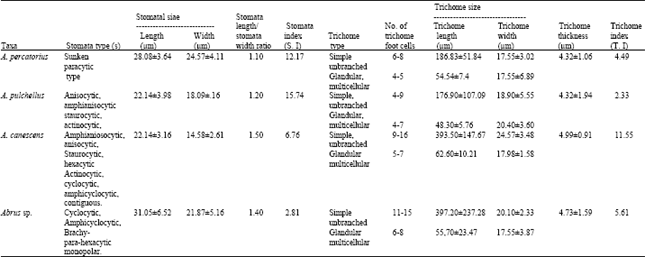 | |
These are paracytic, anisocytic, staurocytic, actinocytic, cyclocytic, para-hexacytic and brachy-parahexacytic–monopolar, amphicyclocytic, amphianisocytic (helicocytic) (Fig. 1-4). Also stomata in groups of two to thirteen occur in A. pulchellus and A. canescens.
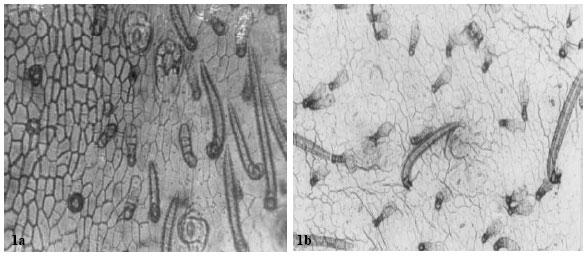 | |
| Fig. 1: | a: (x 560) showing fruit epidermises in A. precatorius. b: (x 560) notice the almost sunken stomata and the characteristic 2-3 glandular trichomes originating from one cell stalk |
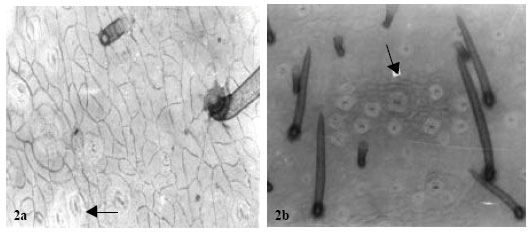 | |
| Fig. 2: | Showing fruit epidermises in A. pulchellus. a: ( x 875) Notice one glandular and one simple trichome. Arrow points to contiguous stomata. b: (x 560) arrow shows a parahexacytic stomata with a cluster of 12 other stomata. Notice the characteristic guarding of the stomata by several trichomes |
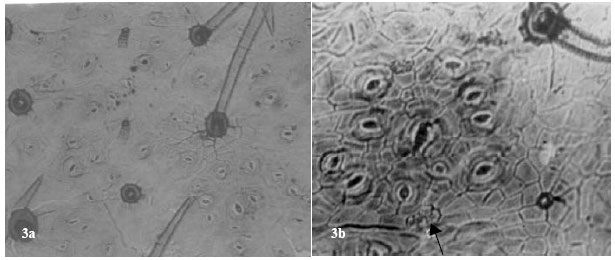 | |
| Fig. 3: | Showing fruit epidermises of A. canescens. a: (x 560) several cyclocytic stomata, glandular multicellular and simple unbranched trichome. b: (x 875) a large amphicyclocytic stomata surrounded by a cluster of 10 other cyclocytic types. Arrow shows 4 crystals encrusted into the cell |
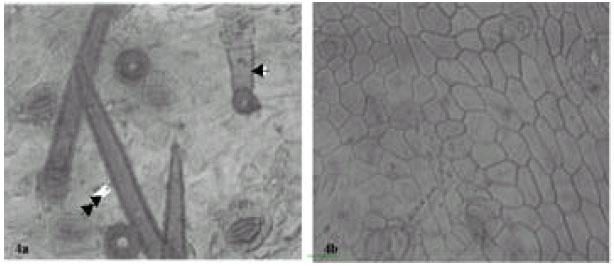 | |
| Fig. 4: | Showing fruit epidermises in Abrus sp. a: (x 875) arrow point to multicellular glandular trichome. Double arrow indicates simple unbranched trichome. b: (x 560) notice the thick epidermises wall |
Contiguous stomata occur in A. pulchellus and Abrus sp. Sunken paracytic stomata were observed in A. precatorius, amphianisocytic and para-hexacytic types in A. pulchellus and Abrus sp. Abrus pulchellus had the highest stomatal index of (15.74). The stomatal sizes of the species ranged from 22.14±3.16x14.58±2.61μm in A. canescens to 31.05±6.52x21.87±5.16 μm in Abrus sp. (Table 3).
Cell inclusion: Crystal sand and prismatic crystals are of common occurrence in the fruit surfaces of the species of Abrus. In Abrus sp., they occur ranging from cluster of four to several chains linked together (Fig. 4), but are few in the cells of the other species.
Trichomes: Two types of trichomes, glandular multicellular and simple unbranched, were observed on the fruit surfaces of all the species. In A. precatorius, 2-5 glandular multicellular trichomes often originate from the same cell stock. The highest trichome index of 11.55 for simple, unbranched trichomes in the species was recorded in A. canescens while A. pulchellus had the least trichome index value of 2.33 (Table 3).
DISCUSSION
Epidermal cell shape in the species is generally irregular with straight or arcuate anticlinal wall pattern (Table 2). In the new collection Abrus sp. a strict adherence to straight anticlinal pattern, which contrasts with the other three species where straight and arcuate types occur was observed. The quantitative results of the epidermal cell wall thickness follow the anticlinal wall pattern with affinity between A. precatorius, A. pulchellus and A. canescens, but distant values of 4.05±1.85 for Abrus sp. (Table 2). Ogundipe and Akinrinlade[21] reported high variability in the dimensions of epidermal cells of another leguminous genera Albizia and concluded that it was difficult to make clear distinction between the species using such character. However, we note that such a character can be made useful by amassing much quantitative data and making systematic comparison based on it, as was the case in this study. For instance, the seemingly high average mean values of 47.30±21.47x14.83±3.66 μm and 61.40±43.92x13.77±5.61 μm for the fruit surfaces of A. pulchellus and Abrus sp., respectively distinguishes them from the other two species (Table 2).
The ten types of stomata observed on the fruit surfaces of the species (Fig. 1-4 and Table 3) in one way or the other could be harnessed for taxonomic purposes. Sunken paracytic stomata, the only type observed in A. precatorius does not occur in any other species. Similarly, the brachy-para-hexacytic monopolar type occurs only in Abrus sp. while the hexacytic and contiguous types, which occur only in A. canescens, differentiates it from the others. Strikingly, all stomatal types in A. pulchellus and two out of the three in Abrus sp. were observed in A. canescens., however, A. pulchellus and Abrus sp. do not have any stomata in common (Table 3). In this study stomata types specific to species delimit those species while the common ones were indications of generic affinity. Since most stomata in the genus are represented in A. canescens, it is possible that in stomatal ontogeny of the genus the stomata of A. canescens are of earlier origin. Strong taxonomic relationship exists in stomatal types between these Abrus species and other members of the Leguminosae, where similar characters have been previously used in taxa elucidation.
| Table 4: | Morphology and density distribution of trichomes in Abrus species |
 | |
For instance, anomocytic, anisocytic, paracytic and cyclocytic stomata types, which occur in this genus, have been reported in other leguminous genera[18-21,26,30-32]. In these other genera, stomata types and in some cases their developmental patterns were variously utilized for taxa elucidation. Stomata in-groups of two occurring in clusters of between 10 and 13 as observed in A. pulchellus and A. canescens is a useful generic diagnostic character in some families, which in the Papilionaceae has only been reported in Euchestra[26]. This feature suggests taxonomic or evolutionary relatedness of Abrus to Euchestra, though the later does not occur in tropical West Africa. It also delimits these two species of Abrus from the others. Olowokudejo[22] successfully revealed remarkable topographic diversity in the genus Biscutella L. using fruit epidermal characters (stomata and trichome). Further applied these epidermal characters including anisocytic stomatal type (the only type in the genus) to achieve systematic classification of members of the genus[22]. In the present study, apart from achieving systematic elucidation of the genus, several of the stomatal features have been previously reported in other genera of Leguminosae, which implies taxonomic and evolutionary relationship to these genera.
Taxonomic variation on stomatal size and index useful in distinguishing species were observed on the fruit surfaces (Table 3). While Abrus sp. has an S.I of 2.81, in A. pulchellus it is 15.74. Edeoga and Osawe[19] and Ogundipe and Akinrinlade[21] reported the diagnostic significance of these characters in Senna and Albizia respectively. Carlquist[33] emphasized the contributions of the variation in stomatal size and index in delimiting species within a genus. High stomata index has been reported for the Leguminosae[19,32]. However, our observation in Abrus (Table 3) does not agree with these earlier reports. We expect this situation for a family as large as the Leguminosae with cosmopolitan distribution of species. For instance the reduced stomatal index and dimensions observed in A. canescens could be more of an adaptation to function. From distributional studies[23] A. canescens exists in the xerophytic savanna region of Nigeria. The glandular multicellular and simple unbranched trichomes were present on fruit surfaces of the four species, which depicts taxonomic and evolutionary relationship (Table 4). However, the ontogeny of the glandular multicellular heads differs in A. precatorius where 2-5 glandular heads originate anticlinally from one cell stock. In the other three species, each glandular head originates from one cell. Abubakar and Yunusa[20] recently demonstrated the importance of stomatal ontogeny in the taxonomic delimitation of species of Acacia, a leguminous genus. A. canescens was observed to have the highest values for simple unbranched trichome length (393.50±47.67 μm), glandular multicellular trichome length (62.60±10.21 μm), as well as trichome index (11.55), which according to Agbagwa[23] is related to the xerophytic habit of this species and the need to guide against water loss. Observations on trichomes in this study are similar to those of earlier authors on the Leguminosae that vividly applied and emphasized the taxonomic importance of these features in making reliable taxonomic conclusions[19-21,26].
REFERENCES
- Hutchinson, J. and M.J. Dalziel, 1958. The Flora of West Tropical Africa. 2nd Edn., Crown Agents for Oversea Governments and Admiistrations, Mulbank, London.
CrossRefDirect Link - El-Shabrawy, O.A., S. El-Gengaihi, N.A. Ibrahim and N. Ali-Ibrahim, 1987. Toxicity and teratogenicity of abrin. Egypt. J. Vet. Sci., 24: 135-142.
Direct Link - Reda, A.S., N.Z. Dimetry, S.A. Amer and H.M. Motawe, 1989. Activity of Abrus precatorius L. extracts and compounds isolated on orientation and oviposition behaviour of the two-spotted spider mite Tetranychus urticae Koch. Applied Entomol., 107: 395-400.
CrossRef - Hoareau, L. and E.J. da Silva, 1999. Medicinal plants: A re-emerging health aid. Electron. J. Biotechnol., 2: 56-70.
Direct Link - Dilcher, D.L., 1974. Approaches to the identification of angiosperm leaf remains. Bot. Rev., 40: 1-157.
Direct Link - Olowokudejo, J.O., 1990. Comparative morphology of leaf epidermis in the genus Annona (Annonaceae) in West Africa. Phytomorphology, 40: 407-422.
Direct Link - Carlquist, S.J., 1961. Comparative Plant Anatomy: A Guide to Taxonomic and Evolutionary Application of Anatomical Data in Angiosperms. Holt, Rinehart and Winston, New York, USA., Pages: 146.
Direct Link








