ABSTRACT
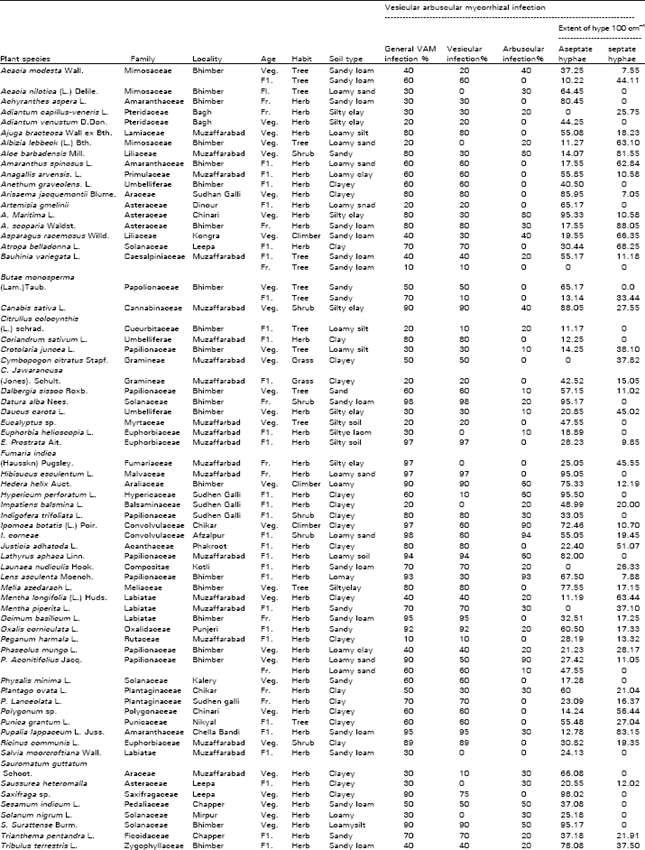
Seventy six medicinal plants were investigated for vesicular-arbuscular mycorrhizal (VAM) association in a survey of the Azad Jammu and Kashmir. Vesicular-arbuscular mycorrhiza was found to be of universal occurrence in all plants located at different habitats. Great variations were found in the VAM infection percentage and the extent of hyphal infection. Plants at vegetative stage exhibited more VAM infection percentage compared to those at flowering and fruiting stages. Herbaceous plants showed more infections in comparison with the shrubby and woody plants. The extent of root colonization by VA endohphytes varied with the soil type and plant species. Endophytes other than VA were also recorded.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2002.383.387
URL: https://scialert.net/abstract/?doi=ajps.2002.383.387
INTRODUCTION
The vesicular-arbuscular mycorrhizae are ubiquitous in roots of vascular plants in nature. (Harley and Smith, 1983; Powel and Bagyaraj, 1986; Gabor, 1992). They are common and wide spread among the plants of great economic importance growing on most of the soils (Nicolson, 1959). This wide distribution of VAM is due to obligate mycotrophy of the plants (Trappe, 1987). Indigenous nature to soil and distribution patterns of Endogonaceous fungi. The VAM fungi helps the plant in uptake of P, N, Zn, K, Cu, Sr, S and other mineral elements (Powel and Bagyaraj, 1984). The benefit of VAM fungi is due to an increased zone of nutrient depletion by mycorrhizal roots as compared to non-mycorrhizal ones (Owusu Bennoach and Wild, 1979).
The state of Azad Jammu and Kashmir is very rich in medicinal and aromatic plants. These medicinal plants occur naturally and most of them propagate vegetatively by under ground rhizomes, stems, bulbs or corms. This herbal wealth is being used not only by developing countries but also by developed countries for their health care systems. A bulk of our rural population relies on drug resources of plant origin. Locally collected plants are sold, where they are exploited commercially for preparation of medicines. The medicinal plants from some areas of Azad Jammu and Kashmir have been described by Haq and Hussain (1995), Bukhari (1996), Dastagir (2001), Shahzad (2001), Gorsi and Shahzad (2002).
However, no survey has so far been conducted on the mycorrhizal association of medicinal plants of Azad Kashmir. Therefore, an intensive survey of different areas of Azad Jammu and Kashmir was conducted in a preliminary attempt to observe the mycorrhizal associations of these plants.
MATERIALS AND METHODS
The medicinal plant species were sampled from different areas of Azad Jammu and Kashmir during the period 1999-2000. The areas of sampling included Bhimber, Chapper, Chikar, Chinari, Dhir Kot, Dinour, Leepa, Muzaffarabad, Pattika, Mirpur, Kotli, Punjeri, Afzalpur, Sudhen Gali, Bagh, Hevali and Nikyal. The plants were carefully dug up along with their rhizospheric soil in triplicate and transported to the laboratory in polythene bags. Plant roots were gently washed under tap water to remove soil particles. These roots were fixed in FA (formalin, acetic acid, rectified spirit, 5:5:90 by volume). These roots were cleared in 10% KOH by simmering at 90 °C for 1 hour. The KOH cleared roots were then stained in lactophenol following the procedure of Phillips and Hayman (1970). Stained roots were cut up into 1cm long pieces. Ten such stained root pieces were mounted in lactic acid on a microscope slide and were studied for VA mycorrhizal infections under the microscope. The extent of VA infections was calculated 100 cm-1 of roots.
Plants were pressed in presser papers and were dried carefully. The collected plants were identified with the help of available literature (Nasir and Ali, 1970-1987). Further identification was done by comparing the collected plant specimens with the Herbarium specimens available at the Department of Botany, University of Azad Jammu and Kashmir, Muzaffarabad.
RESULTS AND DISCUSSION
All medicinal plants in the present survey were found to be VA mycorrhizal (Table 1), despite the fact that they have an active principle in them, which is responsible for their medicinal value. Many of the medicinal plants have their active principle in under ground plant parts e.g., Arisaema jacquemontii and Valeriana wallichii (Table 1). Most probably, the mycorrhizal fungi can resist the presence of such compounds which the other common fungi can not. Earlier, the presence of VA mycorrhizal fungi has been observed in medicinal rhizomes (Iqbal and Nasim, 1986) and medicinal bulbs (Iqbal and Firdaus-e-Bareen, 1986).
The extent of root colonization by VA endophytes varied with the soil types and plant species. The percentage of VAM infections in the investigated plant species ranged from 10 to 98. Septate and aseptate hyphae of Atropa belladona forming coils (Fig. 1) and clamp -connections of Solanum surattense (Fig. 2) were observed in the root samples. Extent of aseptate hyphal infections varied from 11.17 to 95.57 100 cm-1, while the range of septate hyphal infections was 1.02 to 87.05 100 cm-1. Aseptate hyphae were counted more than septate hyphae. Profusely spread hyphae with average diameter ranging from 1-26 μ were noticed in most of the root samples. Two types of appressoria, dichotomously branched and single stranded with characteristic pegs entering the root cortex were also observed in Ajuga bracteosa (Fig. 3). Plants at vegetative stage were noted with higher VAM infection percentage than at flowering and fruiting stages. Vesicular and arbuscular infections were predominant at vegetative stage, while flowering with vesicles only which after fructification give rise to spores. Higher number of spores and low extent of hyphae were observed in plants at fruiting stage. Moreover herbaceous plants exhibited more VA mycorrhizal infection than shrubby and woody plants. Plants of woody nature restrict mycorrhizal infection frequently to herbaceous stages. This is probably due to the incorporation of mycorrhizal infection during more active stage, the herbaceous stage.
Vesicular infections ranging from 10 to 96% was of common occurrence, whereas arbuscular infection ranged from 10 to 60% and was less frequently observed. Vesicles of various dimensions (60-900 F2) and shapes i.e., spherical; Datura alba (Fig. 11) oval; Indigofera trifoliata (Fig. 8) rounded, oval-lobed, elongated; Ocimum basilicum (Fig. 4) dumbell-shaped and V-shaped were found in the root cortical cells. In some cases vesicles and spores of the genus Glomus, the most frequent VA mycorrhizal associate and most common of soil borne fungi were found intermixed within root cells. Terminal Eucalyptus sp. (Fig. 5) and intercalary vesicles containing oil droplets were frequently observed in many samples. Dichotomously branched arbuscules were seen in Xanthium strumarium (Fig. 6) and in some other plants. Collapsed network of arbuscules was also found in the root cortical cells.
| Table 1: | Vesicular arbuscular mycorrhizal status of some medicinal plants of Azad Kashmir |
 | |
 | |
 | |
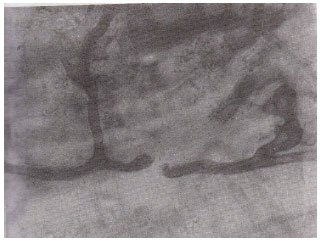
| Fig. 1: | Septate hyphae forming coils within root cortical cells of Atropa belladona. (X400) |
 | |
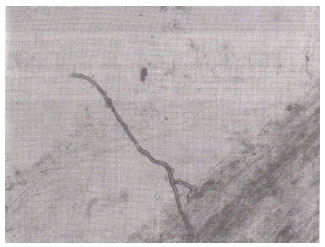
| Fig. 2: | Septate hyphae forming clamp-connections in the root cortex of Solanum surattense.(X400) |
 | |
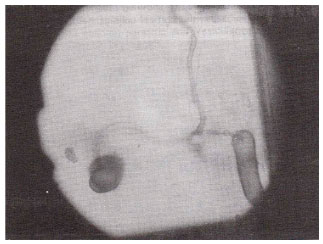
| Fig. 3: | A root segment showing dichotomously branched appressorium with entry point in Ajuga bracteosa. (X400) |
 | |
| Fig. 4: | Elongated and oval vesicles on the same mycelium in the root cortex of Ocimum basilicum. (X400) |
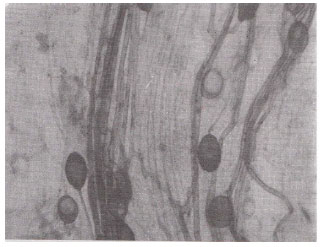 | |
| Fig. 5: | A portion of root showing hyphae with terminal vesicles in Eucalyptus sp. (X400) |
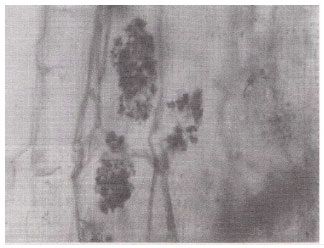 | |
| Fig. 6: | A root segment showing dichotomously branched arbuscules in the cortical cells of Xanthium strumarium. (X400) |
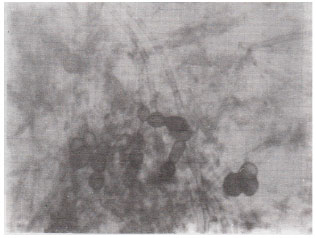 | |
| Fig. 7: | Septate mycelium with thick – walled, rounded and elongated spores in the root cortex of Coriandrum sativum. (X400) |
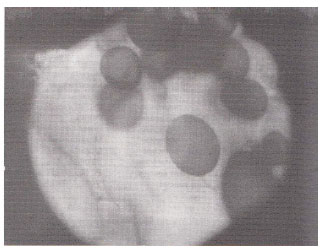 | |
| Fig. 8: | Large oval and terminal vesicles in the root cortex of Indigofera trifoliata. (X400) |
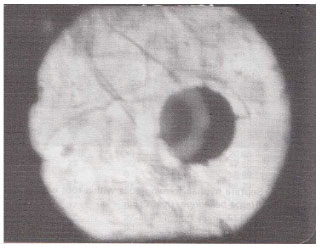 | |
| Fig. 9: | A sporocarp with several hyphal connections in the cortex of a root segment of Trigonella foenum– raecum. (X400) |
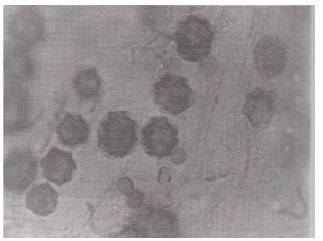 | |
| Fig. 10: | Star – shaped VAM spores within root cortical cells of Anagallis arvensis . (X400) |
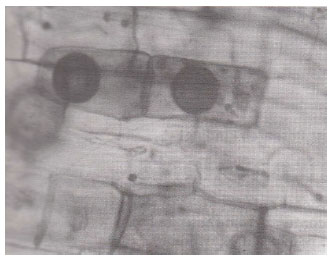 | |
| Fig. 11: | Datura alba , thick – walled spherical vesicles within the root cortical cells. (X400) |
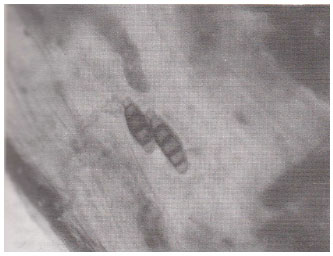 | |
| Fig. 12: | Xanthium strumarium, pathogenic fungus forming conidia within root cortex. (X400) |
Spores of various sizes and shapes; Coriandrum sativa (Fig. 7) were noticed individually and/ or in the form of root-attached sporocarpsl; Trigonella foenum-graecum (Fig. 9) mostly in mature plants of which rounded oblong, echinulated and star-shaped; Anagallis arvensis (Fig. 10) double wall spores were most pronounced. The diversity of vesicles and spores indicates the multiple VA infections. The invasion of roots by fungi other than VA endophytes; Xanthium strumarium (Fig. 12) was also observed. Variation in the VAM infection percentage may be attributed to differences in physico-chemical and biological characteristics of the soil including their VAM inoculum. The incidence of mycorrhizal colonization may very with season, soil moisture availability (Staffeldt and Vogt, 1975), habitat (Miller, 1979) and composition of a plant community (Hirrel et al., 1978 and Read et al., 1976).
These findings confirms the statement that mycorrhize are of universal occurrence (Micolson, 1967), beneficial for plant (Mosse, 1973 and Tinker, 1975) and have a significant role in ecosystem functioning (Baylis, 1976).
REFERENCES
- Dastagir, G., 2001. Medicinal plants of Mai Dhani Hill, Muzaffarabad, Azad Jammu and Kashmir. Hamdard Med., 44: 29-35.
Direct Link - Gorsi, M.S. and R. Shahzad, 2002. Medicinal uses of plants with particular reference to the people of Dhirkot, Azad Jammu and Kashmir. Asian J. Plant Sci., 1: 222-223.
CrossRefDirect Link - Iqbal, S.H. and G. Nasim, 1986. Vesicular-arbuscular mycorrhiza in roots and other underground portions of Curcuma longa. Biologia, 32: 223-228.
Direct Link - Miller, R.M., 1979. Some occurrences of vesicular-arbuscular mycorrhiza in natural and disturbed ecosystems of the red desert. Can. J. Bot., 57: 619-623.
Direct Link - Nicolson, T.H., 1967. Vesicular-arbuscular mycorrhiza: A universal plant symbiosis. Sci. Prog., 55: 561-581.
Direct Link - Phillips, J.M. and D.S. Hayman, 1970. Improved procedures for clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection. Trans. Br. Mycol. Soc., 55: 158-161.
CrossRefDirect Link








