ABSTRACT
Seeds from one triazine-resistant and two triazine-susceptible giant foxtail seed accessions were collected from plants grown in a greenhouse to characterize their germinability. Plants were grown in a greenhouse during mid to late summer and again during late autumn. Differential plant development from planting to first flowering among biotypes was not evident. Seed production of the susceptible biotypes either equaled or exceeded that of the resistant biotype. After harvest, seeds were placed under stratification involving 4oC, darkness, and moisture. Seed germination assays conducted after 0 and 4 weeks stratification resulted in very low percent germination for all biotypes. After 10 weeks stratification and subsequent germination assays, the resistant biotype had a lower percent germination than either susceptible biotype. Germination rates were similar among biotypes.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2002.334.336
URL: https://scialert.net/abstract/?doi=ajps.2002.334.336
INTRODUCTION
Triazine-resistance has been reported in more than 55 weed species (Le Baron and Mc Farland, 1990) and more than five have been characterized for their germinability. Germination differences between resistant (R) and susceptible (S) biotypes of a species varied among species. Darmency and Pernes (1989) reported a lower percent germination for triazine-resistant Setaria italica than for a genetically similar susceptible biotype. Triazine-resistant Brassica campestris was found to germinate later but had similar final percent germination as the susceptible biotype (Mapplebeck et al., 1982). Varied responses within weed families have also been documented. Triazine-resistant and susceptible biotypes of Amaranthus retroflexus had similar final percent germination and germination rate, while the triazine-susceptible A. powellii biotype germinated at a higher percent and faster than the resistant biotype (Weaver and Thomas, 1986). This variation in seed germination requires characterizing each triazine-resistant species separately to understand a triazine-resistant biotype's germinability relative to the wild biotype (Gasquez et al., 1981).
Giant foxtail (Setaria faberii L.) is one of the most significant weeds in Midwest corn and soybean crops (Holm et al., 1977, 1979 and 1997). The occurrence of triazine-resistant S. faberii (Ritter et al., 1989; Thornhill and Dekker, 1993) has been documented in the Midwestern U.S.
This study was conducted to characterize triazine-resistant and wild type S. faberii percent seed germination and rate of seed germination.
Materials and Methods
Plant material: A triazine-susceptible S. faberii seed lot from Ames, IA, accession 1816, has previously been characterized for dormancy requirements (Haar and Milton, 1995; Personal Communication) and was used as a reference for germination. A triazine-susceptible S. faberii seed lot, accession 1292, and a triazine-resistant S. faberi seed lot, accession 1299, were obtained from Pennsylvania (Wang et al., 1995). These three seed lots were termed triazine-susceptible 1 (S1), triazine-susceptible 2 (S2), and triazine-resistant (R), respectively, based -on chlorophyll fluorescence assays of each plant from which seed was collected (Thornhill and Dekker, 1993).
Plant growth: Plants were grown in a greenhouse (20-30 °C, natural light supplemented with 16 hr halogen lighting, and 60% humidity). Fertilizer (Water soluable, Peat-Lite special 20-10-20, Grace-Sierra Horticultural Products Company) was added every 10-14 days. Seeds were planted in 10 cm pots. For experiment 1, seeds were planted on 6 dates over a 22 day period. For experiment 2, seeds assayed for germination in experiment 1 were used. The seeds used for experiment 2 had germinated over a 38 day period and were transplanted from petri dishes to 10 cm pots at 2 to 6 days after germination. When plants were in the 3-5 leaf stage, they were transplanted to 1 plant per 13 cm pot.
Pollination bags were added to primary panicles at panicle emergence to insure self pollination and aid in collection of abscised seeds. The calendar date of first flower appearance on each primary panicle was recorded; therefore, a specific day after first flower (DAFF) may occur on different calendar dates for different primary panicles. First flowering indicates initial seed development.
Germination assay: Post-abscission seeds from primary panicles were collected every 5 days starting at 13 DAFF to ensure germination assessment soon after abscission. Each seed collection is termed a harvest. A cohort is a group of a biotype's primary panicles from which seed was collected at the same DAFF harvest. Seed harvest continued until seed rain of "dark" seed diminished. "Dark" seed had a fully expanded hull which is about 50% or more brown and no more than about 10% green. Seed harvest involves gentle tapping of panicle, removing of pollination bag, and placing new pollination bag on panicle. Both “dark” and “non-dark” seeds were counted upon collection.
“Dark” seeds only were selected for the three germination assays. These seeds were divided among three stratification treatments of 0, 4, and 10 weeks. Stratification involves storage of seed in the moistened petri dish at 4 °C and darkness, and began within 12 hours after harvest. For each stratification treatment and germination assay, a minimum of 10 seeds were placed on 5 cm filter paper in a petri dish 5 cm in diameter, moistened with 2 ml of water and maintained moist, and sealed in the petri dish with parafilm. After stratification, the petri dishes were placed in a germination cabinet (26 °C, continuous light) and seed germination was assessed daily for twelve days. The time required for seed germination to occur was recorded. Seed germination is defined by emergence of the coleoptile or coleorhiza from the hull or germination lid on the hull.
Data analysis: Data from experiment 1 and experiment 2 was analyzed separately because parent plant growth and development occurred during different times of the year. Results were determined for each biotype. “Dark”, “non-dark”, and total seed production of each primary panicle was recorded for each harvest. Each biotype had a minimum of nine primary panicles from which seed was collected. Percent seed germination and seed germination rate was determined for each primary panicle, a minimum of six per biotype.
Seed germination rate was determined by the mean germination rate (MGR):
MGR= Σ (TiNi)/N
where Ti is the days from placement of seed in germination cabinet until seed germination was recorded, Ni is the number of seeds germinated on dayi and N is the total number of seeds germinated (Darmency and Pernes, 1989). The average percent seed germination and seed germination rate for each biotype was determined. Standard errors were determined for individual biotypes.
Results and Discussion
Plant ontogeny: Time of plant development varied between experiments more than it varied among populations. No trend in differential plant ontogeny among populations was found (Table 1). In experiment 1, time from planting to first flowering for S1 was less than S2 and R. In experiment 2, R had the shortest development time from planting to first flowering followed by S2. S1 had the longest development time. Plant growth for experiment 1 occurred during mid to late summer while plant growth for experiment 2 occurred during late fall. Although greenhouse conditions were maintained at a relatively constant temperature and light amount, the warmer summer conditions may be the reason for quickened plant development.
| Table 1: | Plant development (days) from planting to first flowering (Mean ± S.E.) |
 | |
| Table 2: | Average number of seed produced per primary panicle (Mean ± S.E.) |
 | |
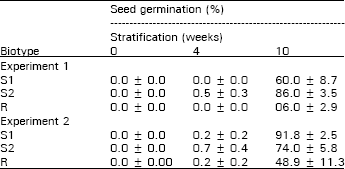
| Table 3: | Percent of dark seed germination after stratification periods (Mean ± S.E.) |
 | |
Seed production: In both experiments, “dark” and total seed production in susceptible biotypes either equaled or exceeded seed production of the resistant biotype (Table 2).
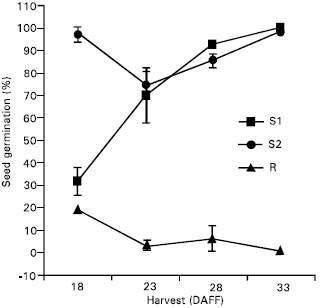 | |
| Fig. 1: | Percent (%) seed germination (Mean ± S.E.) of triazine-susceptible populations after 10 weeks startification for harvest dates of individual days after first flower (DAFF) in experiment 1 |
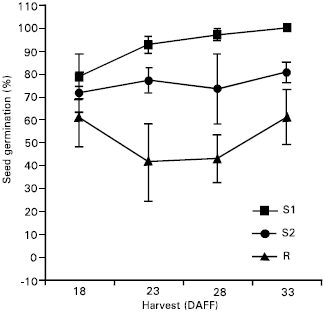 | |
| Fig. 2: | Percent (%) seed germination (Mean ± S.E.) of triazine-susceptible populations after 10 weeks startification for harvest dates of individual days after first flower (DAFF) in experiment 2 |
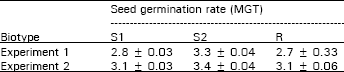
| Table 4: | Seed germination rate in mean germination time (MGT) of dark seed after 10 weeks stratification (Mean ± S.E.) |
 | |
In experiment 1, all biotypes produced an equal amount of total seed, while in experiment 2, S1 total seed production was greater than S2 and R. In experiment 1, S2 produced more “dark” seed than S1 and R which produced similar amounts. In experiment 2, both susceptible biotypes produced more “dark” seed than R. The results are consistent with Darmancy et al. (1989) who reported on two nearly isogenic S. italica populations, one triazine-resistant and one susceptible. The resistant S. italica population had a lower or equal total seed production than the susceptible population.
Seed germination: Percent seed germination was very low after 0 and 4 weeks stratification for both experiments (Table 3). After 10 weeks stratification, the percent germination increased and the R biotype had a significantly lower percent germination than either susceptible biotype. In experiment 1, the resistant biotype had a lower percent germination than S2 and S2 had a lower percent germination than S1. The resistant biotype again had a lower percent germination than the susceptible biotypes in experiment 2. The percent germination from lowest to highest was R < S1 < S2 in experiment 1 and R < S2 < S1 in experiment 2. For both experiments, the differences in percent germination between susceptible biotypes was less than the differences between the resistant biotype and either susceptible biotype. Since “dark” seed production was adequate at 18, 23, 28 and 33 DAFF, the percent seed germination was determined for each harvest as well (Fig. 1 and 2). The resistant biotype had lower percent germination than S1 and S2 at all harvests except the 18 DAFF in experiment 2. These results agree with Darmancy and Pernes (1989), who investigated on S. italica and found a lower percent germination for the resistant biotype compared with the susceptible biotype. Also, Garcia-Baudin and Aguirre (1983) reported that a triazine-susceptible Solanum nigrum ssp. Nigrum biotype lost dormancy before the resistant biotype.
Seed germination rate: Seed germination rates were determined in mean germination time in days. Germination rates were the same for R and S1 in both experiments. The germination rate of S2 was statistically different from S1 and R in both experiments, although the germination rate of S2 was only slightly greater (Table 4). All biotypes had mean germination times of about 2.7 to 3.4 days for both experiments. These results of similar germination rates are agreeable to Darmancy and Pernes (1989).
Differences in seed yield and percent germination between triazine-resistant and susceptible giant foxtail agree with some previous studies comparing triazine-resistant and susceptible plant species. The pleiotropic differences associated with triazine-resistant species may be the cause for such differences as suggested by Mapplebeck et al. (1982), or be due to changes in membranes in the resistant genotype (Pillai and St. John, 1981).
This investigation concurs but does not resolve that differences in seed yield or percent germination results from genetic differences associated between triazine-resistant and susceptible biotypes.
REFERENCES
- Mapplebeck, L.R., V.S. Machado and B. Grodzinski, 1982. Seed germination and seedling growth characteristics of atrazine-susceptible and resistant biotypes of Brassica campestris. Can. J. Plant Sci., 62: 733-739.
Direct Link - Pillai, P. and J.B.S. John, 1981. Lipid composition of chloroplast membranes from weed biotypes differentially sensitive to triazine herbicides. Plant Physiol., 68: 585-587.
Direct Link - Ritter, R.L., L.M. Kaufman, T.J. Monaco, W.P. Novitzky and D.E. Moreland, 1989. Characterization of triazine-resistant giant foxtail (Setaria faberi) and its control in no-tillage corn (Zea mays). Weed Sci., 37: 591-595.
Direct Link - Thornhill, R. and J. Dekker, 1993. Mutant weeds of Iowa: V. S-triazine resistant Setaria faberi herrm. J. Iowa Acad. Sci., 100: 13-14.
Direct Link








