ABSTRACT
Previously isolated cDNA clones encoding glutamine synthetase (GS, EC 6.3.1.2) genes from asparagus and broccoli were observed that the level of transcripts decreased in both plant species during storage. In order to identify factors regulating the postharvest deterioration of another important Asian vegetable bamboo (Phyllostachys edulis) shoot, the present study was conducted to isolate glutamine synthetase (pBA-GS) cDNA clone from a cDNA library prepared from harvested bamboo shoot using reverse transcriptase-polymerase chain reaction. The partial cDNA clone encodes an mRNA of 832 bp and the derived amino acid sequence is highly homologous to GS from rice, barely and maize. Northern blot analysis showed decrease level of pBA-GS mRNA until 9 d at 20°C, which coincided well with enzyme activity and ammonia accumulation. Results demonstrated that GS activity decrease during the postharvest storage of bamboo shoot which is a highly perishable vegetable like asparagus spears and broccoli head and is likely to be a critical feature of their postharvest physiology.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2004.290.294
URL: https://scialert.net/abstract/?doi=ajps.2004.290.294
INTRODUCTION
Bamboo shoots are immature, expanding culms that emerge from nodes of the rhizome of bamboo plants. The edible part consists of meristematic tissues with regions of rapid cell division and differentiation, which is enveloped in protective, non-edible leaf sheaths[1]. We are investigating the early changes in physiology, biochemistry and gene expression during postharvest storage of highly perishable vegetables like asparagus spears, broccoli heads and bamboo shoots to identify factors regulating postharvest deterioration. Postharvest management of bamboo shoot which have a good analogy with asparagus spears, can be as crucial as pre-harvest management. It is previously reported that harvested asparagus spears accumulate ammonia in their tips towards the end of shelf-life and glutamine synthetase (GS; EC 6.3.1.2) is the primary enzyme responsible for assimilating ammonia in asparagus spears[2]. GS catalyses the ATP-dependent conversion of glutamate to glutamine utilizing ammonia as a substrate. Treatment of asparagus with phosphinothricin (PPT), a potent inhibitor of GS in plants, results in a rapid accumulation of ammonia in the spear tips and a reduction of shelf life[3]. Ammonia accumulation is thought to be a factor contributing to the perishability of harvested asparagus spears and occur due to alterations in GS activity after harvest. As ammonia is thought to be toxic to plant cells, the changes in ammonia content and GS gene expression in moso bamboo shoot in order to further understand the factors influencing ammonia assimilation during storage were studied.
Although the leaf cells of some other higher plant species, contain two isoforms of GS that are distinguished by their subcellular position: GS-1 is only present in the cytosol whereas GS-2 is exclusively detectable in the choloroplast matrix[4], the partial cDNA clone (pBA-GS) that was isolated from bamboo shoot showed higher homology to both GS-1 and GS-2 and total GS sequences from other species in the database. Therefore, the present study characterized the cDNA clone as pBA-GS and used as probe for Northern blot analysis of total GS without separation of cytosolic and plastid forms.
MATERIALS AND METHODS
Plant material: Moso bamboo shoots were harvested from farmer’s field in Kagawa Prefecture, Japan in April, 2002. The shoots were immediately brought to the laboratory (Faculty of Agriculture, Kagawa University, Japan) and stored at 1 and 20°C in perforated plastic bags for 0, 1, 3, 6 and 9 d in darkness. After storage treatment shoots were cut into small pieces (3 mm in thickness), weighed and immediately stored at –80°C until needed for GS and ammonia assay and for total RNA extraction.
Enzyme extraction: Approximately 5 g sample was homogenized under ice –cold condition (ca. 0-4°C) with 1% polyvinylpolypyrrolidone (PVPP), proportional to the sample weight and 1 g of sea sand in Buffer A by using a mortar and pestle. One ml extraction buffer per g fresh weight of plant materials was used. Extraction was performed according to the method of Hurst and Clark[5] in which Buffer A contained 50 mM Tris-Hcl (pH 7.6), 10 mM MgSO4, 1 mM EDTA, 1 mM dithiothreitol (DTT), 12 mM 2-mercaptoethanol, 5 mM Na L-glutamate and 100 ml L-1 glycerol. The homogenate was squeezed through a four-layer cotton cloth and the filtrate was centrifuged at 11000 x g for 10 min. The residual tissues were re-extracted in 5 ml Buffer A and dialyzed with 40 times dilution of the same buffer for 1 h and then centrifuged. The resulting supernatant were mixed together and used for the enzyme assay.
GS and ammonia assay: GS was measured in a total volume of 1.0 ml. The assay mixture contained 80 mM L-glutamate-Na, 500 mM Tricine-KOH Buffer (pH 7.0), 600 mM NH2OH, 200 mM MgSO4•7H2O, 10 mM Diethylenetriamine pentaacetic acid (DTPA), 80 mM ATP and 800 mM mercaptoethanol. After incubating at 35°C for 8 min, the reaction was stopped by adding 1 ml ferric reagent (25 ml FeCl3• 6H2O, 50 ml HCL and 20 ml TCA). GS activity was measured using a double beam spectrophotometer (Shimadzu model UV-150-02) at 540 nm.
For assay of ammonia 5 g samples from each portion were extracted with 10% Trichloroacetic acid and centrifuged at 11000 x g for 10 min as described by Kun and Kearney[6]. Ammonia content of the supernatant was determined from a triplicate 500 μl sample by adding 200 μl 0.5 M Tris-buffer (pH 8.0), 100 μl 0.1 M 2-oxoglutarate solution (pH 7.4), 30 μl 8 mM β–NADH solution and 150 μl distilled water. The absorbance was recorded at 340 nm against a reagent blank.
RNA isolation: Total RNA was extracted according to the Hot Borate method of Wan and Wilkins[7].
Amplification of poly (A)+ RNA by RT-PCR: The first strand cDNA was synthesized from 2 g of the total RNA by reverse transcriptase with Oligo-(dT) primer according to the instruction of SUPER SCRIPTTM Preamplification
System for First Strand cDNA Synthesis (GIBCOBRL, Tokyo, Japan). PCR was performed in a total volume of 25 μl containing the first strand cDNA reaction products, 10 x PCR Buffer, MgCl2, dNTP, First Start Taq DNA Polymerase (Roche) and primers. The primers (5'-TCNAGCACNGGGCARGCNCC -3' as the sense primer and 5'-TCRTTNCCYTCNCCRTANGC -3' as the antisense primer) were designed and synthesized on the basis of amino acid domains conserved in various GS genes. The Sal 1 and Not 1 restriction site sequences were also included at 5’-end of the sense and antisense primer, to facilitate cloning of PCR product. The PCR procedure started with 10 min at 95°C and was carried out 35 cycles of 30 s at 95°C, 30 s at 50°C and 30 s at 72°C and 10 min at 72°C with ASTEC Program Temperature Control System PC-700. The PCR products were confirmed by agarose gel electrophoresis.
Cloning and sequencing of cDNA: Sequence data analysis, synthesis of RNA probe and Northern blot analysis was performed as described in our previous reports[2].
GenBank accession number: The nucleotide sequence data reported in this paper will appear in the DDBJ/EMBL/GenBank nucleotide sequence databases with the accession number AB 162423.
RESULTS
GS activity: GS activity began to decline at both 1 and 20°C during the first 24 h storage (Fig. 1). After that activity at 20°C continued to decline until 3 days and remains almost unchanged thereafter. At 1°C, on the other hand, activity increased on the three days of storage and then decreased in 6 days. It again increased a little at the end of storage period. Enzyme activity was significantly higher in shoots stored at 1°C than those stored at 20°C. In other words, at 20°C GS activity declined to 80% of its level at harvest but at 1°C it declined by only about 12%.
GS gene expression: The cDNA pBA-GS is a partial clone encoding a glutamine synthetase transcript from moso bamboo shoot. The encoded mRNA is 832 bp long (Fig. 2) and is highly homologous to the glutamine synthetase gene of other plant species (Fig. 3). The pBA-GS sequence is 93.9% identical to GS from maize (D14578), 87.6% identical to GS from rice shoot (X14244) and 83.9% identical to the rice root GS1 (X14243) (Table 1). Allowing for conservative amino acid substitutions, the similarities are 99.3, 94.2 and 87.4% for the maize, rice shoot and rice root GS sequences, respectively (Fig. 4).
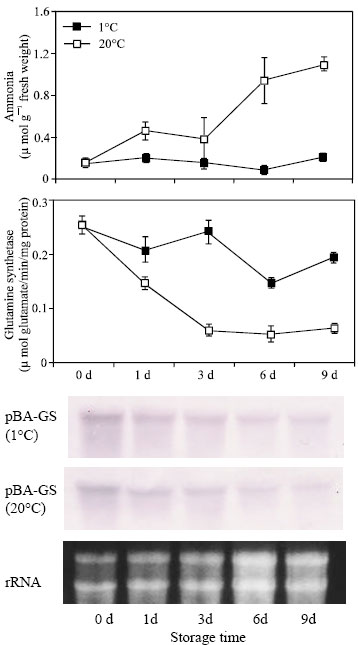 | |
| Fig. 1: | Changes in glutamine synthetase activity, ammonia accumulation and expression of pBA-GS during storage of bamboo shoot at 1 and 20°C. Each point represents the mean of three replicates and bars show SD about mean. Equal loading of RNA was confirmed by staining a gel with ethidium bromide |
The levels of transcripts for GS decreased after harvest at both 1 and 20°C (Fig. 1) and correlated well with enzyme activity. Expression of GS gene at 20°C was somewhat weak as compared to 1°C that may reflect in much lower retention of GS activity at that temperature.
Ammonia: A general increase in ammonia content for the shoots stored at 20°C was observed throughout the experimental period but no significant ammonia accumulation was observed in the shoots stored at 1°C (Fig. 1).
| Table 1: | Percentage of nucleic acid and amino acid identity between glutamine synthetase from bamboo and other plants in database |
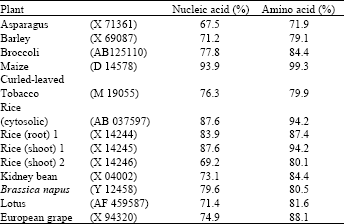 | |
| Bamboo GS (AB162423) was calculated as 100% | |
Ammonia accumulation increased to about 40% of initial level after 3 d of storage and reached about 70% of the initial level at the end of 9 d storage period.
DISCUSSION
Ammonia is thought to be toxic to plants and detoxified by the ammonia assimilating enzyme glutamine synthetase. In recent report with asparagus[2] it is shown that ammonia accumulation is a factor in the perishability of asparagus and it occurred due to changes in GS activity. In this experiment we have measured the ammonia accumulation and changes in GS activity in stored bamboo shoot and observed almost the same trend that was found in asparagus.
We have cloned and sequenced a 832 bp partial cDNA encoding pBA-GS in bamboo shoot. The comparison of amino acid sequence predicted for the protein encoded by pBA-GS to the polypeptide sequence of maize and rice GS showed that our bamboo cDNA clone encodes GS proteins. Our northern blot results suggest that induction of GS in harvested bamboo shoot is regulated by transcription of pBA-GS. The accumulation of ammonia in senescing leaves has been shown to coincide with almost complete disappearance of GS. In the present study we observed the same trend in which, GS activity and the level of transcripts for total GS declined in shoots stored at 20°C with an increased ammonia accumulation to about 70% after 9 d storage period. Hurst and Clark[5] reported 30% decline of total GS activity in harvested spear tips after 5 day storage at 20°C. The levels of plastid GS activity, polypeptide and mRNA all declined in the tips of harvested asparagus spears[4]. It has been reported that plastid GS has a role in assimilating ammonia produced during photo respiration in higher plants[8].
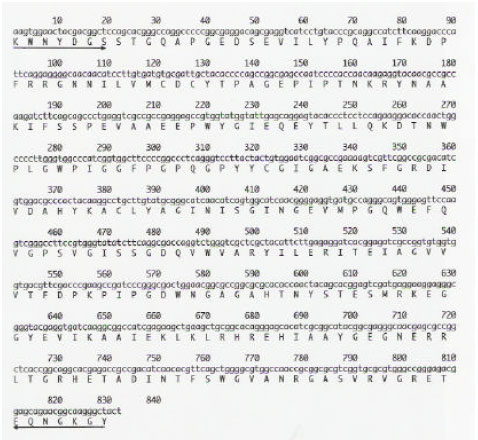 | |
| Fig. 2: | Nucleotide sequence and deduced amino acid sequence of the cDNA clone corresponding to pBA-GS. The predicted amino acid sequence is given in single-letter code for each amino acid. The arrows indicate the positions of degenerated primers (→sense, 7antisense) used for RT-PCR. Numbering refers to total nucleotide residues on each line |
 | |
| Fig. 3: | Comparison of the deduced amino acid sequences from barley (X69087), broccoli (AB125110), maize (D14578), rice cytosolic (AB037597), rice shoot (X14244, X14246) and rice root (X14243) by multi alignment. The amino acid residues are numbered at the beginning and end of the sequences on each line. Asterisks denote the amino acid residues that are identical. Dashes in the amino acid sequences represent gaps introduced to maximize alignment of the polypeptides |
 | |
| Fig. 4: | Phylogenetic tree of the alignment of pBA-GS deduced amino acid sequence with other glutamine synthetase in the database. Protein sequences were aligned using UPGMA and phylogenetic tree was constructed using GENETYX-MAC software |
Hurts et al.[3] reported that although about 65% of tip GS activity was lost when spears were held at 20°C ammonia accumulation was not due to limiting GS levels. They have studied postharvest inhibition of glutamine synthetase and used PPT 40 and 200 ppm which reduced GS activity by 50 and 80%, respectively, but did not increase ammonia accumulation. Ammonia accumulation was only enhanced, when greater than 95% of GS was inhibited. Thus, it seems that there is a critical level of GS activity necessary for the postharvest life of perishable vegetables and that under normal postharvest conditions, this critical level is exceeded and is more than the requirement of ammonia salvage. It is likely that ammonia accumulation results from a shortage of carbon skeletons required for its assimilation, i.e. the demand for respiratory carbon ultimately outstrips the availability of carbon for ammonia reassimilation and ammonia accumulation ensues.
Therefore, it can be concluded that the increase in ammonia content and decrease in total GS activity in bamboo shoot is a response to significant stress after harvest.
In summary, we isolated a partial cDNA clone encoding GS of bamboo shoot. Using this cDNA clone, it could be suggested that bamboo shoot having analogy with asparagus spears undergo ammonia accumulation during storage which is regulated by the low GS activity and the low induction of GS mRNA.
REFERENCES
- Bhowmik, P.K., T. Matsui, H. Suzuki and Y. Kosugi, 2003. Glutamine synthetase in harvested asparagus spears: Changes in activity and gene expression during storage. Asian J. Plant Sci., 2: 102-107.
CrossRefDirect Link - Hurst, P.L., G.A. King and W.M. Borst, 1993. Postharvest inhibition of glutamine synthetase activity with phosphinothricin reduces the shelf-life of asparagus. Postharvest Biol. Technol., 3: 327-334.
Direct Link - Downs, C.G., W.M. Borst, P.L. Hurst and D.G. Stevenson, 1994. Isoforms of glutamine synthetase in asparagus spears: The cytosolic enzyme increases after harvest. Plant Cell Environ., 17: 1045-1052.
Direct Link - Hurst, P.L. and C.J. Clark, 1993. Postharvest changes in ammonium, amino acids and enzymes of amino acid metabolism in asparagus spear tips. J. Sci. Food Agric., 63: 465-471.
CrossRefDirect Link - Wan, C.Y. and T.A. Wilkins, 1994. A modified hot borate method significantly enhances the yield of high quality RNA from cotton (Gossypium hirsutum L.). Anal. Biochem., 223: 7-12.
Direct Link








