Research Article
Effect of Waste Water Effluents From a Tannery on the Growth of Some Crop Plants
Not Available
T.Z. Bokhari
Not Available
A.A. Dasti
Not Available
S. Zaib-Un-Nisa Abidi
Not Available
The world today is facing with three main problems: ever-increasing human population, food shortage and environmental pollution. The last one is a serious problem in the developed countries while developing countries too have their share of it through their increased rate of industrialization and uncontrolled discharge into the water and air etc. Industrial wastes contain various poisonous salts, alkalies, acids, odor, gases, heavy metals, insecticides and pesticides. These polluted wastes are thrown into the canals, streams or rivers where they deteriorate the quality of water making the water unfit for irrigation purposes and for the use of animals. The harmful effects of effluents and waste products from different industries have been reported by Tripathi (1978), Chin et al. (1987) and Chadderton (1988).
In Pakistan, ways and means have to be adopted to combat the pollution of our environment which in the case of tanning industries, in addition to emissions of foul odors and toxic substances, is evident in the form of their wastewater effluents which are being thrown out relentlessly into the environment causing ecodisasters of varying types and magnitudes. Therefore, it is imperative to assess the quality of various wastewater effluents from industries and their effects on soils and vegetation. In Pakistan, very little effort has yet been directed towards this pressing objective (Sheikh and Irshad, 1980; Sheikh and Ahmad, 1981; Malik et al., 1999; Ansari et al., 2001). Much more detailed studies are obviously needed not only to determine the quality of industrial effluents, but also to investigate their effects on water, soils and vegetation.
In the present study attempts have been made to investigate the quality of wastewater effluents of Muzaffar-ud-Din Tannery and their effect on soil, seed germination and seedling growth of some crop plants. The findings will provide a basis for the further extensive study.
Samples of wastewater were taken from the out-fall of Muzaffar-ud-Din Tannery, situated on the Nawab-pur Road, Multan. The sample was collected in 10-liter capacity polyethylene containers from along the channel at randomly selected points. The wastewater was light-green in colour and had a bad smell. It contained a number of waste materials such as hair, pieces of skin and flesh etc. The wastewater was allowed to stand for sufficient time in the containers and the clear water above the residue was then used for analysis.
Wastewater sample was analyzed for sodium, potassium, calcium, magnesium, carbonates, bicarbonates, chloride, iron, manganese, electrical conductivity and pH. The values of Residual Sodium Carbonates (RSC) and Sodium Adsorption Ratio (SAR) were also calculated. Sodium, potassium and calcium were determined by Flame photometer. Magnesium, iron and manganese were determined by Atomic Absorption Spectrophotometer and the remaining determinations were made by following the methods of Richards (1954).
Following four treatments having three replicates were used to investigate the effects of wastewater on germination of Brassica campestris and Triticum aestivum:
| T1 | : | Tap water; |
| T2 | : | Wastewater as such; |
| T3 | : | 1:1 dilution, (wastewater: tap water) |
| T4 | : | 1:3 dilutions, (wastewater: tap water) |
Seed germination was studied by putting the seeds on filter papers placed in sterilized Petri dishes of 14 cm diameter. The filter papers were well moistened by using equal amounts of the respective waters. Twenty healthy seeds of Brassica campestris or Triticum aestivum were placed in each petridish. The petridishes were arranged on a laboratory bench in a completely rancomized block design. The positions of the dishes in a block were randomized each day. A seed was considered to be germinated when the radical had emerged from the seed coat.
The experiment was completed in 10 days. After the termination of the experiment, the seedlings were taken out from the petridishes and the excess of moisture was removed by drying them gently between the tissue papers. The length of plumule and radical of each seedling was measured and fresh weight of 10 randomly selected seedlings of a replicate was taken. Then the seedlings were dried in an oven at 80°C for 24 h and oven dry weight of each replicate batch of seedlings was determined. The data obtained on seed germination and seedling growth were subjected to statistical analysis.
Quality of wastewater: The results of the analysis of Muzaffar-ud-Din Tannery wastewater used in different treatments are presented in Table 1. The wastewater had higher amounts of sodium, potassium and chloride. The amounts of calcium and magnesium were also high. Among anions, carbonates were absent while the amount of bicarbonates was relatively high. As the amount of bicarbonates was less than that of calcium and magnesium, the water had no Residual Sodium Carbonates (RSC). The Tannery wastewater was found to be basic in reaction and had high value for electrical conductivity. Among trace elements only iron was present while manganese was absent in this water.
On the basis of electrical conductivity (EC) and Sodium Adsorption Ratio (SAR) values as criteria for the classification of water from the point of view of its quality (Richards, 1654) the wastewater of Muzaffar-ud-Din Tannery falls in C4-S1 class i.e., very high salinity and low sodium water.
 | |
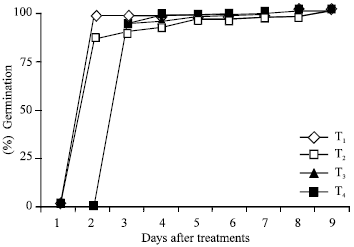
| Fig. 1: | Percentage germination of seeds of Brassica caperstris in different treatments of Muzaffar-ud-Din Tannery wastewater |
 | |
| Fig. 2: | Percentage germination of seeds of Triticum aestivum in different treatments of Muzaffar-ud-Din Tannery wastewater |
Seed germination: Fig. 1 and 2 shows changes with time in percentage germination of Brassica campestris and Triticum aestivum, respectively. The rate of seed germination in case of Brassica campestris was much faster in all the treatments and there was a delay in the start of germination. The rate of seed germination was relatively lower in T2 (wastewater as such) and T3 (1:1 dilution) as compared to T1 (tap water) and T4 (1:3 dilutions), while in case of Triticum aestivum, the rate of seed germination was initially faster in treatments T1 (tap water) and T2 (wastewater as such) than those in other treatments i.e., T3 (1:1 dilution) and T4 (1:3 dilutions).
| Table 1: | Analysis of wastewater sample form Muzaffar-ud-Din Tannery |
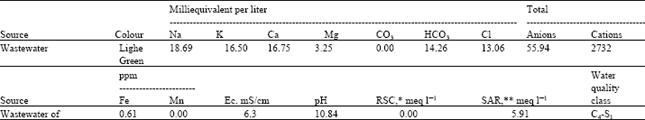 | |
| * Residual sodium sarbonate (RSC), ** Sodium adsorption ratio (SAR) | |
| Table 2: | Growth measurements per seedling of Brassica campestris grown in different treatments |
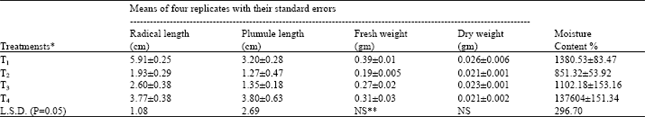 | |
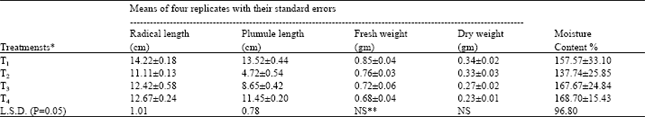
| * T1=Tap water; T2=Wastewater as such; T3=1:1 dilution of wastewater; T4= 1:3 dilution of wastewater NS**= Non significant | |
| Table 3: | Growth measurements per seedling of Triticum aestivum grown in different treatments |
 | |
Ultimately there were no significant differences in the total seed germination among the four treatments in either of the species.
Seedling growth: Significant differences were observed among the various treatments with regard to the radical and plumule length and moisture contents of seedlings in either of the species. The plumule length and moisture content of the seedlings of Triticum aestivum did not show significant difference in various treatments (Table 2 and 3). From the finding of thr present project we can say that wastewater as such was hazardous for seedling growth of Brassica campestris and Triticum aestivum. However, when it was diluted by mixing tap water to it, it resulted in the improved growth of plants of both the species as compared to that in the wastewater as such.
Irrigation water quality must be considered not only with regards to its immediate effect on soils and crops but also with regards to the welfare of consumers. Pesticides, pathogens and even some naturally occurring water constituents may not affect the crops directly but may affect animals or human beings and so are equally important criteria of water quality (Rhoades and Bernstein, 1971).
The wastewater of Muzaffar-ud-Din Tannery had toxic amounts of sodium and chloride (Table 1). The possibility of toxic effects, which might determine the choice of crop, should be attributed to the presence of greater amount of chlorides in wastewater. Higher contents of chlorides are dangerous for plants growth (Hayward et al., 1946). This water is being added in canal water used to irrigate the agricultural fields. The canal water with the addition of wastewater effluents carries a heavy load of sediments in addition to the eroded soils etc. that it already carries as suspended matter. The addition of this industrial residue to the water, besides its involvement in some chemical transformations in soil (after reaching there with the irrigation water), may result in silting-up of channels, etc. and hence cause problems of its own. Sediments in the water may also adversely affect surface soil structure by filling soil pores and decreasing permeability of air and water (Rhoades and Bernstein, 1971).
It was indicated above that the wastewater of the industrial plant is not being used directly for purposes of irrigation. Instead it was the canal water, into which the tannery wastewater effluent is being discharged, that is being used for irrigation purpose. Accordingly it was considered appropriate to investigate the effects of this wastewater and its dilutions on seed germination and seedling growth of Brassica campestris and Triticum aestivum, which are commonly grown crops in the area. The results generally indicate that as a whole the wastewater as such was found to be unfit for agriculture, although it had no significant effects on seed germination of crops like Brassica campestris and Triticum aestivum. However, when it was diluted by mixing tap water to it, it resulted in the improved growth of plants as compared to that in the wastewater as such. The lower growth of the seedlings might have been due to high osmotic pressure and high salt concentration, which prevented the intake of necessary water, or possibly, due to the toxicity of specific ions, as has been reported by Hayward and Wadleigh (1949).